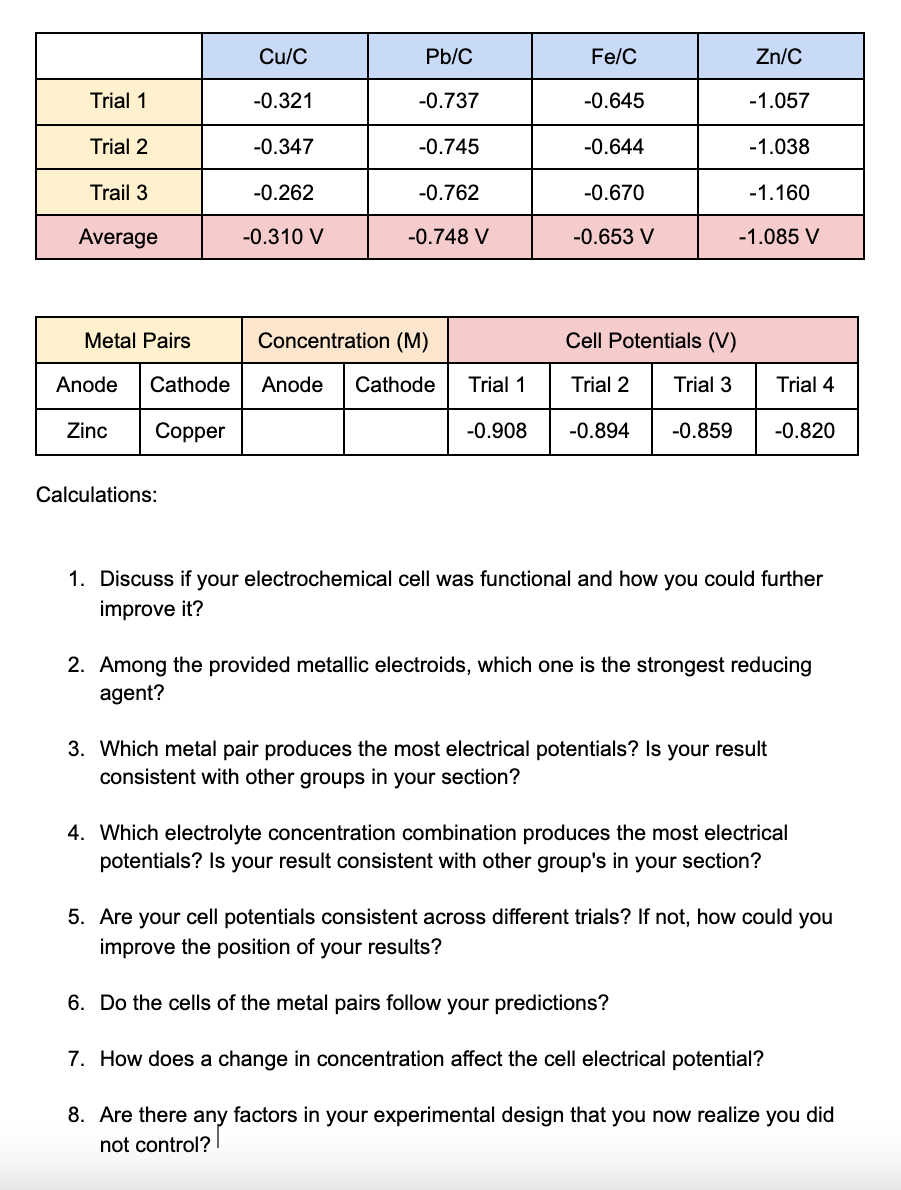
Trial 1 Trial 2 Trail 3 Average Metal Pairs Anode Cathode Zinc Copper Calculations: Cu/C -0.321...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Trial 1 Trial 2 Trail 3 Average Metal Pairs Anode Cathode Zinc Copper Calculations: Cu/C -0.321 -0.347 -0.262 -0.310 V Pb/C -0.737 -0.745 -0.762 -0.748 V Concentration (M) Anode Cathode Fe/C -0.645 -0.644 -0.670 -0.653 V Cell Potentials (V) Trial 1 Trial 2 Trial 3 -0.908 -0.894 -0.859 Zn/C -1.057 -1.038 -1.160 -1.085 V Trial 4 -0.820 1. Discuss if your electrochemical cell was functional and how you could further improve it? 3. Which metal pair produces the most electrical potentials? Is your result consistent with other groups in your section? 2. Among the provided metallic electroids, which one is the strongest reducing agent? 4. Which electrolyte concentration combination produces the most electrical potentials? Is your result consistent with other group's in your section? 5. Are your cell potentials consistent across different trials? If not, how could you improve the position of your results? 6. Do the cells of the metal pairs follow your predictions? 7. How does a change in concentration affect the cell electrical potential? 8. Are there any factors in your experimental design that you now realize you did not control? Trial 1 Trial 2 Trail 3 Average Metal Pairs Anode Cathode Zinc Copper Calculations: Cu/C -0.321 -0.347 -0.262 -0.310 V Pb/C -0.737 -0.745 -0.762 -0.748 V Concentration (M) Anode Cathode Fe/C -0.645 -0.644 -0.670 -0.653 V Cell Potentials (V) Trial 1 Trial 2 Trial 3 -0.908 -0.894 -0.859 Zn/C -1.057 -1.038 -1.160 -1.085 V Trial 4 -0.820 1. Discuss if your electrochemical cell was functional and how you could further improve it? 3. Which metal pair produces the most electrical potentials? Is your result consistent with other groups in your section? 2. Among the provided metallic electroids, which one is the strongest reducing agent? 4. Which electrolyte concentration combination produces the most electrical potentials? Is your result consistent with other group's in your section? 5. Are your cell potentials consistent across different trials? If not, how could you improve the position of your results? 6. Do the cells of the metal pairs follow your predictions? 7. How does a change in concentration affect the cell electrical potential? 8. Are there any factors in your experimental design that you now realize you did not control?
