1. Balance the following equations C7H14 02 CO2 H20 PCI3 H20 H3PO3 HCI QUESTION 2 Ammonia...
Fantastic news! We've Found the answer you've been seeking!
Question:

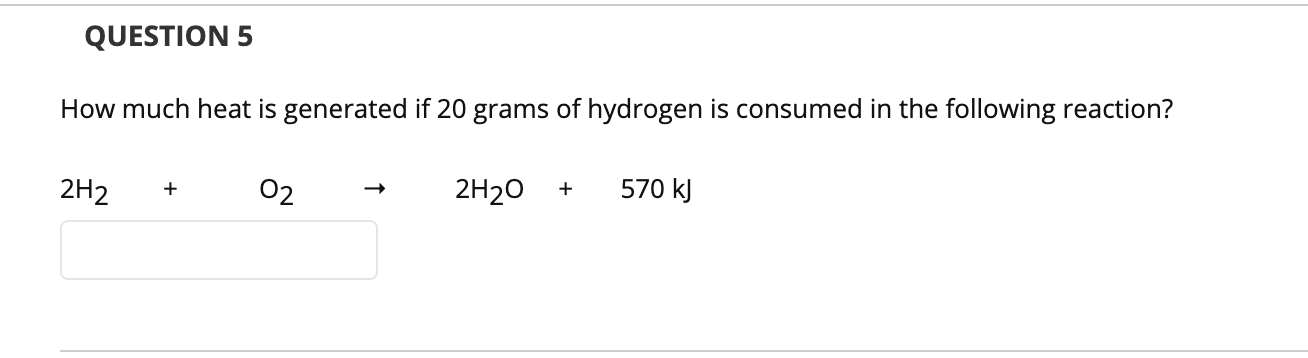
Transcribed Image Text:
1. Balance the following equations C7H14 02 CO2 H20 PCI3 H20 H3PO3 HCI QUESTION 2 Ammonia reacts with diatomic oxygen to form nitrogen monoxide and water vapor: NH3 + 02 ® NO H20 When 40.0 g NH3 and 50.0 g 02 are allowed to react, what is the theoretical yield of this reaction? If 30 g of NO is produced, what is the % yield? QUESTION 3 Aqueous solutions of sodium sulfide and lead (II) nitrate are combined. The products are sodium nitrate and lead (II) sulfide. Write the net ionic equation. Na2s + Pb(NO3)2 → 2 NaNO3 PbS s2. (aд) + Pb2+, (aq) + 2NO3 (aq) 2NO3'aq) 2 Na' (aq) PbS(s) + + + (aq) 2 Na"(aq) + 2NO3 aq) - 2 NANO3(s) s2- (aq) Pb2+, PbS(s) + (aq) QUESTION 4 What element is oxidized in the chemical reaction Cu + HNO3 → Cu(NO3)2 + NO + H20 Cu The nitrogen of the HNO3 is oxidized The hydrogen of the HNO3 is oxidized The oxygen of the HNO3 is oxidized QUESTION 5 How much heat is generated if 20 grams of hydrogen is consumed in the following reaction? 2H2 02 2H20 570 kJ 1. Balance the following equations C7H14 02 CO2 H20 PCI3 H20 H3PO3 HCI QUESTION 2 Ammonia reacts with diatomic oxygen to form nitrogen monoxide and water vapor: NH3 + 02 ® NO H20 When 40.0 g NH3 and 50.0 g 02 are allowed to react, what is the theoretical yield of this reaction? If 30 g of NO is produced, what is the % yield? QUESTION 3 Aqueous solutions of sodium sulfide and lead (II) nitrate are combined. The products are sodium nitrate and lead (II) sulfide. Write the net ionic equation. Na2s + Pb(NO3)2 → 2 NaNO3 PbS s2. (aд) + Pb2+, (aq) + 2NO3 (aq) 2NO3'aq) 2 Na' (aq) PbS(s) + + + (aq) 2 Na"(aq) + 2NO3 aq) - 2 NANO3(s) s2- (aq) Pb2+, PbS(s) + (aq) QUESTION 4 What element is oxidized in the chemical reaction Cu + HNO3 → Cu(NO3)2 + NO + H20 Cu The nitrogen of the HNO3 is oxidized The hydrogen of the HNO3 is oxidized The oxygen of the HNO3 is oxidized QUESTION 5 How much heat is generated if 20 grams of hydrogen is consumed in the following reaction? 2H2 02 2H20 570 kJ
Expert Answer:

