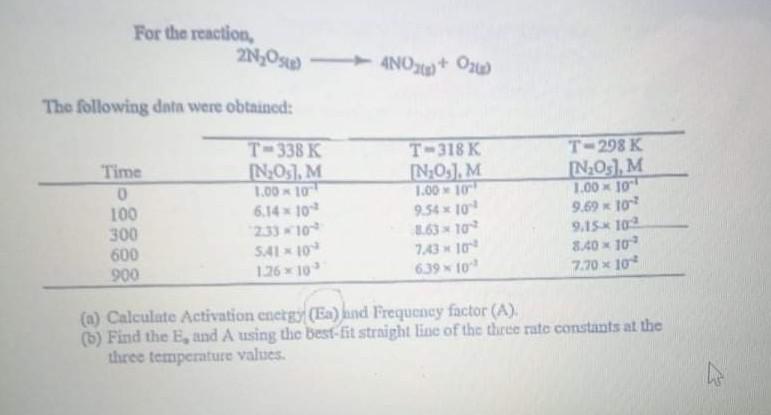
For the reaction, 2NO4NO2+0) The following data were obtained: Time 0 100 300 600 900 T-338...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
For the reaction, 2N₂O4NO2+0) The following data were obtained: Time 0 100 300 600 900 T-338 K [N₂0s], M 1.00-10 6.14 × 10 2.33-10 5.41-10 1.26×10³ T-318 K [N₂0₂], M 1.00 10 9.54 × 10¹ 8.63 10² 7.43 × 10¹¹ 6.39 × 10³ T-298 K IN₂0s], M 1.00 10 9.69 × 10² 9.15 102 8.40 10 7.70 × 10² (a) Calculate Activation energy (Ea) and Frequency factor (A). (b) Find the E, and A using the best-fit straight line of the three rate constants at the three temperature values. For the reaction, 2N₂O4NO2+0) The following data were obtained: Time 0 100 300 600 900 T-338 K [N₂0s], M 1.00-10 6.14 × 10 2.33-10 5.41-10 1.26×10³ T-318 K [N₂0₂], M 1.00 10 9.54 × 10¹ 8.63 10² 7.43 × 10¹¹ 6.39 × 10³ T-298 K IN₂0s], M 1.00 10 9.69 × 10² 9.15 102 8.40 10 7.70 × 10² (a) Calculate Activation energy (Ea) and Frequency factor (A). (b) Find the E, and A using the best-fit straight line of the three rate constants at the three temperature values.
Expert Answer:
Answer rating: 100% (QA)
a Calculating Activation Energy Ea and Frequency Factor A The Arrhenius equation is used to calculate the activation energy Ea and frequency factor A ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
In problem 8.16, a college chemistry instructor thinks the use of embedded tutors will improve the success rate in introductory chemistry courses. The instructor carried out a hypothesis test and...
-
You have exams in economics and chemistry coming up, and you have 5 hours available for studying. The following table shows the trade-offs you face in allocating the time you will spend studying each...
-
A college chemistry instructor thinks the use of embedded tutors will improve the success rate in introductory chemistry courses. The passing rate for introductory chemistry is 62%. During one...
-
Question 1 of 5 Which statement best illustrates Crusoe's reasons for teaching Friday about his religion? OA. He does not see Friday as a savage but as a soul worthy of saving. B. He thinks Friday...
-
Considering the case U. S. v. Bonnie Bain, develop a list of items the defense might seek to obtain through the discovery process.
-
In problem, information is given about a polynomial function f(x) whose coefficients are real numbers. Find the remaining zeros of f. Degree 4; zeros: 2 -i, -i
-
Sherwin Corporation invest its excess cash in equity securities when such founds are not needed to support operations. At the beginning of the year the companys portfolio consisted of the following...
-
DCF, sensitivity analysis, no income taxes. (CMA, adapted) Whimsical Corporation is an international manufacturer of fragrances for women. Management at Whimsical is considering expanding the product...
-
5 Connect Only Problem 13-13 NPV (LG13-3) 10 points eBook Suppose your firm is considering investing in a project with the cash flows shown below, that the required rate of return on projects of this...
-
Write a query to display the patron ID, book number, and days kept for each checkout. Days Kept is the difference from the date on which the book is returned to the date it was checked out. Sort the...
-
Assume a charged object with a mass of 3.6 gram moves in the positive direction of the z- axis with a kinetic energy of 18 kj. This object enters in a region of magnetic field of 7.2 mT whose...
-
Sasha plans to retire in 8 years from today with 283,422 dollars in her account. If she receives payments of 69,000 dollars per year and she receives her first 69,000 dollar payment in 9 years and...
-
Discuss considerations that should be fully taken into account when developing inventory related relevant costs for use in an economic order quantity (EOQ) model. Why do conflicts arise between the...
-
Hewlett Packard (HP) 2018 FY 10K filings highlights of financial analysis, and contrast to a non-manufacturing (or manufacturing) corporation's financial statements. about the company What areas are...
-
If the price of one of your stocks were to decline 80%, what return would it need to get back to even?
-
You plan to purchase equipment for your business. The equipment costs $ 2 5 , 0 0 0 and you plan to make a $ 5 , 0 0 0 down payment and finance the remaining amount over 1 2 months at an 8 % interest...
-
Page 8 CEO Samuel Drugs Ltd. Exhibit 1 ORGANIZATIONAL STRUCTURE OF THE HOUSTON GROUP Chairman 9B11C006 Managing Director Eastern Pharmaceuticals Ltd. Managing Director Tamil Nadu Pharmaceuticals Ltd....
-
In Exercises 15 through 30, find the derivative dy/dx. In some of these problems, you may need to use implicit differentiation or logarithmic differentiation. y ex + et -2x 1 + e
-
Define each of the following. a. Elementary step b. Molecularity c. Reaction mechanism d. Intermediate e. Rate-determining step
-
The radius trend and the ionization energy trend are exact opposites. Does this make sense? Define electron affinity. Electron affinity values are both exothermic (negative) and endothermic...
-
Consider the reaction 3A + B + C D + E where the rate law is defined as An experiment is carried out where [B]0 = [C]0 = 1.00 M and [A]0 = 1.00 Ã 10-4 M. a. If after 3.00 minutes [A] = 3.26...
-
Rose Ubens, buys and sells goods. Her trial balance is presented below. Note: 1. Closing inventory is 4,000 Required: Prepare Rose Ubens, income statement for the year ended 31 December 2012 and a...
-
Paul Icasso has the following trial balance. Required: Prepare Paul Icassos trading and income statement for the year ended 31 March 2013 and a statement of financial position as at 31 March 2013. P....
-
Michael Anet has the following trial balance. Required: Prepare Michael Anets income statement for the year ended 31 December 2012 and statement of financial position as at 31 December 2012. M. Anet...

Study smarter with the SolutionInn App


