Colligative Properties (ALEKS lab) Introduction Lab Data Verify your mass calculation Verify your molality calculation Did...
Fantastic news! We've Found the answer you've been seeking!
Question:
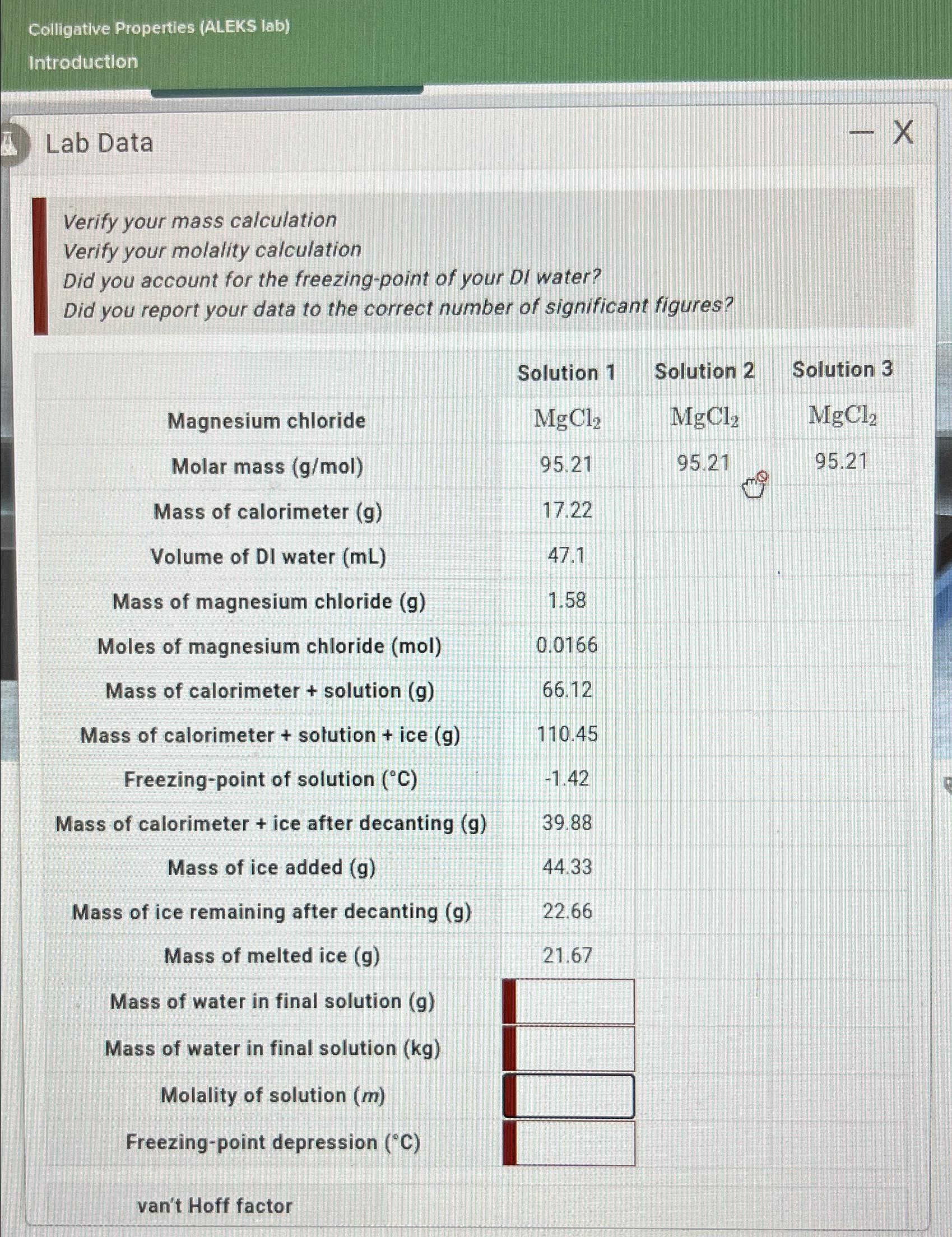
Transcribed Image Text:
Colligative Properties (ALEKS lab) Introduction Lab Data Verify your mass calculation Verify your molality calculation Did you account for the freezing-point of your DI water? Did you report your data to the correct number of significant figures? Magnesium chloride Molar mass (g/mol) Mass of calorimeter (g) Volume of DI water (mL) Mass of magnesium chloride (g) Moles of magnesium chloride (mol) Mass of calorimeter + solution (g) Mass of calorimeter + solution + ice (g) Freezing-point of solution (C) Mass of calorimeter + ice after decanting (g) Mass of ice added (g) Mass of ice remaining after decanting (g) Mass of melted ice (g) Mass of water in final solution (g) Mass of water in final solution (kg) Molality of solution (m) Freezing-point depression (C) van't Hoff factor Solution 1 MgCl 95.21 17.22 47.1 1.58 0.0166 66.12 110.45 -1.42 39.88 44.33 22.66 21.67 Solution 2 MgC1, 95.21 - X Solution 3 MgCl 95.21 Colligative Properties (ALEKS lab) Introduction Lab Data Verify your mass calculation Verify your molality calculation Did you account for the freezing-point of your DI water? Did you report your data to the correct number of significant figures? Magnesium chloride Molar mass (g/mol) Mass of calorimeter (g) Volume of DI water (mL) Mass of magnesium chloride (g) Moles of magnesium chloride (mol) Mass of calorimeter + solution (g) Mass of calorimeter + solution + ice (g) Freezing-point of solution (C) Mass of calorimeter + ice after decanting (g) Mass of ice added (g) Mass of ice remaining after decanting (g) Mass of melted ice (g) Mass of water in final solution (g) Mass of water in final solution (kg) Molality of solution (m) Freezing-point depression (C) van't Hoff factor Solution 1 MgCl 95.21 17.22 47.1 1.58 0.0166 66.12 110.45 -1.42 39.88 44.33 22.66 21.67 Solution 2 MgC1, 95.21 - X Solution 3 MgCl 95.21
Expert Answer:
Answer rating: 100% (QA)
Based on the data provided these are the calculations and verifications you need to perform 1 Verify ... View the full answer

