:CI: B (a) NO NO You wish to prepare various isomers of chloronitrobenzene from benzene. For...
Fantastic news! We've Found the answer you've been seeking!
Question:
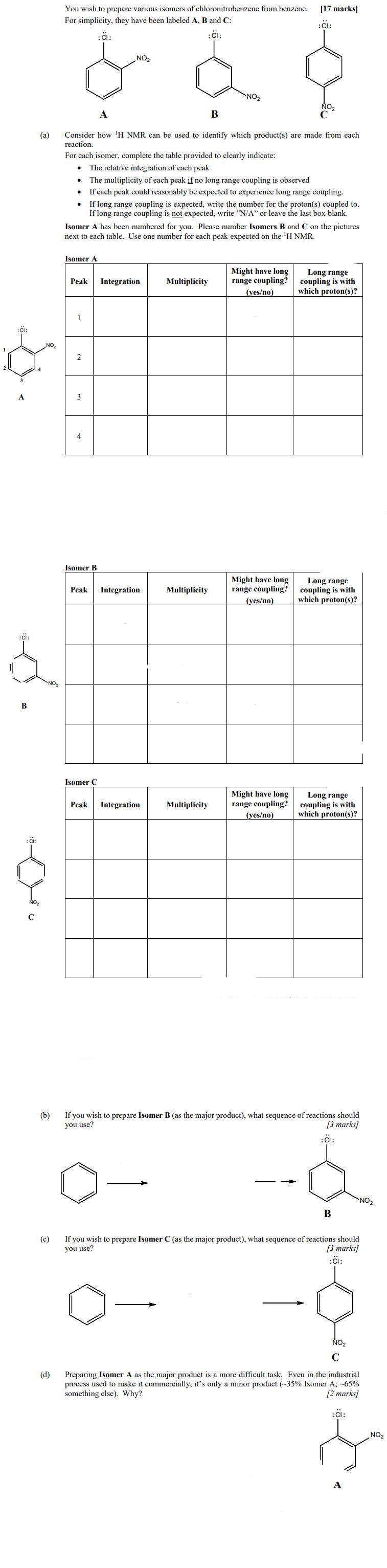
Transcribed Image Text:
:CI: B (a) NO₂ NO₂ You wish to prepare various isomers of chloronitrobenzene from benzene. For simplicity, they have been labeled A, B and C: :CI: (c) . A B Consider how 'H NMR can be used to identify which product(s) are made from each reaction. ● For each isomer, complete the table provided to clearly indicate: The relative integration of each peak The multiplicity of each peak if no long range coupling is observed If each peak could reasonably be expected to experience long range coupling. Isomer A Peak If long range coupling is expected, write the number for the proton(s) coupled to. If long range coupling is not expected, write "N/A" or leave the last box blank. Isomer A has been numbered for you. Please number Isomers B and C on the pictures next to each table. Use one number for each peak expected on the ¹H NMR. 1 4 NO₂ Isomer B Isomer C Peak Integration Peak Integration Integration NO₂ Multiplicity Multiplicity Multiplicity [17 marks] :CI: Might have long range coupling? (yes/no) Might have long range coupling? (yes/no) Might have long range coupling? (yes/no) Long range coupling is with which proton(s)? Long range coupling is with which proton(s)? Long range coupling is with which proton(s)? (b) If you wish to prepare Isomer B (as the major product), what sequence of reactions should you use? [3 marks] : CI: B If you wish to prepare Isomer C (as the major product), what sequence of reactions should you use? [3 marks] :CI: NO₂ C (d) Preparing Isomer A as the major product is a more difficult task. Even in the industrial process used to make it commercially, it's only a minor product (-35% Isomer A; -65% something else). Why? [2 marks] : CI: NO₂ NO₂ :CI: B (a) NO₂ NO₂ You wish to prepare various isomers of chloronitrobenzene from benzene. For simplicity, they have been labeled A, B and C: :CI: (c) . A B Consider how 'H NMR can be used to identify which product(s) are made from each reaction. ● For each isomer, complete the table provided to clearly indicate: The relative integration of each peak The multiplicity of each peak if no long range coupling is observed If each peak could reasonably be expected to experience long range coupling. Isomer A Peak If long range coupling is expected, write the number for the proton(s) coupled to. If long range coupling is not expected, write "N/A" or leave the last box blank. Isomer A has been numbered for you. Please number Isomers B and C on the pictures next to each table. Use one number for each peak expected on the ¹H NMR. 1 4 NO₂ Isomer B Isomer C Peak Integration Peak Integration Integration NO₂ Multiplicity Multiplicity Multiplicity [17 marks] :CI: Might have long range coupling? (yes/no) Might have long range coupling? (yes/no) Might have long range coupling? (yes/no) Long range coupling is with which proton(s)? Long range coupling is with which proton(s)? Long range coupling is with which proton(s)? (b) If you wish to prepare Isomer B (as the major product), what sequence of reactions should you use? [3 marks] : CI: B If you wish to prepare Isomer C (as the major product), what sequence of reactions should you use? [3 marks] :CI: NO₂ C (d) Preparing Isomer A as the major product is a more difficult task. Even in the industrial process used to make it commercially, it's only a minor product (-35% Isomer A; -65% something else). Why? [2 marks] : CI: NO₂ NO₂
Expert Answer:
Related Book For 

Physics
ISBN: 978-0077339685
2nd edition
Authors: Alan Giambattista, Betty Richardson, Robert Richardson
Posted Date:
Students also viewed these chemistry questions
-
You wish to prepare 0.12 M HNO3 from a stock solution of nitric acid that is 15.8 M. How many milliliters of the stock solution do you require to make up 1.00 L of 0.12 M HNO3?
