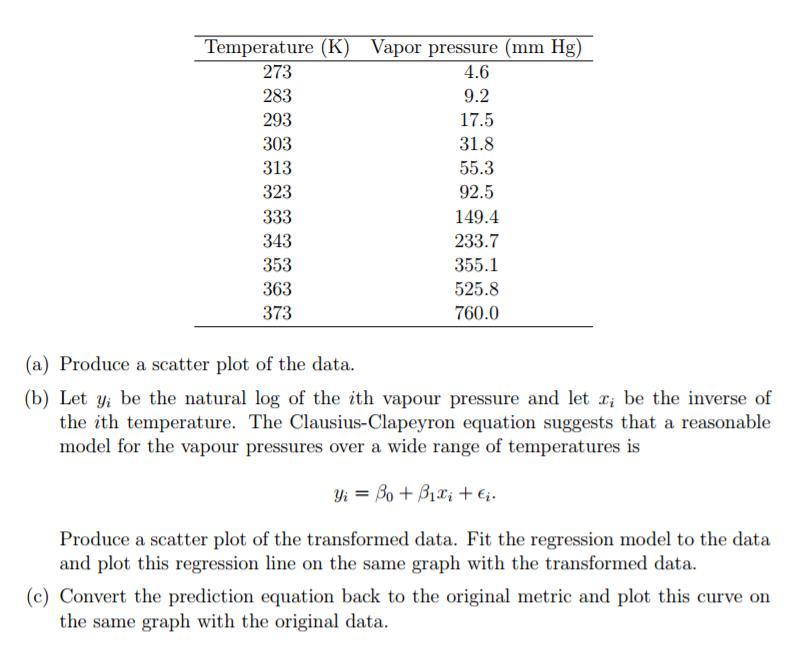
Chemical and mechanical engineers often need to know the vapour pressure of water for specific temperatures....
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
Chemical and mechanical engineers often need to know the vapour pressure of water for specific temperatures. Physical chemistry suggests that the vapour pressure should follow an exponential relationship to the inverse of the temperature. Specifically, let p, be the vapour pressure and T be the temperature, the Clausius-Clapeyron equation states that In p, is directly proportional to -1/T, that is 1 In p, ox T The following table lists the vapour pressures of water for various temperature (in Kelvin) from 0°C to 100°C. Temperature (K) Vapor pressure (mm Hg) 273 4.6 283 9.2 293 17.5 303 31.8 313 55.3 323 92.5 333 149.4 343 233.7 353 355.1 363 525.8 373 760.0 (a) Produce a scatter plot of the data. (b) Let y; be the natural log of the ith vapour pressure and let r, be the inverse of the ith temperature. The Clausius-Clapeyron equation suggests that a reasonable model for the vapour pressures over a wide range of temperatures is Yi = Bo + B1x; +6. Produce a scatter plot of the transformed data. Fit the regression model to the data and plot this regression line on the same graph with the transformed data. (c) Convert the prediction equation back to the original metric and plot this curve on the same graph with the original data. Chemical and mechanical engineers often need to know the vapour pressure of water for specific temperatures. Physical chemistry suggests that the vapour pressure should follow an exponential relationship to the inverse of the temperature. Specifically, let p, be the vapour pressure and T be the temperature, the Clausius-Clapeyron equation states that In p, is directly proportional to -1/T, that is 1 In p, ox T The following table lists the vapour pressures of water for various temperature (in Kelvin) from 0°C to 100°C. Temperature (K) Vapor pressure (mm Hg) 273 4.6 283 9.2 293 17.5 303 31.8 313 55.3 323 92.5 333 149.4 343 233.7 353 355.1 363 525.8 373 760.0 (a) Produce a scatter plot of the data. (b) Let y; be the natural log of the ith vapour pressure and let r, be the inverse of the ith temperature. The Clausius-Clapeyron equation suggests that a reasonable model for the vapour pressures over a wide range of temperatures is Yi = Bo + B1x; +6. Produce a scatter plot of the transformed data. Fit the regression model to the data and plot this regression line on the same graph with the transformed data. (c) Convert the prediction equation back to the original metric and plot this curve on the same graph with the original data.
