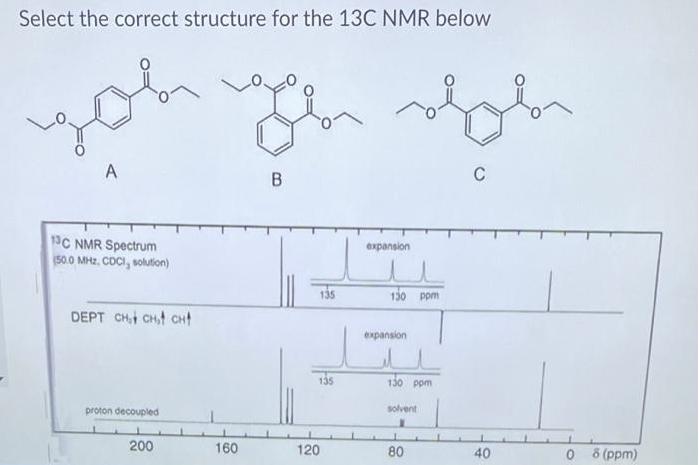
Select the correct structure for the 13C NMR below you A C B 1C NMR Spectrum...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Select the correct structure for the 13C NMR below you A C B 1C NMR Spectrum (50.0 MHz. CDCI, solution) expansion 135 130 ppm DEPT CHt CHt CHt expansion 135 130 pom proton decoupled solvent 200 160 120 0 8 (ppm) 80 40 Select the correct structure for the 13C NMR below you A C B 1C NMR Spectrum (50.0 MHz. CDCI, solution) expansion 135 130 ppm DEPT CHt CHt CHt expansion 135 130 pom proton decoupled solvent 200 160 120 0 8 (ppm) 80 40
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
For each compound shown below, (1) Sketch the 13C NMR spectrum (totally decoupled, with a singlet for each type of carbon), showing approximate chemical shifts. (2) Show the multiplicity expected for...
-
The IR spectrum, 13C NMR spectrum, and 1HNMR spectrum of an unknown compound (C6H8O3) appear next. Determine the structure, and show how it is consistent with the spectra. wavelength (um) 5 5.5 6 710...
-
The standard 13C NMR spectrum of phenyl propanoate is shown here. Predict the appearance of the DEPT-90 and DEPT-135 spectra. 13C NMR 0-C-CH2CH3 pheny! propanoate 200 180 160140 10 100 80 40 20 0 8...
-
Create a T-Account Transaction Account titles Cash d Common stock Supplies Creditors accounts payable) Cash Fees earned Rent expense Cash Creditors(accounts payable) Cash Accounts receivable Fees...
-
Identify the foreign keys.
-
Travolta Company accumulates the following data concerning a proposed capital investment: cash cost $225,000, net annual cash flow $34,000, present value factor of cash inflows for 10 years 6.71...
-
Cubs Incorporated manufactures a product with a selling price of $60 per unit. Units and monthly cost data follow: Cubs Inc. pays all bills in the month incurred. All sales are on account with 50...
-
Start Excel. Download and open the file named Exp19_Excel_Ch09_Cap_HouseCleaners.xfsx. Grader has automatically added your last name to the beginning of the filename. 1 2 The title and column labels...
-
Music and Math Camp, Inc. is a not-for-profit organization dedicated to the development of music skills in kids through weekend and summer camp experiences. Research shows that learning to read music...
-
Placid Lake Corporation acquired 80 percent of the outstanding voting stock of Scenic, Inc., on January 1, 2023, when Scenic had a net book value of $400,000. Any excess fair value was assigned to...
-
3. A researcher suspects that the actual prevalence of generalized anxiety among children and adolescents is higher than the previously reported prevalence of generalized anxiety disorder among...
-
What are the two general types of feedback and what are the advantages and disadvantages of each type?
-
Define the following terms: a. Point estimate b. Confidence interval c. Confidence level
-
State the following: a. The five attestation general standards. b. The two attestation standards of field work. c. The four attestation standards of reporting.
-
According to the Committee on Basic Auditing Concepts, what are the four conditions that create a demand for auditing?
-
CPA firms should establish quality control policies and procedures for professional development to provide reasonable assurance that a. Employees promoted possess the appropriate characteristics to...
-
Question 9 Profit and loss statements for two companies are presented below: Company A Company B Revenue $432,000 Revenue $1,003,331 Expenses Expenses Operating Exp. 44,000 Operating Exp. 243,000...
-
Why is inventory management important for merchandising and manufacturing firms and what are the main tradeoffs for firms in managing their inventory?
-
Show how you would synthesize each compound, starting with an ester containing no more than eight carbon atoms. Any other necessary reagents may be used. (a) Ph3C¬OH (b) (PhCH2)2CHOH (c)...
-
Show how you would use the Robinson annulation to synthesize the following compounds. (a) (b) (c) H,C Ph CH3
-
Propose a mechanism for the triolefin process using a metal alkylidene as the catalyst. CH IM CHCH CH - - - CH3 propylene but-2-ene (cis + trans) ethylene
-
Griffin Metals Co. has provided the following data: The selling price is expected to be \($300\) per tonne for the first six months and \($310\) per tonne thereafter. Variable costs per tonne are...
-
Highjinks Corporation's sales department has estimated revenue of 250,000 for your division. 60% of this will be achieved in the first half-year and 40% in the remaining half-year. Variable operating...
-
The total cost of goods sold for June is a. $103,500 b. $128,800 c. 57,500 d. $232,300

Study smarter with the SolutionInn App