Expert Answer:
Answer rating: 100% (QA)
1 Discuss if your electrochemical cell was functional and how you could further improve it The electrochemical cell we used was functional We were able to measure the potentials of the metals we used ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
In problem 8.16, a college chemistry instructor thinks the use of embedded tutors will improve the success rate in introductory chemistry courses. The instructor carried out a hypothesis test and...
-
You have exams in economics and chemistry coming up, and you have 5 hours available for studying. The following table shows the trade-offs you face in allocating the time you will spend studying each...
-
A college chemistry instructor thinks the use of embedded tutors will improve the success rate in introductory chemistry courses. The passing rate for introductory chemistry is 62%. During one...
-
Write balanced equations based on the information given. (a) Solid magnesium + oxygen gas solid magnesium oxide (b) Nitrogen monoxide gas + oxygen gas nitrogen dioxide gas (c) Gaseous ethane(C2H6)...
-
Rickhaus Company is authorized to issue 10 million shares of $ 1 par value common stock and 800,000 shares of $ 10 par value 7 percent cumulative, convertible, preferred stock. For simplicity assume...
-
Calculate the firms EVA for both years. What does it indicate about the performance of the company?
-
Smith \& Sons reports sales revenue of \(\$ 1,000,000\) on its income statement. Its balance sheet reveals beginning and ending accounts receivable of \(\$ 92,000\) and \(\$ 60,000\), respectively....
-
A group of businessmen formed a corporation to lease for 5 years a piece of land at the intersection of two busy streets. The corporation has invested $50,000 in car-washing equipment. They will...
-
Question 5 Now, we will perform a Regression Discontinuity analysis by taking observations that are close to any of the cutoffs. In this question, we will be comparing rates of corruption for...
-
Client's Facts: The client found a check written out to cash in the amount of $750. The check was completely made out when he found it. He took it to the bank, signed it on the back as instructed by...
-
1- What signal is produced if the sampling frequency is equal to Fm? 2- Find the range of the cutoff frequency funt for LPF required to reconstruct the original signal from the sampled one. 3- If the...
-
When contemplating compensation communications, remember that what occurs when a company follows trends in programs and techniques, and the company needs to benchmark selectively?
-
Simplify the following Boolean function F, together with the don't care conditions d, and implement it with a. two-level NAND gate circuits. b. two-level NOR gate circuits....
-
contribution? J . Lo ( 3 ) Doris purchased a piano with $ 3 0 0 down and end - of - month payments of $ 1 2 4 for 2 percentage What was the purchase price of the piano ?
-
Research Critique Guidelines Part II Use this document to organize your essay. Successful completion of this assignment requires that you provide a rationale, include examples, and reference content...
-
The Lothridge Wholesale Beverage Company purchases soft drinks from producers and then sells them to retailers. The company began the year with inventory of $ 1 2 0 , 0 0 0 on hand. During the year,...
-
Hello can you help me with some question I have trouble with? Consider the following table of available call and put options on the same stock: Type: Call 62 Strike 0.5Maturity 12.42 Price 0.83 Delta...
-
Factor and simplify, if possible. Check your result using a graphing calculator. 3 cot 2 + 6 cot + 3
-
Show that the location of the centroid yc of the half-circle area shown is given by yc = 4R/3.The coordinate yc can be calculated by: dA dA dy
-
In the triangle shown a = 5 in., b = 7 in., and y = 25°. Define a, b, and y as variables, and then: (a) Calculate the length of c by substituting the variables in the Law of Cosines. (Law of...
-
A student has a summer job as a lifeguard at the beach. After spotting a swimmer in trouble, he tries to deduce the path by which he can reach the swimmer in the shortest time. The path of shortest...
-
The Arcadia Company is uncertain whether it should utilize the first-in, first-out (FIFO) method, the last-in, first-out (LIFO) method, or the weighted-average method to account for its inventory....
-
The Rock Creek Company has an ending inventory valuation using FIFO of \($350,000\). Had the company used LIFO the inventory would have been valued at \($320,000\). The company has determined the...
-
Walgreens Boots Alliance, Inc. (Walgreens) is a global pharmacy, health and well-being company. It operates over 13,000 stores in 11 countries, generating net sales of \($103.4\) billion. The...

Study smarter with the SolutionInn App


