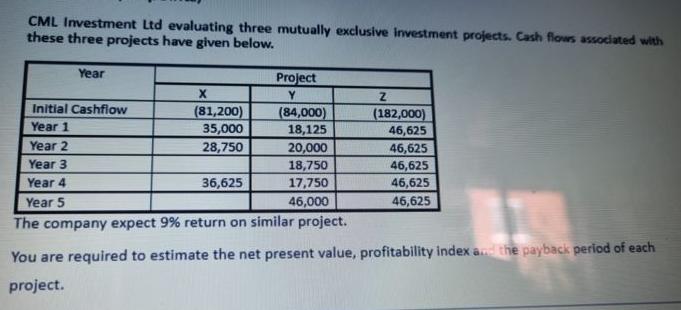
CML Investment Ltd evaluating three mutually exclusive investment projects. Cash flows associated with these three projects...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
CML Investment Ltd evaluating three mutually exclusive investment projects. Cash flows associated with these three projects have given below. Year Initial Cashflow Year 1 Year 2 Year 3 Year 4 Year 5 X (81,200) 35,000 28,750 Project Y 36,625 Z (182,000) 46,625 46,625 46,625 46,625 46,625 (84,000) 18,125 20,000 18,750 17,750 46,000 The company expect 9% return on similar project. You are required to estimate the net present value, profitability index and the payback period of each project. CML Investment Ltd evaluating three mutually exclusive investment projects. Cash flows associated with these three projects have given below. Year Initial Cashflow Year 1 Year 2 Year 3 Year 4 Year 5 X (81,200) 35,000 28,750 Project Y 36,625 Z (182,000) 46,625 46,625 46,625 46,625 46,625 (84,000) 18,125 20,000 18,750 17,750 46,000 The company expect 9% return on similar project. You are required to estimate the net present value, profitability index and the payback period of each project.
Expert Answer:
Answer rating: 100% (QA)
To calculate the net present value NPV profitability index PI and payback period for each project we need to discount the cash flows using the companys expected return of 9 Project X Year 0 81200 Year ... View the full answer

Related Book For 

Foundations of Finance The Logic and Practice of Financial Management
ISBN: 978-0132994873
8th edition
Authors: Arthur J. Keown, John D. Martin, J. William Petty
Posted Date:
Students also viewed these finance questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-4. Ivan and Irene paid the following in 2012 (all by check or can otherwise be...
-
You are the president of a new, capital-rich firm and you must decide on your firm's targeting approach. Three large subgroups exist in your market that have never had their unique needs served. If...
-
A chemical firm is considering how best to supply the world market for sulfuric acid. A manufacturing plant costs about $20 million to construct and requires a moderately skilled workforce. The total...
-
Fred and Martina, senior agents at an airline security checkpoint, carry out advanced screening procedures for hundreds of randomly selected passengers per day. For a random sample of 30 passengers...
-
An engineer studied the effect of four variables on a dimensionless factor used to describe pressure drops in a screen-plate bubble column. Table B. 9 summarizes the experimental results. a. Fit a...
-
Josh Borke recorded the following transactions during the month of April. Post these entries to the Cash T-account of the general ledger to determine the ending balance in cash. The beginning balance...
-
Company M has reported the following information for the month of February: Direct labor $ 4 5 , 0 0 0 Manufacturing overhead applied $ 9 0 , 0 0 0 Direct materials inventory has decreased by $ 1 1 ,...
-
John and Nina Hartwick, married 17 years, have a 13-year-old daughter. Eight years ago, they purchased a home on which they owe about $240,000. They also owe $6,000 on a two-year-old automobile. All...
-
Constance Wozek is the matriarch of her family. She is 75 years of age and still the CEO of "Plastics and Gadgets", a company she started in 1975. Constance's firm makes plastics (custom molding) and...
-
What will the multiplier be when the MPS is 0, 0.4, 0.6, and 1? What will it be when the MPC is 1, 0.90, 0.67, 0.50, and 0? How much of a change in GDP will result if firms increase their level of...
-
Suppose that an economy begins in long-run equilibrium before the price level and real GDP both decline simultaneously. If those changes were caused by only one curve shifting, then those changes are...
-
On 1 October of the current year, Bourdon Company paid \$396 for a two-year comprehensive insurance policy on the business' building. Required: a Prepare the journal entry to record each of the...
-
A bank borrows $100,000 from the Fed, leaving a $100,000 Treasury bond on deposit with the Fed to serve as collateral for the loan. The discount rate that applies to the loan is 4 percent, and the...
-
If the multiplier is 5 and investment increases by $3 billion, equilibrium real GDP will increase by: a. $2 billion. c. $8 billion. b. $3 billion. d. $15 billion. e. none of the above.
-
A 330 kg piano slides 3.60 m down a 28-degree ramp at constant speed, partially supported by a man who pushes upward, parallel to the ramp. The coefficient of kinetic friction between the piano and...
-
For all of the following words, if you move the first letter to the end of the word, and then spell the result backwards, you will get the original word: banana dresser grammar potato revive uneven...
-
Firms often involve themselves in projects that do not result directly in profits. For example, Apple, which we featured in the chapter introduction, donated $ 50 million to Stanford University...
-
The Mitchem Marble Company has a target current ratio of 2.0 but has experienced some difficulties financing its expanding sales in the past few months. At present, the firm has current assets of $...
-
In 20 years youd like to have $ 250,000 to buy a vacation home, but you have only $ 30,000. At what rate must your $ 30,000 be compounded annually for it to grow to $ 250,000 in 20 years? Use a...
-
A blender does 5000 J of work on the food in its bowl. During the time the blender runs, 2000 J of heat is transferred from the warm food to the cooler environment. What is the change in the thermal...
-
Which system contains more atoms: 5 mol of helium (A = 4) or 1 mol of neon (A = 20)? A. Helium B. Neon C. They have the same number of atoms.
-
A sample of ideal gas is in a sealed container. The temperature of the gas and the volume of the container are both increased. What other properties of the gas necessarily change? (More than one...

Study smarter with the SolutionInn App


