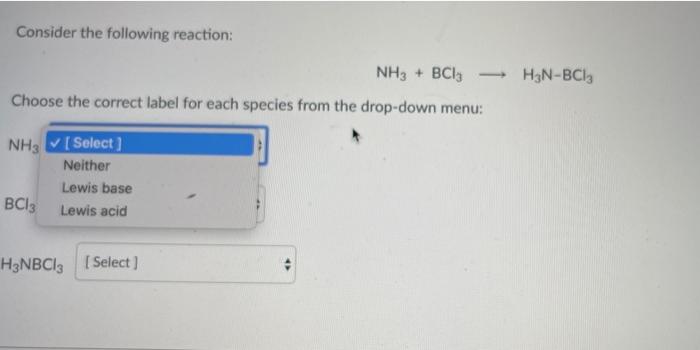
Consider the following reaction: NH3 + BCI3 H3N-BCI3 Choose the correct label for each species from...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Consider the following reaction: NH3 + BCI3 H3N-BCI3 Choose the correct label for each species from the drop-down menu: NH3 [Select] Neither Lewis base BCI3 Lewis acid H3NBCI3 (Select] Consider the following reaction: NH3 + BCI3 H3N-BCI3 Choose the correct label for each species from the drop-down menu: NH3 [Select] Neither Lewis base BCI3 Lewis acid H3NBCI3 (Select]
Expert Answer:
Answer rating: 100% (QA)
1 NH3 Lewis base 2 BCl3 Lewis acid 3 H3NBCl3 Neither BCl3 molecule accepts ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Consider the following reaction at some temperature: H2O(g) + CO(g) H2(g) + CO2(g) K = 2.0 Some molecules of H2O and CO are placed in a 1.0- L container as shown below. When equilibrium is reached,...
-
Consider the following reaction between mercury (II) chloride and oxalate ion: 2 HgCI2(aq) + C2O42- (aq) 2 CIË (aq) + 2 CO2(g) + Hg2CI2(s) The initial rate of this reaction was determined for...
-
Consider the following reaction between oxides of nitrogen: NO2(g) + N2O(g) 3 NO (g) (a) Use data in Appendix C to predict how G for the reaction varies with increasing temperature. (b) Calculate G...
-
Ultimate Consulting is a management consulting firm that offers a wide range of services to companies in many industries. To stay on the cutting edge of developments in the business world and to...
-
Suppose that a person lives for two periods, earning $30,000 in income in period 1, which she consumes or saves for period 2. What is saved earns interest of 10% per year. a. Draw that persons...
-
Does Mi Ola have a first-mover or late-mover advantage? Explain your answer.
-
Use the Peclet number [Eq. (16-111a)] to determine which model (completely mixed or plug flow) is appropriate for the distillation column calculation at \(\mathrm{x}_{\mathrm{W}}=0.48\) in Problems...
-
Selected accounts for Kennedy Photography at December 31, 2016, follow: Requirements 1. Journalize Kennedy Photographys closing entries at December 31, 2016. 2. Determine Kennedy Photographys ending...
-
A researcher conducted a study with 6 coronary artery bypass surgeries and found the sample mean of complete the surgery to be 16.7 hours. Suppose the accepted population standard deviation is 12.7 ....
-
End A of the slender pole is given a velocity v A to the right along the horizontal surface. Show that the magnitude of the velocity of end B equals v A when the midpoint M of the pole comes in...
-
1. 2. 3. 4. 5. 6. 7. 8. Assuming CNE Inc. could receive 1/15, n/30 terms from its suppliers for its accounts payable (A/P), how much cash would CNE Inc. pay to its suppliers from January through...
-
the questions below and provide the FAR citation in accordance with the guidance provided at FAR 1.105-2 1. Discuss some of the best practices in conducting market research, both in the Federal...
-
Grand Corporation reported pretax book income of $ 6 6 3 , 0 0 0 . Tax depreciation exceeded book depreciation by $ 4 4 2 , 0 0 0 . In addition, the company received $ 3 3 1 , 5 0 0 of tax - exempt...
-
Sources of Magnetic Field 1. Estimate the maximum magnetic force per meter that Earth's magnetic field could exert on a current-carrying wire in a 20-A circuit in your house. 2. A straight segment of...
-
A horizontal disk of diameter 40 cm rotates about a vertical axis that passes through its center with angular speed 4 rad./s. The tangential speed, in m/s, of a point on its circumference is a. 0.8...
-
Find the real solutions of the equation. X 2+x-2=0
-
Determine whether pulse rates of college students are the same while the students are standing and sitting. Is this a systematics sample, convenience sample, stratified sample, cluster sample
-
Is the modified 5-question approach to ethical decision making superior to the modified moral standards or modified Past in approach?
-
What is the electron configuration for the transition metal ion(s) in each of the following compounds? a. (NF4)2[Fe(h2O)2CI4] b. [Co(NH3)2(NH2CH2CH2NH2)2]I2 c. Na2[TaF7] d. [Pt(NH3)4I2][Ptl4] Pt...
-
Without looking at data in the text, sketch a qualitative graph of the third ionization energy versus atomic number for the elements Na through Ar, and explain your graph.
-
Although y rays are far more penetrating than a particles, the latter are more likely to cause damage to an organism. Why? Which type of radiation is more effective at promoting the ionization of...
-
Job Cost Journal Entries and T Accounts Following are certain operating data for Durango Manu- facturing Company for January 2019: Total sales were \($1,800,000\), on which the company earned a 40%...
-
Summarized data for the first month's operations of Dobson Welding Foundry during 2019 are presented below. A job order costing system is used. 1. Materials purchased on account, \($58,000\). 2....
-
During June 2019, its first month of operations, Weston Manufacturing Company completed the transactions listed below. Weston uses a job order costing system. Materials requisitions and the wages...

Study smarter with the SolutionInn App


