Activity 2: Effect of solute concentration on cell membranes For this activity, you will investigate what...
Fantastic news! We've Found the answer you've been seeking!
Question:
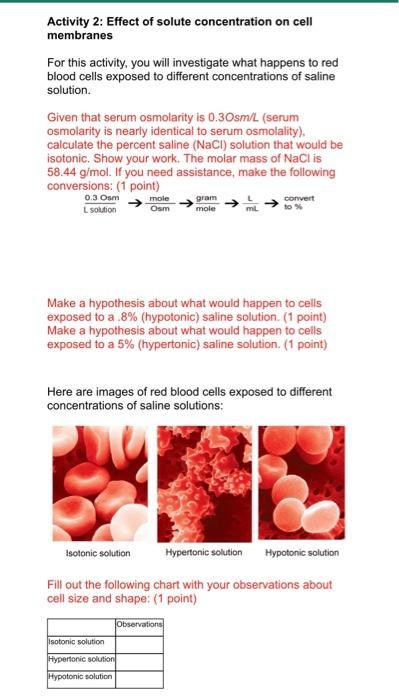
Transcribed Image Text:
Activity 2: Effect of solute concentration on cell membranes For this activity, you will investigate what happens to red blood cells exposed to different concentrations of saline solution. Given that serum osmolarity is 0.30sm/L (serum osmolarity is nearly identical to serum osmolality), calculate the percent saline (NaCl) solution that would be isotonic. Show your work. The molar mass of NaCl is 58.44 g/mol. If you need assistance, make the following conversions: (1 point) 0.3 Osm L solution mole Osm gram mole Isotonic solution L mL Make a hypothesis about what would happen to cells exposed to a .8% (hypotonic) saline solution. (1 point) Make a hypothesis about what would happen to cells exposed to a 5% (hypertonic) saline solution. (1 point) Isotonic solution Hypertonic solution Hypotonic solution convert to % Here are images of red blood cells exposed to different concentrations of saline solutions: Hypertonic solution Hypotonic solution Fill out the following chart with your observations about cell size and shape: (1 point) Observations Activity 2: Effect of solute concentration on cell membranes For this activity, you will investigate what happens to red blood cells exposed to different concentrations of saline solution. Given that serum osmolarity is 0.30sm/L (serum osmolarity is nearly identical to serum osmolality), calculate the percent saline (NaCl) solution that would be isotonic. Show your work. The molar mass of NaCl is 58.44 g/mol. If you need assistance, make the following conversions: (1 point) 0.3 Osm L solution mole Osm gram mole Isotonic solution L mL Make a hypothesis about what would happen to cells exposed to a .8% (hypotonic) saline solution. (1 point) Make a hypothesis about what would happen to cells exposed to a 5% (hypertonic) saline solution. (1 point) Isotonic solution Hypertonic solution Hypotonic solution convert to % Here are images of red blood cells exposed to different concentrations of saline solutions: Hypertonic solution Hypotonic solution Fill out the following chart with your observations about cell size and shape: (1 point) Observations
Expert Answer:
Answer rating: 100% (QA)
Calculating Percent Saline Solution To calculate the percent saline solution that would be isotonic we will use the following formula Molarity Moles of soluteVolume of Solution x 1000 Given that the s... View the full answer

Related Book For 

Posted Date:
Students also viewed these finance questions
-
Your lab instructor has asked you to measure a spring constant using a dynamic methodletting it oscillaterather than a static method of stretching it. You and your lab partner suspend the spring from...
-
Your lab partner tells you that she mixed two solutions that contain ions. You analyze the solution and find that it contains the ions and precipitate shown in the beaker. a. Write the molecular...
-
You have prepared dilute solutions of equal molar concentrations of HC2H3O2 (acetic acid), HNO2, HF, and HCN. Rank the solutions from the highest pH to the lowest pH. (Refer to Table 16.1.)
-
George bought the following amounts of Stock A over the years: (Loss amounts should be indicated with a minus sign.) Number of Adjusted Basis $ 7,200 6,270 Date Purchased Shares Stock A 11/21/1993...
-
What information would you need to fully answer the questions that IBM Rochester uses for selecting measures and indicators that are listed in Section 3 of this chapter? Where would you get this...
-
Examine if the following differential forms are closed, explaining why: (i) $A \in \Omega^{1}\left(\mathbb{R}^{2} \backslash\{0\} ight)$, defined as $$A=\frac{h c}{2 \pi e} \frac{x d y-y d...
-
The document that identifies and explains all differences between the companys record of cash and the banks record of that cash is the a. bank reconciliation. b. bank collection. C. bank statement....
-
Thomas Green College is contemplating opening a European campus where students from the main campus could go to take courses for 1 of the 4 college years. At the moment, it is considering five...
-
Use empathy mapping to understand and identify problems and opportunities for Health Care Workers during Covid-19. Implement the stages of the empathy map and present your findings to your colleagues...
-
Julio buys a koi fishpond (and fish to put in it) for his wife on their anniversary. He pays $8000 for the pond and fish with $2000 down. The dealer charges add-on interest of 3.5% per year, and...
-
why should pre participating exam ( PPE ) begin with the medcal history?
-
Consider the following apportionment problem: Use the apportionment plan requested in Problems 46-50 assuming that there must be 26 representatives. Adams' plan North: 18,200 South: 12,900 East:...
-
A group of fun-loving people have decided to play a practical joke on one of their friends, but they can't decide which friend, Alice (A), Betty (B), or Connie (C). Their preferences are: Use this...
-
Consider the following apportionment problem: Use the apportionment plan requested in Problems 46-50 assuming that there must be 26 representatives. HH's plan North: 18,200 South: 12,900 East: 17,600...
-
Use this information to answer the questions in Problems 46-49. The fraternity \(\Sigma \Delta \Gamma\) is electing a national president, and there are four candidates: Alberto (A), Bate (B), Carl...
-
Packard-Hue manufactures testing equipment at four locations, and has just hired 300 new employees. Those employees are to be apportioned using production levels at the four locations according to...
-
In Beckhard's formula for successful change, the V stands for the vision of the desired state. In Beckhard's formula for successful change, the R stands for a realistic approach to the change. Select...
-
What can you do to reduce hunger where you live? To reduce hunger globally?
-
Under the doctrine of fair representation, unions are required to represent both members and nonmembers in the bargaining unit. Do you think unions should be required to represent nonmembers? Explain.
-
Distinguish between dual-career couples and dual- earner couples and identify some of the challenges these groups face.
-
It is impossible to evaluate the effectiveness of a supervisory development program. Discuss.
-
Define a derivative, and a hedge.
-
What areas of disclosure are required for financial instruments?
-
On-the-Crest Ltd. (OCL) is a company operating in the used-vehicle industry. OCL derives its revenue from selling, licensing, and servicing software products for car dealers, from the sale of...

Study smarter with the SolutionInn App