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
Write the net ionic equation for CaCl2(aq) + Pb(NO3)2(aq) ( Ca(NO3)2(aq) + PbCl2(s)
-
Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate): (a) (b) (c) Ba(OH)-(aq) + HPO 4 (aq )- HCIO4 (aq) + Mg(OH )2 (s)
-
Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate): (a) (b) (c) CH3COOH (aq) + KOH(aq)- .co.(aq) + NaO H (aq) - HNO3(aq ) + Ba(OH)2(aq )-
-
Blaine is a practicing dentist. He operates his business from the basement of his house, with a separate entrance and facilities. Blaine uses the simplified method every year to calculate his...
-
Suppose that the parts produced by a machine can have three different levels of functionality: working, impaired, defective. Let p1, p2, and p3 = 1 p1 p2 be the probabilities that a part is working,...
-
Sardano and Sons is a large, publicly held company that is considering leasing a warehouse. One of the company's divisions specializes in manufacturing steel, and this particlar warehouse is the only...
-
Which of the following is an acceptable method to report total comprehensive income according to U.S. GAAP? (a) On the face of the balance sheet. (b) In the discontinued operations section of the...
-
The Gidewall Corporation uses part 210 in a manufacturing process. Information as to balances on hand, purchases, and requisitions of part 210 is given in the following table: Instructions: What is...
-
The below table shows the number of operations with 78 Beds (26 patients per day). Each row in the table shows the number of patients who checked in on a given day. The columns indicate the number of...
-
The lifetime (in months) of a battery is modeled by a random variable X that has pdf f ( x ) = K x 1 ( x > 0 ) where K = ln ( 1 / ) for an unknown parameter ( 0 , 1 ) . (Here 1 ( x > 0 )...
-
11. _____________________________________ concerns the ongoing social, economic, and political process that deepens and broadens the relationships and interdependencies amongst nations, their people,...
-
james and annie bourke sold stock in the current year. the sale resulted in a short term capital loss of 4,000 the bourkes had no other capital transactions during the year. their taxable income was...
-
Introduction Talk about the history of Publix where everything started and how the company is still active now. 1-Describe Publix management system. 2-How Publix manage their inventory and warehouse?...
-
Explain to me how to get the manufacturing overhead number? Trees Sod Selling Price Per Unit $ 12.99 $ 0.38 Direct Materials Per Unit $ 0.99 $ 0.01 Direct Labor Per Unit $ 2.00 $ 0.09 Direct Labor...
-
Using the SEC 10-K report for Vertex pharmaceuticals please respond to the following: What is the name of the auditor? Are they one of the "Big Four"? conduct a search to see who the "Big Four" audit...
-
On 1 June 2023, Zul Ghani established a management consultancy firm, ZG Consultancy Sdn Bhd. During the month, Zul carried out the following transactions: June 1 Zul deposited RM25,000 into bank...
-
A convex mirror with a radius of curvature of 3 m is used as rear-view mirror for a vehicle. If a bus is located at 5 m from this mirror, find the position, nature and size of the image.
-
Propose a reasonable mechanism for the following reaction. OH
-
What is the freezing point of an aqueous solution that boils at 105.0 oC?
-
Specify what ions are present in solution upon dissolving each of the following substances in water: (a) ZnCl2 (b) HNO3 (c) (NH4)2SO4 (d) Ca(OH)2.
-
Detailed calculations show that the value of Zeff for the outermost electrons in Si and Cl atoms is 4.29+ and 6.12+, respectively. (a) What value do you estimate for Zeff experienced by the outermost...
-
An individual has the genotype Aa Bb Cc and makes an abnormal gamete with the genotype AaBc. Does this gamete violate the law of independent assortment or the law of segregation (or both)? Explain...
-
Marfan syndrome is a rare inherited human disorder characterized by unusually long limbs and digits plus defects in the heart (especially the aorta) and the eyes, among other symptoms. Following is a...
-
With regard to biological evolution, which of the following statements is incorrect? Explain why. A. During its lifetime, an animal evolves to become better adapted to its environment. B. The process...

Study smarter with the SolutionInn App



