Chemical reactions in foods that follow zero-order kinetics exhibit a constant rate of change in the...
Fantastic news! We've Found the answer you've been seeking!
Transcribed Image Text:
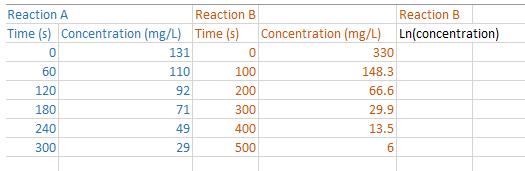
Chemical reactions in foods that follow zero-order kinetics exhibit a constant rate of change in the concentration of a reactant or a product. When the concentration is plotted against time, a straight line graph is obtained. The slope of the straight line gives the zero-order rate constant. On the other hand, many chemical reactions that occur during food processing and storage follow first-order kinetics. If we plot the concentration of a reactant against time we get an exponential type plot (i.e., a curve). The rate constant for a first- order reaction can be obtained by taking the natural logarithm of concentrations and then plotting those values against time. If the reaction follows first-order kinetics, then a plot of the natural logarithm of concentration against time should yield a straight line. Two sets of data are shown in the table below. a) Obtain a scatterplot of Concentration vs Time for Reaction A. (Be sure to put time on the x-axis). What trend do you see? Obtain a correlation coefficient. What does it tell you? What order of kinetics is being shown by Reaction A? b) Obtain a scatterplot of Concentration vs Time for Reaction B. (Be sure to put time on the x-axis). What trend do you see? What order of kinetics is being shown by Reaction B? c) Transform the concertation data for Reaction B by taking the natural logarithm. Obtain a scatterplot of Ln Concentration vs Time. What trend do you see now? d) Obtain a correlation coefficient for the relationship between Ln Concentration and Time. What does it tell you? Reaction A Reaction B Time (s) Concentration (mg/L) Time (s) 0 60 120 180 240 300 131 110 92 71 49 29 0 100 200 300 400 500 Concentration (mg/L) 330 148.3 66.6 29.9 13.5 6 Reaction B Ln(concentration) Chemical reactions in foods that follow zero-order kinetics exhibit a constant rate of change in the concentration of a reactant or a product. When the concentration is plotted against time, a straight line graph is obtained. The slope of the straight line gives the zero-order rate constant. On the other hand, many chemical reactions that occur during food processing and storage follow first-order kinetics. If we plot the concentration of a reactant against time we get an exponential type plot (i.e., a curve). The rate constant for a first- order reaction can be obtained by taking the natural logarithm of concentrations and then plotting those values against time. If the reaction follows first-order kinetics, then a plot of the natural logarithm of concentration against time should yield a straight line. Two sets of data are shown in the table below. a) Obtain a scatterplot of Concentration vs Time for Reaction A. (Be sure to put time on the x-axis). What trend do you see? Obtain a correlation coefficient. What does it tell you? What order of kinetics is being shown by Reaction A? b) Obtain a scatterplot of Concentration vs Time for Reaction B. (Be sure to put time on the x-axis). What trend do you see? What order of kinetics is being shown by Reaction B? c) Transform the concertation data for Reaction B by taking the natural logarithm. Obtain a scatterplot of Ln Concentration vs Time. What trend do you see now? d) Obtain a correlation coefficient for the relationship between Ln Concentration and Time. What does it tell you? Reaction A Reaction B Time (s) Concentration (mg/L) Time (s) 0 60 120 180 240 300 131 110 92 71 49 29 0 100 200 300 400 500 Concentration (mg/L) 330 148.3 66.6 29.9 13.5 6 Reaction B Ln(concentration)
Expert Answer:
Answer rating: 100% (QA)
A Obtain a scatterplot of Concentration vs Time for Reaction A Be sure to put time on the xaxis What trend do you see Obtain a correlation coefficient ... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Doing calculations with relevant formulae (from lectures) and calculator is worth 8 marks, and spreadsheet work is worth 4 marks. For part (a), the spreadsheet work requires presenting this as an...
