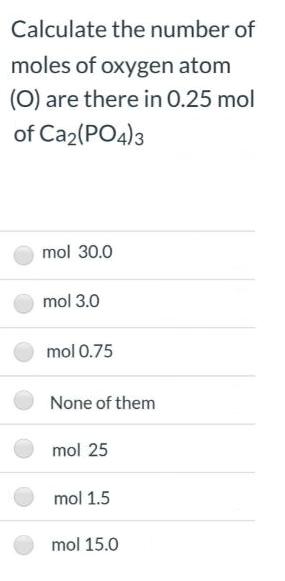
Calculate the number of moles of oxygen atom (0) are there in 0.25 mol of Ca2(PO4)3...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Calculate the number of moles of oxygen atom (0) are there in 0.25 mol of Ca2(PO4)3 mol 30.0 mol 3.0 mol 0.75 None of them mol 25 mol 1.5 mol 15.0 Calculate the number of moles of oxygen atom (0) are there in 0.25 mol of Ca2(PO4)3 mol 30.0 mol 3.0 mol 0.75 None of them mol 25 mol 1.5 mol 15.0
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Calculate the number of moles of HCl(g) that must be added to 1.0 L of 1.000 M NaC2H3O2 to produce a solution buffered at each pH. (Ka for HC2H3O2 = 1.800x10^-5) a) pH = pKa b) pH = 3.92 c) pH = 5.49
-
Calculate the number of moles of a gas that occupies a volume of 20 liters at a temperature of 25 C and 25 atm of pressure.n
-
Calculate the number of moles of barium chloride in 427 g of a 3.17% by mass barium chloride solution? a. 6.50 10 02 mol b. 7.83 10 02 mol c. 4.31 10 02 mol d. 7.81 10 02 mol e. 4.27 10 02 mol
-
Suppose the following game is repeated once (that is, played twice in total). Bridget Alex SB (40,40) (70,20) SA SA (20, 70) (30,30) (a) How many strategies does Alex have? (b) In an SPNE, what...
-
Briefly explain the five principal assertions that can be made about assets and liabilities, and auditors objectives related to each.
-
Comets travel around the sun in elliptical orbits with large eccentricities. If a comet has speed 2.0 X 10 m/s when at a distance of 2.5 X 1011 m from the center of the sun, what is its speed when at...
-
If the relevant price indices for trade accounts receivable and trade accounts payable are as follows, calculate the monetary working capital adjustment for Seafield Ltd, using the details given in...
-
James Albemarle created a trust fund at the beginning of 2016. The income from this fund will go to his son Edward. When Edward reaches the age of 25, the principal of the fund will be conveyed to...
-
Jack and Jill are married. This year Jack earned $75,500 and Jill earned $83,500 and they received $4,700 of interest income from a joint savings account. How much gross income would Jack report if...
-
Jiminys Cricket Farm issued a 30-year, 4.5 percent semiannual bond three years ago. The bond currently sells for 104 percent of its face value. The companys tax rate is 22 percent. a. What is the...
-
Wildhorse Inc. is a private company reporting under ASPE. The following selected account balances were reported in Wildhorse Inc.'s financial statements at year end: Cash Building Equipment Land...
-
A steel alloy has a melting temperature = 1450C, its density = 7.87 g/cm2, specific heat=0.46 /gC, and heat of fusion - 270 1/8 Assume specific heat has the same value for solid and molten metal. The...
-
The Sales Manager said he didn't have authority to match that low a price- Kevin would have to wait until Charles Cabot got home and ask him. The Sales Manager also reminded Kevin that Charles really...
-
Maxwell Inc. uses the periodic inventory system. During its first year of operations, Maxwell made the following purchases, listed in chronological order of acquisition: 40 units at $100 per unit 70...
-
Oriole Corporation manufactures safes-large mobile safes, and large walk-in stationary bank safes. As part of its annual budgeting process, Oriole is analyzing the profitability of its two products....
-
Working as an employee in a busy hospital, you overhear two of your co-workers in the public cafeteria discussing confidential patient information. You notice that other people in the cafeteria...
-
The following information relates to the Better Lawns and Gardens Company at the end of December 2020. The company is a publisher of a monthly lawn and garden magazine. The company's revenue comes...
-
Distinguish between the work performed by public accountants and the work performed by accountants in commerce and industry and in not-for-profit organisations.
-
Sodium-23 is the only stable isotope of sodium. Predict how sodium-20 will decay and how sodium-26 will decay.
-
Give the normal state (solid, liquid, or gas) of each of the following. a. Sodium hydrogen carbonate (baking soda) b. Isopropyl alcohol (rubbing alcohol) c. Oxygen d. Copper
-
An aqueous solution of an unknown salt of vanadium is electrolyzed by a current of 2.50 amps for 1.90 hours. The electroplating is carried out with an efficiency of 95.0%, resulting in a deposit of...
-
Assume that Protex Company, a U.S. company, is involved in petroleum operations in Thailand. Protex Company has a 40% WI, while the Local Oil Company has a 60% WI. Annual gross production is to be...
-
Jones Oil Company operates under a PSC agreement in the South China Sea. Jones has 49% of the working interest, and Sinhai Oil Company (which is owned by the Chinese government) has 51% of the...
-
Fortune Company enters into a risk service agreement with the Chilean government. Fortune pays the government, in U.S. dollars, a \($5,000,000\) signing bonus and also agrees to pay all of the costs...

Study smarter with the SolutionInn App



