9. The kinetics of the oxidation of ferrodoxin by oxygen were investigated in a careful series...
Fantastic news! We've Found the answer you've been seeking!
Question:
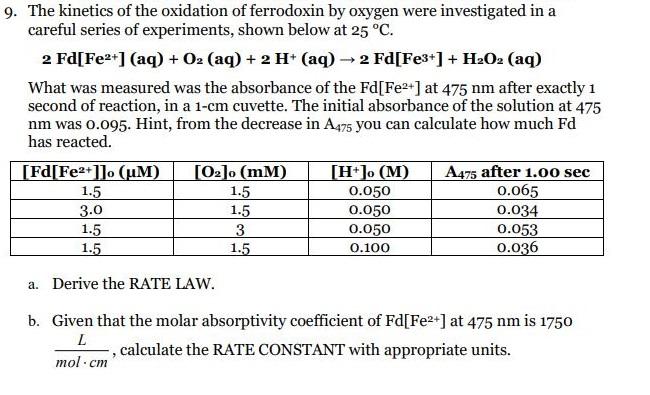
Transcribed Image Text:
9. The kinetics of the oxidation of ferrodoxin by oxygen were investigated in a careful series of experiments, shown below at 25 °C. 2 Fd[Fe2*] (aq) + O2 (aq) + 2 H* (aq) → 2 Fd[Fe3*] + H:O2 (aq) What was measured was the absorbance of the Fd[Fe2+] at 475 nm after exactly 1 second of reaction, in a 1-cm cuvette. The initial absorbance of the solution at 475 nm was 0.095. Hint, from the decrease in A475 you can calculate how much Fd has reacted. [Fd[Fe2+]]o (uM) [02]o (mM) [H*]o (M) A475 after 1.0o sec 0.065 0.034 0.053 0.036 1.5 1.5 1.5 3. 1.5 0.050 3.0 1.5 1.5 0.050 0.050 0.100 a. Derive the RATE LAW. b. Given that the molar absorptivity coefficient of Fd[Fe2+] at 475 nm is 1750 , calculate the RATE CONSTANT with appropriate units. mol - ст 9. The kinetics of the oxidation of ferrodoxin by oxygen were investigated in a careful series of experiments, shown below at 25 °C. 2 Fd[Fe2*] (aq) + O2 (aq) + 2 H* (aq) → 2 Fd[Fe3*] + H:O2 (aq) What was measured was the absorbance of the Fd[Fe2+] at 475 nm after exactly 1 second of reaction, in a 1-cm cuvette. The initial absorbance of the solution at 475 nm was 0.095. Hint, from the decrease in A475 you can calculate how much Fd has reacted. [Fd[Fe2+]]o (uM) [02]o (mM) [H*]o (M) A475 after 1.0o sec 0.065 0.034 0.053 0.036 1.5 1.5 1.5 3. 1.5 0.050 3.0 1.5 1.5 0.050 0.050 0.100 a. Derive the RATE LAW. b. Given that the molar absorptivity coefficient of Fd[Fe2+] at 475 nm is 1750 , calculate the RATE CONSTANT with appropriate units. mol - ст
Expert Answer:

Related Book For 

Introduction to Statistics and Data Analysis
ISBN: 9781337793612
6th edition
Authors: Roxy Peck, Chris Olsen, Tom Short
Posted Date:
Students also viewed these chemistry questions
-
College student attitudes about e-books were investigated in a survey of 1625 students. Students were asked to indicate their level of agreement with the following statement:? ?I would like to be...
-
A NaOH solution was 0.1019 M immediately after standardization. Exactly 500.0 mL of the reagent was left exposed to air for several days and absorbed 0.652 g of CO2. Calculate the relative carbonate...
-
What is the shear capacity of the RC beam described below considering the steel reinforcement and using the formula: VRsyAw 2fyd cot 8/s The shear reinforcement in the beam is provided by sets of...
-
Describe the Economic Analysis of the Valero Energy Corporation. Demonstrate Valero Energy Corporation is susceptible to Macroeconomic outlook both in the U.S.& foreign markets
-
Conduct a test of H0: μ1 ¥ μ2 2.3 versus Ha: μ1 Population Sample size Sample mean Sample standard deviation 13 50.3 21 58.6 7.23 6.98
-
Let R be a ring that contains at least two elements. Suppose for each nonzero a R, there exists a unique b R such that aba = a. a. Show that R has no divisors of 0. b. Show that bab = b. c. Show...
-
The following information pertains to a fire insurance policy in effect during the calendar year 2020, covering Vail Company's inventory: Vail's inventory averages \(\$ 500,000\) uniformly throughout...
-
The GASB has identied four classes of nonexchange revenues: Derived tax Imposed Government mandated Voluntary For each of the following revenue transactions affecting a city, identify the class...
-
ASAP. Use the information below to answer the two questions that follow. A company has two divisions: Division A and B. Division A Division B Total Sales $90,000,000 $7,000,000 $97,000,000 Variable...
-
Peyton Smith enjoys listening to all types of music and owns countless CDs. Over the years, Peyton has gained a local reputation for knowledge of music from classical to rap and the ability to put...
-
An overall conclusion on how covid-19 pandemic responses impacted the children's right to education in Scotland including aultism and the human rights law.
-
1-year, money market account investment at 2.17 percent? (APY), a 1.16 percent inflation? rate, a 15 percent marginal tax? bracket, and a constant ?$50,000 ?balance, calculate the? after-tax rate of?...
-
A car rental firm needs to relocate cars for the coming month. Three of the cities are projected to have a surplus of cars, and three are expected to have a shortage. The following table shows the...
-
A horizontal board of negligible thickness and area 4.0 m hangs from a spring scale that reads 60 N when a 2.0 m/s wind moves below the board. The air above the board is stationary. When the wind...
-
Courtney's insurance company has a capital structure of 45% debt, 15% preferred stock and 40% common stock. Its cost of equity is 12%, the cost preferred stock is 8%, and its cost of debt is 4%. The...
-
Selected current year-end financial statements of Cabot Corporation follow. (All sales were on credit; selected balance sheet amounts at December 31 of the prior year were inventory, $53,900; total...
-
When consumers research products online and then go to an actual store to make their purchase, the practice is known as Group of answer choices digital retailing. showrooming. webrooming. dual-line...
-
Identify the Critical Infrastructure Physical Protection System Plan.
-
Researchers were interested in comparing regularintensity exercise and high-intensity exercise for patients recovering from hospitalization due to chronic obstructive pulmonary disease (COPD). The...
-
Kyle and Lygia are going to play a series of Trivial Pursuit games. The first person to win four games will be declared the winner. Suppose that outcomes of successive games are independent and that...
-
Based on survey of a representative sample of 1000 adult Americans, YouGov estimated that the proportion of adult Americans who have less than $1000 in savings is 0.430 (People More Likely to Save...
-
Stonewall Corporation issued \(\$ 20,000\) of \(5 \%, 10\)-year convertible bonds. Each \(\$ 1,000\) bond is convertible to 10 shares of common stock (par \$50) of Stonewall Corporation. The bonds...
-
On January 1, 2020, Rocket Corporation issued \(\$ 250,000\) of \(6 \%, 20\)-year bonds at 98 . The interest is payable each December 31. Rocket uses straight-line amortization. The company's...
-
Dillon Corp. issued \(\$ 100,000\) of \(6 \%\) (cash payable each December 31), 10 -year bonds on January 1, 2020. The bonds are callable at any point after 2024 at 103. The bonds sold on January 1,...

Study smarter with the SolutionInn App