Related Book For 

Intermediate Accounting
ISBN: 978-0324592375
17th Edition
Authors: James D. Stice, Earl K. Stice, Fred Skousen
Posted Date:
Students also viewed these chemical engineering questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Given the following expression: E = ((y'x) (x + y))' a) Draw the circuit that represents this expression as is (do not simplify). b) Simplify the expression using Boolean algebra rules. State your...
-
CPA firm brings in a yoga instructor during the tax busy season to help relieve stress of the employees. Which is true about the CPA firm's ability to take a deduction for the yoga instructor's...
-
Determine the force in member BD and the components of the reaction at C. 310 N 30 r 1.4 m 1.92 m 0.56 m
-
One of your friends states that "operations management and supply chain management are primarily of interest to manufacturing rms." Is this true or false? Give some examples to support your answer.
-
Just before hitting the ground, a partially inflated \(0.625-\mathrm{kg}\) basketball has a speed of \(3.30 \mathrm{~m} / \mathrm{s}\). Then it loses half of its kinetic energy as it bounces. (a)...
-
The vice president of operations of Rucker-Putnam Bike Company is evaluating the performance of two divisions organized as investment centers. Invested assets and condensed income statement data for...
-
Help me explaining the questions with examples of the following: Understanding Decision-Making as a Process Decision-Making and Problem Solving as a Process Decision-Making as a Strategy...
-
After Chris completed the ratio analysis for S&S Air (see Chapter 3), Mark and Todd approached him about planning for next years sales. The company had historically used little planning for...
-
Nonpolar solutes dissolve more readily in nonpolar solvents than in polar solvents. Describe the interactions occurring between the solute and the solvent. A) The induced dipole of the solute...
-
A company is evaluating an investment project with the following forecast cash flows: Year 0 1 2 3 4 Cash flow ($m) (6.5) 2.4 3.1 2.1 1.8 Using discount rates of 15% and 20%, what is the internal...
-
The McCarthys have a nanny/housekeeper whom they paid $13,000 during 2022. They did not withhold income or FICA taxes. The McCarthys paid Pennsylvania state unemployment tax of $380 in 2022. How do...
-
At the end of the 6th year, the bonds were redeemed at 102: - Write the Journal Entry Premium on Bonds Payable Balance: Amortization: $24,924.60 * 6 years = $149,547.60 Account balance: $249,246 -...
-
Slides in a report deck ________. Group of answer choices include headlines with periods at the end include headlines that are centered and have every word capitalized have less text on them than a...
-
Which statement does not apply to VA Entitlements? It is possible to use up only part of an entitlement. The amount that is used up is equal to the amount that is being guaranteed by an existing VA...
-
How much 12 ppg mud is needed to prepare exactly 250 bbls of 14 ppg mud?
-
During 2012, Cheng Book Store paid $483,000 for land and built a store in Georgetown. Prior to construction, the city of Georgetown charged Cheng $1,300 for a building permit, which Cheng paid. Cheng...
-
Using the following information, prepare a multiple-step income statement. Cost of goods sold . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . $ 8,000 Interest expense...
-
Hertzel Advertising Agency handles advertising for clients under contracts that require the agency to develop advertising copy and layouts and place ads in various media, charging clients a...
-
On December 3, Ainge Printing purchased inventory listed at $7,400 from Craig Paper Supply. Terms of the purchase were 3/10, n/20. Ainge Printing also purchased inventory from Tippetts Ink Wholesale...
-
Use the \(\gamma\)-matrices in the Weyl representation to show that the Dirac equation (14.31) is equivalent to Eq. (14.25). Data from Eq. 14.31 Data from Eq. 14.25 (y"Pu-m)(p) = (iy" - m)(p) = 0
-
Prove that the boosted right-handed spinor \(\psi_{\mathrm{R}}(\boldsymbol{p})\) is related to the corresponding rest spinor by Eq. (14.21).
-
Prove the identity \((\sigma \cdot \boldsymbol{p})^{2}=\mathrm{I}^{(2)} p^{2}\), where \(\sigma=\left(\sigma_{1}, \sigma_{2}, \sigma_{3} ight)\) are the Pauli matrices, \(\boldsymbol{p}\) is the...

Study smarter with the SolutionInn App


