COMPARING LEWIS STRUCTURES TO2/S01 H :F: :0: ++ H=0=8 :0: H A B BF4 (HO)...
Fantastic news! We've Found the answer you've been seeking!
Question:
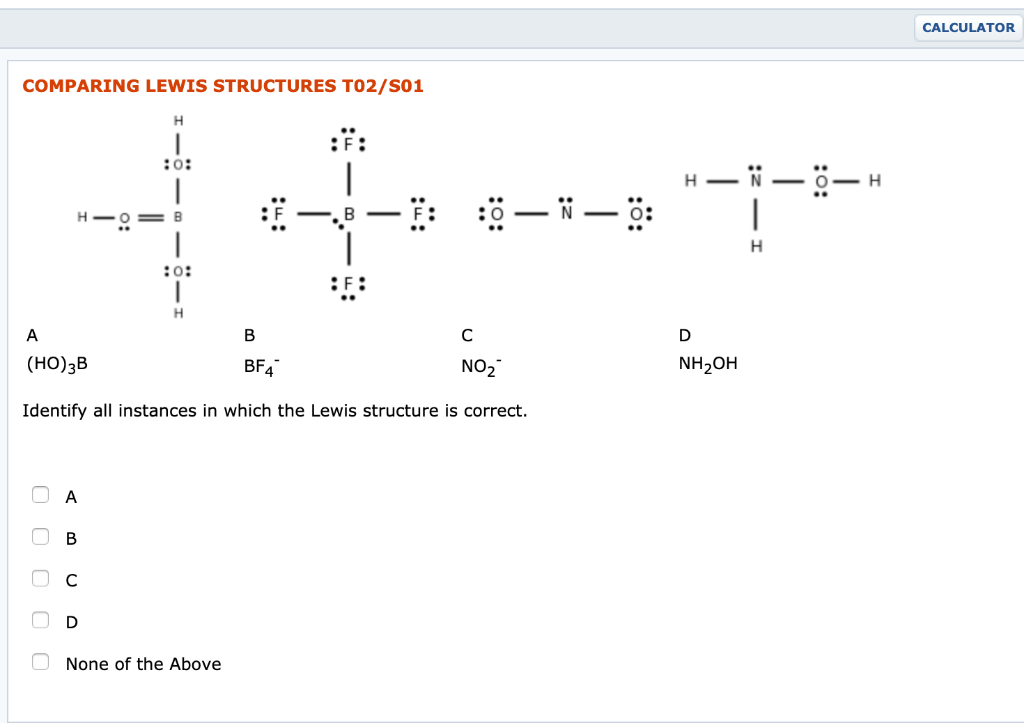
Transcribed Image Text:
COMPARING LEWIS STRUCTURES TO2/S01 H :F: :0: ++ H=0=8 :0: H A с B BF4 (HO) 3B NO₂ Identify all instances in which the Lewis structure is correct. ооооо A B None of the Above :0—N—0: H- -N-8- I H D NH₂OH CALCULATOR COMPARING LEWIS STRUCTURES TO2/S01 H :F: :0: ++ H=0=8 :0: H A с B BF4 (HO) 3B NO₂ Identify all instances in which the Lewis structure is correct. ооооо A B None of the Above :0—N—0: H- -N-8- I H D NH₂OH CALCULATOR COMPARING LEWIS STRUCTURES TO2/S01 H :F: :0: ++ H=0=8 :0: H A с B BF4 (HO) 3B NO₂ Identify all instances in which the Lewis structure is correct. ооооо A B None of the Above :0—N—0: H- -N-8- I H D NH₂OH CALCULATOR COMPARING LEWIS STRUCTURES TO2/S01 H :F: :0: ++ H=0=8 :0: H A с B BF4 (HO) 3B NO₂ Identify all instances in which the Lewis structure is correct. ооооо A B None of the Above :0—N—0: H- -N-8- I H D NH₂OH CALCULATOR COMPARING LEWIS STRUCTURES TO2/S01 H :F: :0: ++ H=0=8 :0: H A с B BF4 (HO) 3B NO₂ Identify all instances in which the Lewis structure is correct. ооооо A B None of the Above :0—N—0: H- -N-8- I H D NH₂OH CALCULATOR COMPARING LEWIS STRUCTURES TO2/S01 H :F: :0: ++ H=0=8 :0: H A с B BF4 (HO) 3B NO₂ Identify all instances in which the Lewis structure is correct. ооооо A B None of the Above :0—N—0: H- -N-8- I H D NH₂OH CALCULATOR COMPARING LEWIS STRUCTURES TO2/S01 H :F: :0: ++ H=0=8 :0: H A с B BF4 (HO) 3B NO₂ Identify all instances in which the Lewis structure is correct. ооооо A B None of the Above :0—N—0: H- -N-8- I H D NH₂OH CALCULATOR COMPARING LEWIS STRUCTURES TO2/S01 H :F: :0: ++ H=0=8 :0: H A с B BF4 (HO) 3B NO₂ Identify all instances in which the Lewis structure is correct. ооооо A B None of the Above :0—N—0: H- -N-8- I H D NH₂OH CALCULATOR COMPARING LEWIS STRUCTURES TO2/S01 H :F: :0: ++ H=0=8 :0: H A с B BF4 (HO) 3B NO₂ Identify all instances in which the Lewis structure is correct. ооооо A B None of the Above :0—N—0: H- -N-8- I H D NH₂OH CALCULATOR COMPARING LEWIS STRUCTURES TO2/S01 H :F: :0: ++ H=0=8 :0: H A с B BF4 (HO) 3B NO₂ Identify all instances in which the Lewis structure is correct. ооооо A B None of the Above :0—N—0: H- -N-8- I H D NH₂OH CALCULATOR COMPARING LEWIS STRUCTURES TO2/S01 H :F: :0: ++ H=0=8 :0: H A с B BF4 (HO) 3B NO₂ Identify all instances in which the Lewis structure is correct. ооооо A B None of the Above :0—N—0: H- -N-8- I H D NH₂OH CALCULATOR COMPARING LEWIS STRUCTURES TO2/S01 H :F: :0: ++ H=0=8 :0: H A с B BF4 (HO) 3B NO₂ Identify all instances in which the Lewis structure is correct. ооооо A B None of the Above :0—N—0: H- -N-8- I H D NH₂OH CALCULATOR COMPARING LEWIS STRUCTURES TO2/S01 H :F: :0: ++ H=0=8 :0: H A с B BF4 (HO) 3B NO₂ Identify all instances in which the Lewis structure is correct. ооооо A B None of the Above :0—N—0: H- -N-8- I H D NH₂OH CALCULATOR COMPARING LEWIS STRUCTURES TO2/S01 H :F: :0: ++ H=0=8 :0: H A с B BF4 (HO) 3B NO₂ Identify all instances in which the Lewis structure is correct. ооооо A B None of the Above :0—N—0: H- -N-8- I H D NH₂OH CALCULATOR COMPARING LEWIS STRUCTURES TO2/S01 H :F: :0: ++ H=0=8 :0: H A с B BF4 (HO) 3B NO₂ Identify all instances in which the Lewis structure is correct. ооооо A B None of the Above :0—N—0: H- -N-8- I H D NH₂OH CALCULATOR COMPARING LEWIS STRUCTURES TO2/S01 H :F: :0: ++ H=0=8 :0: H A с B BF4 (HO) 3B NO₂ Identify all instances in which the Lewis structure is correct. ооооо A B None of the Above :0—N—0: H- -N-8- I H D NH₂OH CALCULATOR COMPARING LEWIS STRUCTURES TO2/S01 H :F: :0: ++ H=0=8 :0: H A с B BF4 (HO) 3B NO₂ Identify all instances in which the Lewis structure is correct. ооооо A B None of the Above :0—N—0: H- -N-8- I H D NH₂OH CALCULATOR COMPARING LEWIS STRUCTURES TO2/S01 H :F: :0: ++ H=0=8 :0: H A с B BF4 (HO) 3B NO₂ Identify all instances in which the Lewis structure is correct. ооооо A B None of the Above :0—N—0: H- -N-8- I H D NH₂OH CALCULATOR