-
A car accelerates from rest at a constant rate of 2.0 m/s2 for 5.0 s. (a) What is the speed of the car at the end of that time? (b) How far does the car travel in this time?
-
A tire 0.500 m in radius rotates at a constant rate of 200 rev/min, find the speed and acceleration of a small stone lodged in the tread of the tire (on its outer edge).
-
Write the surface x 2 + y 2 z 2 = 2 (x + y) as an equation r = (, z) in cylindrical coordinates.
-
Extract Corporation, a publicly traded mining company, acquires a mine at a cost of $500,000. Capitalized development costs total $125,000. After the mine is depleted, $75,000 will be spent to...
-
The Bureau of Labor Statistics reported that in 2016, the median weekly earnings for people employed full time in the United States was $837. a. What proportion of full-time employees had weekly...
-
Provide an example of a known liability, an estimated liability, and a contingent liability.
-
Glendo Industries' balance sheet at December 31, 2010, is presented below and on the next page. Additional information accumulated for the budgeting process is as follows.Budgeted data for the year...
-
Code the Following Report Read the following report and enter the proper code(s) in the blank(s) provided. Remember to code for ICD-10-CM, CPT, and HCPCS codes. If a specific category of code is not...
-
Noah and Joan Arc live with their family in Dayton, OH. Noahs Social Security number is 434-11-3311. Noah was born on February 22, 1983, and Joan was born on July 1, 1984. Both enjoy good health and...
-
2. Given the following matrix X and Y h X = = = 16 41 81 Y = [3 [3+ 6i l2-31 1-7i -1- 6il Based on the following MATLAB code, determine Z. Show your answer step-by-step. Z (X.\(Y'))*2+X
-
The yields of $1000 dollar par value zero-coupon government bonds (ignore liquidity premiums) are tabled below: Years to Maturity Yield to Maturity 1 4.00% 2 5.30% 3 6.1% 4 6.5% 5 7.85% The expected...
-
(8%) Problem 8: A point charge of 5.9 C is placed at the origin (x=0) of a coordinate system, and another charge of -1.6 C is placed placed on the x-axis at x = 0.28 m. 50% Part (a) Where on the...
-
Required Information [The following Information applies to the questions displayed below.] Manuel Company predicts it will operate at 80% of its productive capacity. Its overhead allocation base is...
-
Edit the Folder Redirection settings in the policy Open the Folder Redirection GPO for editing. Under User Configuration , browse to Policies\Windows Settings\Folder Redirection . Configure the...
-
Four charges QA 8.00 C, QB = -18.0 C, 16.0 C, and QD = -17.0 C are placed at the = 2.00 cm, d 5.00 cm, and = Qc = locations shown. Here, d = d3 = 5.00 cm. Vo = Calculate the electric potential Vo at...
-
Yummy Rice Cereal offers an all-star bowl in exchange for three coupons. Yummy Rice estimates that 30% of coupons will be redeemed. The bowls cost Yummy Rice $1 each. In 2016, 5,000,000 coupons were...
-
Difference between truncate & delete
-
(a) For a parallel plate capacitor with a fixed plate separation distance, a larger plate area results in (1) a larger capacitance value, (2) an unchanged capacitance value, (3) a smaller capacitance...
-
Modern bowling alleys have automatic ball returns. The ball is lifted to a height of 2.00 m at the end of the alley and, starting from rest, rolls down a ramp. It continues to roll horizontally and...
-
A steam engine operates between 100 oC and 20 oC. What is the Carnot efficiency of the ideal engine that operates between these temperatures?
-
The following table presents prices, in thousands of dollars, of single-family homes for some of the largest metropolitan areas in the United States for the third quarter of 2015 and the third...
-
Following are the closing prices of Google stock for each trading day in May and June of a recent year. a. Find the mean and median price in May. b. Find the mean and median price in June. c. Does...
-
Following are the weights, in pounds, for offensive and defensive linemen on the New York Giants National Football League team at the beginning of a recent year. a. Find the mean and median weight...

Study smarter with the SolutionInn App