-
You wish to prepare 1 L of a 0.02-M potassium iodate solution. You require that the final concentration be within 1% of 0.02 M and that the concentration must be known accurately to the fourth...
-
True or false? a. Sensitivity analysis can be used to identify the variables most crucial to a project's success. b. Sensitivity analysis is used to obtain expected, optimistic, and pessimistic...
-
At December 31, 2014, Torrealba Company reported the following as plant assets. During 2015, the following selected cash transactions occurred. April 1 Purchased land for $1,200,000. May 1 Sold...
-
What supply chain logistics alternatives are available in JIT environments? How does each work in a make-to-stock versus a make-to-order environment? What objective customer service measurements are...
-
1. Did the STR ban adopted by the association comport with or contravene its status as a benefit corporation? Discuss. 2. Suppose that instead of adopting an STR ban on its own, the association had...
-
You throw a \(100-\mathrm{g}\) ball upward with a speed of \(19.8 \mathrm{~m} / \mathrm{s}\). How much work does the force of gravity do on the ball during its trip to its maximum height?
-
1. What was Walmarts early global expansion strategy? Why did it choose to first enter Mexico and Canada rather than expand into Europe and Asia? 2. What cultural problems did Walmart face in some of...
-
3. A particle of mass m is moving in the xy-plane such that its velocity at a point (x, y) is given as v = a(yx+2xy), where a is a non-zero constant. What is the force F acting on the particle? (A) F...
-
Determine the moments of inertia I x and I y of the area shown with respect to centroidal axes respectively parallel and perpendicular to side AB. 60 mm 320 mm 40 mm so mm 80 inm 330 mm 80 mm
-
An annuity will make a payment of $100 in one year; this is the first of a series of twenty payments. If each succeeding annual payment is 5 percent larger than the previous one, what is the present...
-
True Or False In states that prohibit leaving keys in a parked car, a defendant is not necessarily negligent per se if they leave their keys in their car and the thief who steals the car is involved...
-
In an emergency, a defendant is expected to act __________ or can be found to be liable for their actions.
-
What are the duties of a tenant?
-
True Or False In determining whether a defendant acted reasonably, the jury must consider the situation from the defendants perspective and state of mind.
-
True Or False In evaluating a defendants conduct, a jury is allowed the benefit of information the defendant did not have at the time they acted.
-
Bethesda Mining Company reports the following balance sheet information for 2018 and 2019. Assets Current assets Cash $ 65,470 $82,487 Accounts receivable 65,281 85,639 181,549 Inventory 116,676...
-
The production budget of Artest Company calls for 80,000 units to be produced. If it takes 30 minutes to make one unit and the direct labor rate is $16 per hour, what is the total budgeted direct...
-
Suppose that an ideal capacitor and an ideal inductor are connected in series in an ac circuit. (a) What is the phase difference between vC(t) and vL (t)? (b) If the rms voltages across the capacitor...
-
What is the ratio of the wavelength of a 0.100-keV photon to the wavelength of a 0.100-keV electron?
-
An aluminum cylinder weighs 1.03 N. When this same cylinder is completely submerged in alcohol, the volume of the displaced alcohol is 3.90 105 m3. If the cylinder is suspended from a scale while...
-
What other questions and objections can you anticipate? If you revised the message, which questions and objections would you want to address? Are there any you would choose not to address? If so, why?
-
Review how the ACE process led to the improved message by answering the following questions. 1. What information in the revised version addresses the need for persuasion? 2. How does the email...
-
As consumers increasingly interact with one another in digital form, what are the ramifications for real-world human relationships?

Study smarter with the SolutionInn App



