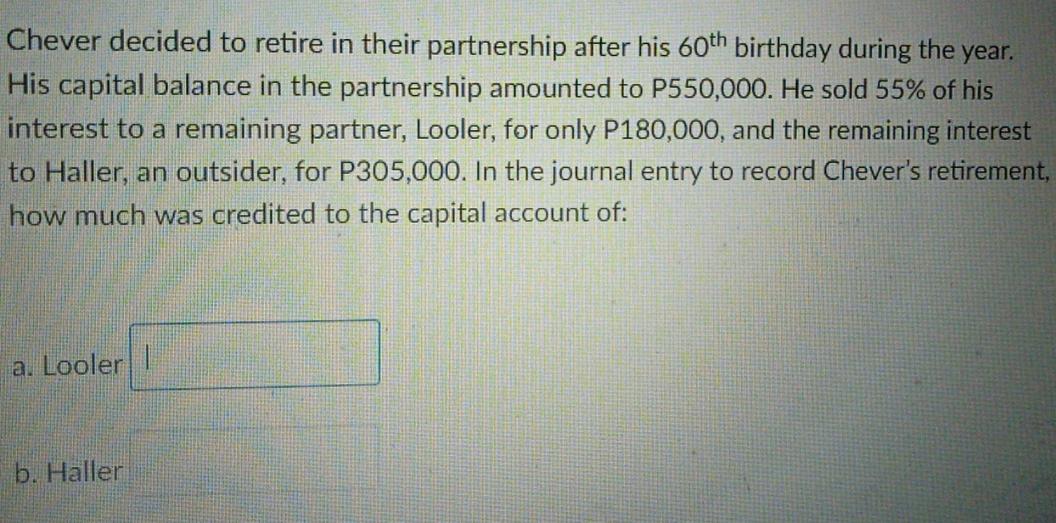
Chever decided to retire in their partnership after his 60th birthday during the year. His capital...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Chever decided to retire in their partnership after his 60th birthday during the year. His capital balance in the partnership amounted to P550,000. He sold 55% of his interest to a remaining partner, Looler, for only P180,000, and the remaining interest to Haller, an outsider, for P305,000. In the journal entry to record Chever's retirement, how much was credited to the capital account of: a. Looler I b. Haller Chever decided to retire in their partnership after his 60th birthday during the year. His capital balance in the partnership amounted to P550,000. He sold 55% of his interest to a remaining partner, Looler, for only P180,000, and the remaining interest to Haller, an outsider, for P305,000. In the journal entry to record Chever's retirement, how much was credited to the capital account of: a. Looler I b. Haller
Expert Answer:
Answer rating: 100% (QA)
SOLUTION To record Chevers retirement and the sale of his interest we need to adjust his capital bal... View the full answer

Related Book For 

Smith and Roberson Business Law
ISBN: 978-0538473637
15th Edition
Authors: Richard A. Mann, Barry S. Roberts
Posted Date:
Students also viewed these accounting questions
-
A uniform rod AB, of mass 4 kg and length L = 1.5 m, is released from rest in the position shown. Knowing that = 20?, determine the values immediately after release of (a) The angular acceleration...
-
The accompanying statements of the parks, recreations, and municipal capital improvement bond fund (a capital projects fund) were drawn from an annual report of Parkville. According to a note in the...
-
The accompanying statements of the parks, recreations, and municipal capital improvement bond fund (a capital projects fund) were drawn from an annual report (dates changed) of Boulder, Colorado....
-
Use the Chain Rule to calculate the partial derivatives. Express the answer in terms of the independent variables. dh t -; h(x, y) = X x = tt, y = tt " y
-
How could you convert butanoic acid into the following compounds? Write each step showing the reagents needed. (a) 1 -Butanol (b) 1Bromobutane (c) Pentanoic acid (d) 1-Butenc (e) Octane
-
Deal with the following concepts: A point p is called a positive combination of the points v 1 ,........., v k if p = c 1 v 1 +..........+c k v k , with all c i 0. The set of all positive...
-
The following controi procedures over payroll transactions are prescribed by the Hahn Company: 1. Operating department requisition for each hiring. 2. Personnel department authorizations for...
-
Moreno Company publishes a monthly sports magazine, Fishing Preview. Subscriptions to the magazine cost $20 per year. During November 2014, Moreno sells 15,000 subscriptions beginning with the...
-
The LOL Company had the following items in its complex capital structure on December 31, 2023: Preferred stock, $8 par value, $1 dividend, 10,000 shares issued and outstanding. Common stock, $5...
-
Here are my code. And the documentation for reference class DeliveryPlanner_PartA: """ Required methods in this class are: plan_delivery(self, debug = False) which is stubbed out below. You may not...
-
List two secondary sexual characters of males that are different from that of females.
-
The Blindjammer Co. bonds are currently selling for $1,007.27. These bonds mature in fouryears, pay interest annually, and have a yield-to-maturity of 8.63%. What is the coupon rate?
-
Now suppose company A issued a 1% cash dividend instead of a stock dividend? How would things change?
-
Company A desires a fixed-rate loan. Company A presently has access to floating interest rate funds at LIBOR 2%. Its direct borrowing cost is 15% in the fixed-rate bond market. In contrast, company...
-
Why is it important to plan your search on a particular topic? Give an example when planning has not turned out well for you. Perform an internet search on any tax, accounting, or auditing topic that...
-
What cost savings, if any, has been observed as a direct result of using the EMR?
-
Suppose the return R on a stock satisfies R = +AY where u and X are fixed and Y has a t-distribution with degrees of freedom. (a) (5pt) If you hold So position in this stock, show that for a one day...
-
Nitrogen monoxide reacts with hydrogen as follows: 2NO(g)+ H2(g) N2O(g) + H2O(g) The rate law is [H2]/ t = k[NO]2[H2], where k is 1.10 107 L2/(mol2s) at 826oC. A vessel contains NO and H2 at...
-
You have an employee from another country who is very shy. After a time, you notice that the quality of her performance is deteriorating rapidly. You find an appropriate time to speak with her and...
-
Marcia executed a mortgage on Blackacre to secure her indebtedness to Ajax Savings and Loan Association in the amount of $25,000. Later, Marcia sold Blackacre to Morton. The deed contained the...
-
In 1969 a deed for land in Pitt County was executed and delivered by Joel and Louisa Tyson unto M. H. Jackson and wife Maggie Jackson, for and during the term of their natural lives and after their...
-
For liquid acetone at \(20^{\circ} \mathrm{C}\) and \(1 \mathrm{bar}\), \[ \beta=1.487 \times 10^{-3} /{ }^{\circ} \mathrm{C} \quad \alpha=62 \times 10^{-6} / \mathrm{bar} \quad V=1.287...
-
Determine the molar volume of ammonia vapour and ammonia liquid at \(321.55 \mathrm{~K}\) and 1.95 \(\mathrm{MPa}\). Ammonia is assumed to follow van der Waals equation of state.
-
Any equation that relates to the pressure, temperature and volume is called an equation of state. Justify the statement.

Study smarter with the SolutionInn App


