Compare the structure of CO and SO. Which of them has a greater dipole moment and...
Fantastic news! We've Found the answer you've been seeking!
Question:
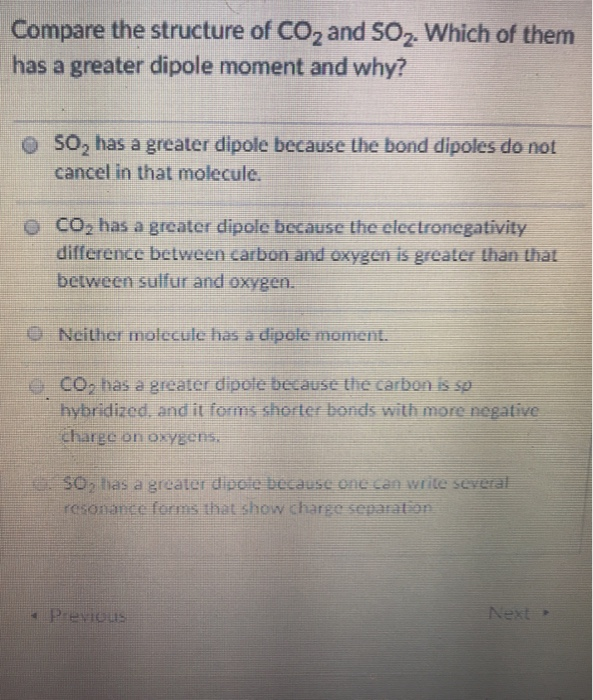
Transcribed Image Text:
Compare the structure of CO₂ and SO₂. Which of them has a greater dipole moment and why? SO₂ has a greater dipole because the bond dipoles do not cancel in that molecule. CO₂ has a greater dipole because the electronegativity difference between carbon and oxygen is greater than that between sulfur and oxygen. Neither molecule has a dipole moment. CO₂ has a greater dipole because the carbon is sp hybridized, and it forms shorter bonds with more negative charge on oxygens. SO₂ has a greater dipole because one can write several resonance forms that show charge separation Previous Next Compare the structure of CO₂ and SO₂. Which of them has a greater dipole moment and why? SO₂ has a greater dipole because the bond dipoles do not cancel in that molecule. CO₂ has a greater dipole because the electronegativity difference between carbon and oxygen is greater than that between sulfur and oxygen. Neither molecule has a dipole moment. CO₂ has a greater dipole because the carbon is sp hybridized, and it forms shorter bonds with more negative charge on oxygens. SO₂ has a greater dipole because one can write several resonance forms that show charge separation Previous Next
Expert Answer:
Answer rating: 100% (QA)
so2 has a greater dipole because the bond dipoles do not cancel in t... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
Compare the structure of multi-level governance in these four countries; Spain, Canada, Germany, and the United Kingdom. In your paper, please discuss the structures in which countries look like each...
-
Compare the structure of the FWPCA with that of the Clean Air Act.
-
Compare the structure of cyclodecane in an all-chair conformation with that of trans-decalin. Explain why all-chair cyclodecane is highly strained, and yet trans-decalin is nearly strain free. Make...
-
You measure 30 textbooks' weights, and find they have a mean weight of 72 ounces. Assume the population standard deviation is 4 ounces. Based on this, construct a 90% confidence interval for the true...
-
Norton Company has a debt-to-equity ratio of 1.65, ROA of 11.3 percent, and total equity of $1,322,796. What are the companys equity multiplier, debt ratio, and ROE?
-
Stewart received a check as a payee drawn on the Citizens Bank. The bank refused to pay the check even though there were sufficient funds in the drawers account. Stewart sued the bank on the check....
-
The one-dimensional heat equation is solved on a grid consisting of \(N\) spatial grid points and \(M\) time layers. The boundary conditions are of Dirichlet type. Calculate, in terms of \(N\) and...
-
1. Prepare a context diagram for the new system. 2. Prepare a diagram 0 DFD for the new system. 3. Write a brief memo that explains at least five attributes that you can use to define a process in...
-
[The following information applies to the questions displayed below.] A pension fund manager is considering three mutual funds. The first is a stock fund, the second is a long-term government and...
-
Gary Hudson was born and raised in Pensacola, Florida. He obtained his bachelors degree in business from Florida State University, where he enrolled in the NROTC (Naval Reserve Officers Training...
-
Real-world markets that approximate the four assumptions of the theory of perfect competition include the a. clothing market. b. stock market. c. soft drink market. d. automobile market
-
Filing a mechanic's lien can be a fantastic collections tool for your company unless you file the lien incorrectly. Regardless of where you're filing across the country, it's of paramount importance...
-
Selected current year-end financial statements of Cabot Corporation follow. (All sales were on credit; selected balance sheet amounts at December 31 of the prior year were inventory, $55,900; total...
-
An object's momentum changed from (-5.0. 1) kg m/s to (14,-14,-4) kg m/s in 1.6s. The object's mass is 5.5 kg. How much work was done on this object during this time? work-
-
Fanning Medical Equipment Company makes a blood pressure measuring kit. Jason McCoy is the production manager. The production department's static budget and actual results for Year 3 follow....
-
The diagram below shows three charges at the corners of a square of sides d = 2.50 m. Here, q = 9 = -9 and 93 = -2.5g where q = 8.00 nC. 92 9 d (a) What is the magnitude of the electric field at the...
-
What would you list as signs of psychosocial maturation over the years of middle childhood? Why did you choose these signs? What ideas do you have to effectively address bullying and why do you...
-
Planning: Creating an Audience Profile; Collaboration: Team Projects. Compare the Facebook pages of three companies in the same industry. Analyze the content on all available tabs. What can you...
-
(a) Using Figure 19.13 as a model, sketch how the entropy of water changes as it is heated from -50C to 110C at sea level. Show the temperatures at which there are vertical increases in entropy. (b)...
-
Write the molecular and structural formulas for the compounds represented by the following models: (a) (b) (c) (d) Cl Cl
-
Little is known about the properties of astatine, At, because of its rarity and high radioactivity. Nevertheless, it is possible for us to make many predictions about its properties. (a) Do you...
-
How would you factor in the absence of liquidity into your valuation?
-
An analyst who looks at real estate decides to apply the capital asset pricing model to estimate the risk (beta) for real estate. He regresses returns on a real estate index (based on appraised...
-
An alternative way of estimating risk for real estate is to use prices on traded REITs to compute returns, and to regress these returns against a stock index to arrive at a beta estimate. Would this...

Study smarter with the SolutionInn App


