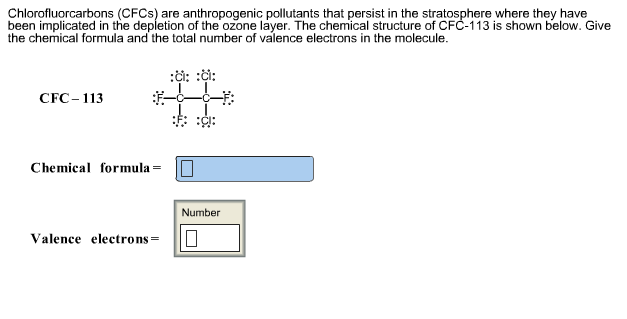
Chlorofluorcarbons (CFCs) are anthropogenic pollutants that persist in the stratosphere where they have been implicated in...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Chlorofluorcarbons (CFCs) are anthropogenic pollutants that persist in the stratosphere where they have been implicated in the depletion of the ozone layer. The chemical structure of CFC-113 is shown below. Give the chemical formula and the total number of valence electrons in the molecule. CFC-113 Chemical formula Valence electrons = :: :: Number 0 Dilorofluorcarbons (CFCs) are highly stable gas molecules that travel through the troposphere and deep into the stratosphere where they have contributed to the depletion of the ozone layer. To circumvent the harmful environmental effects of CFCs, chemists developed hydrochlorofluorocarbons (HCFCs). They are less harmful than CFCs because they decompose much more readily in the trophosphere than CFCs. Draw the Lewis dot structures for CFC-11 (CCI3F), CFC-12 (CCl₂F2), and HCFC-22 (CHCIF2). CFC-11 CFC-12 HCFC–22 Chlorofluorcarbons (CFCs) are anthropogenic pollutants that persist in the stratosphere where they have been implicated in the depletion of the ozone layer. The chemical structure of CFC-113 is shown below. Give the chemical formula and the total number of valence electrons in the molecule. CFC-113 Chemical formula Valence electrons = :: :: Number 0 Dilorofluorcarbons (CFCs) are highly stable gas molecules that travel through the troposphere and deep into the stratosphere where they have contributed to the depletion of the ozone layer. To circumvent the harmful environmental effects of CFCs, chemists developed hydrochlorofluorocarbons (HCFCs). They are less harmful than CFCs because they decompose much more readily in the trophosphere than CFCs. Draw the Lewis dot structures for CFC-11 (CCI3F), CFC-12 (CCl₂F2), and HCFC-22 (CHCIF2). CFC-11 CFC-12 HCFC–22
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Draw three reasonable Lewis dot structures for the ONC" anion, including formal charges. N.B. that the N atom is the central atom. Order them in terms of best to worst (just write best next to the...
-
Ozone molecules in the stratosphere absorb much of the harmful radiation from the sun. Typically, the temperature and pressure of ozone in the stratosphere are 250 K and 1.0 - 10-3 atm, respectively....
-
The depletion of ozone (O3) in the stratosphere has been a matter of great concern among scientists in recent years. It is believed that ozone can react with nitric oxide (NO) that is discharged from...
-
Look at the following code. Which line will cause a compiler error? Explain why. Line 1 public class ClassA Line 2 { Line 3 public ClassA () {} Line 4 public final int methodl (int a) () Line 5...
-
Apollo Companys sales manager predicts that annual sales of the companys product will soon reach 40,000 units and its price will increase to $200 per unit. According to the production manager, the...
-
A foundation was endowed with $15,000,000 in July 2010. In July 2014, $5,000,000 was expended for facilities, and it was decided to provide $250,000 at the end of each year forever to cover operating...
-
Redwing Floor Coverings, Inc., reported the following summarized data at December 31, 2009. Accounts appear in no particular order. Requirement 1. Prepare the trial balance of Redwing Floor Coverings...
-
First National Bank hired Longdon as a secretary and obtained a surety bond from Belton covering the bank against losses up to $100,000 resulting from Longdons improper conduct in the performance of...
-
Official Time: 20:48:00 Question 2 [5 points] X-cell Inc. showed the following equity information as at December 31, 2023. a. The preferred shares had sold for an average price of $14. b. The common...
-
Wild West produces two types of cowboy hats. A Type 1 hat requires twice as much labor time as a Type 2. If all the available labor time is dedicated to Type 2 alone, the company can produce a total...
-
The following 25 measurements were taken of the thickness of steel emerging from a rolling mill. 3.97 3.99 4.04 4.00 3.98 4.03 4.00 3.98 3.99 3.96 4.02 3.99 4.01 3.97 4.02 3.99 3.95 4.03 4.01 4.05...
-
Larned Corporation recorded the following transactions for the just completed month. Purchased $ 8 9 , 0 0 0 of raw materials on account. $ 8 7 , 0 0 0 in raw materials were used in production. Of...
-
Justina is recently promoted to be the vice president of global business development in an American MNC. Her position requires her to bring in new business from clients around the world. Her first...
-
Identify the 8 elements of communication process. Does feedback indicate that communication has been successful?
-
What is the relations between globalization in business and the need of intercultural communication which currently happening?
-
8. A yo-yo with mass M and radius R is wound with a light string. Someone drops it from rest with the string attached to a metal pole. The yo-yo falls and unwinds (spins) without slipping. The moment...
-
4. A boundary layer forms on a convex curve. Over this curve the boundary layer detaches and does not reattach. (a) Sketch and label typical velocity profiles for the laminar, turbulent, and detached...
-
In the current year, the City of Omaha donates land worth $500,000 to Ace Corporation to induce it to locate in Omaha and create an estimated 2,000 jobs for its citizens. a. How much income, if any,...
-
Consider the compounds butanoic acid, pentanal, M-hexane, and 1-pentanol. The boiling points of these compounds (in no specific order) are 69C, I03C, 137C, and 164C. March the boiling points to the...
-
Is ocranoic acid more soluble in 1 M HC1, 1 M NaOH, or pure water? Explain. Drugs such as morphine are often treated with strong acids. The most commonly used form of morphine is morphine...
-
Give the balanced equation for each of the following chemical reactions. a. Glucose (C6H12O6) reacts with oxygen gas to produce gaseous carbon dioxide and water vapor. b. Solid iron(III) sulfide...
-
On what procedures does the auditor place heavy reliance in the audit of revenue and expense?
-
Describe an overall test of revenue for reasonableness.
-
Give two reasons for analyzing an expense account.

Study smarter with the SolutionInn App