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Given the following summary statistics, determine the regression equation used to predict y from z. By 1.65 = 82.17 L 0.11 F 21.29 76.17 Round all answers to 2 decimal places. slope y-intercept W...
-
The vapour pressure of a substance at 20.0DCis 58.0 kPa and its enthalpy of vaporization is 32.7 k] mol-1. Estimate the temperature at which its vapour pressure is 66.0 kPa.
-
The vapour pressure of a liquid in the temperature range 200 K to 260 K was found to fit the expression in (p/Torr) = 18.361 - 3036.8/ (TIK). Calculate the enthalpy of vaporization of the liquid.
-
Carbon has the electron configuration 1s 2s2p. The two unpaired electrons in the n = 2 level suggests that carbon will form two bonds. We know, however, that carbon forms four bonds in most compounds...
-
A piston-cylinder device contains 50 kg of water at 250 kPa and 25oC. The cross-sectional area of the piston is 0.1 m2. Heat is now transferred to the water, causing part of it to evaporate and...
-
Explain why the saving rate might have increased and its effect on the supply of loanable funds.
-
If the assembly fits snugly between two rigid supports A and C when the temperature is at T 1 , determine the normal stress developed in both segments when the temperature rises to T 2 . Both solid...
-
A manufactured product has the following information for June. Compute the (1) Standard cost per unit (2) Total cost variance for June. Indicate whether the cost variance is favorable or unfavorable....
-
A firm's stock sells for $25.00 and they recently declared a dividend of $1.25 per share. Their long term growth rate is 2.5%. What is the stock's expected dividend yield for the Not yet coming year?
-
The Sock Company buys hiking socks for $6 a pair and sells then for $10. Management budgets monthly fixed costs to be $12,000 per month. Required (consider each of the following questions...
-
Mailing a Package The post office will accept only packages for which the length plus the "girth" (distance around) is no more than 108 in. Thus for the package in the figure, we must have L + 2(x+y)...
-
Develop the appropriate primary research question to be associated with this design, including the research questions that will address main effects and interactions. Develop a hypothetical research...
-
Discuss the various control techniques that will be used with this specific design. Develop a hypothetical research scenario that would necessitate the use of an A-B-A Design. The research will be...
-
Develop the appropriate primary research question to be associated with this design. Develop a hypothetical research scenario that would necessitate the use of an A-B-A Design. The research will be...
-
Discuss the sampling strategy and technique used to access the appropriate case. Develop a hypothetical research scenario that would necessitate the use of an A-B-A Design. The research will be...
-
Discuss how the statistical procedures control technique will be used as a means to assign participants to each group. Discuss the rationale for this technique. Develop a hypothetical research...
-
Identify any events or outcomes tied to the political, legal or natural resource environments that create opportunities, or threaten efforts to market the insect-based food products in the U.S. now...
-
An 8.0 kg crate is pulled 5.0 m up a 30 incline by a rope angled 18 above the incline. The tension in the rope is 120 N, and the crates coefficient of kinetic friction on the incline is 0.25. a. How...
-
Use the Gibbs-Duhem equation to show that the partial molar volume (or any partial molar property) of a component B can be obtained if the partial molar volume (or other property) of A is known for...
-
Suppose that a molecular orbital has the form N (O.l45A + 0.844B). Find a linear combination of the orbitals A and B that is orthogonal to this combination.
-
The speed of a certain electron is 995 km S-I. If the uncertainty in its momentum is to be reduced to 0.0010 per cent, what uncertainty in its location must be tolerated?
-
With reference to Exercise 11.65, test the null hypothesis \(\beta=1.5\) against the alternative hypothesis \(\beta>1.5\) at the 0.01 level of significance. Data From Exercise 11.65 11.65 The data...
-
With reference to Exercise 11.65, construct a \(99 \%\) confidence interval for \(\alpha\).
-
The data below pertains to the number of hours a laptop has been charged for and the number of hours of backup provided by the battery. (a) Use the first set of expressions on page 330, involving...

Study smarter with the SolutionInn App