Expert Answer:
Answer rating: 100% (QA)
Lewis structure for A HO B Total Valence e 361 3 24 B BFY No of valence e 4 7X4 In the given opti... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Describe instances in which the interests of the management of a company might conflict with those of the shareholders and suggest mechanisms for their resolutions. Discuss your answer in close...
-
Identify instances for which feedback becomes useless in helping to control activities.
-
A second Lewis structure can be drawn for one of the nucleophiles in Problem 36. (a) Identify it and draw its alternate structure (which is simply a second resonance form), (b) Is there a second...
-
Use a calculator to express each number in Problems 39 and 40 as a decimal to the capacity of your calculator. Observe the repeating decimal representation of the rational numbers and the non...
-
What is the difference between nominal and real cash flows? Which rate of return should we use to discount each type of cash flow?
-
For the study of high school female athletes, when we use x = maximum bench press (BP) to predict y = maximum leg press (LP), we get the results that follow. The sample mean of BP was 80. a....
-
The following question concerns auditor's reports other than unqualified reports. Choose the best response. a.If an auditor issues an adverse auditor's report when there is a very material...
-
The ledger of Aiden Durant and Jasmine Adkins, attorneys-at-law, contains the following accounts and balances after adjustments have been recorded on December 31, 2012: The balance in Adkins' capital...
-
Integrative Risk and valuation Giant Enterprises' stock has a required return of 13.4%. The company, which plans to pay a dividend of $2.75 per share in the coming year, anticipates that its future...
-
The legs of a picnic table form an isosceles triangle as indicated in the figure. If mABC = 80, determine mx and my so that the top of the table will be parallel to the ground. , 80
-
Find f(x) if f(x) = and f(1) = 4.
-
Determining Unit Costs Using the Cost of Finished Goods Manufactured The accounting records of Idaho Paper Company include the following information relating to the current year. Materials...
-
Duggins Veterinary Supplies can issue perpetual preferred stock at a price of $50 a share with an annual dividend of $4.50 per share. Duggins must pay flotation costs of 5% of the issue price. What...
-
Evaluate the expression. Write answer without exponents. 15 -3
-
E=15V. R3 is replaced by an open circuit. Calculate: a. VR1 b. VR3 +wi I R1 www + Vi - V3 R3 - + | R
-
Tanner-UNF Corporation acquired as an investment $240 million of 5% bonds, dated July 1, on July 1, 2024. Company management is holding the bands in its trading portfolio The market interest rate...
-
Using the ethical concepts. identify the ethical issues demonstrated in the TNT Television series Monday Mornings, Ep. 9, "Wheels within Wheels" , discuss your thoughts and feelings on the issues...
-
Suppose that a flow network G = (V, E) violates the assumption that the network contains a path s t for all vertices V. Let u be a vertex for which there is no path s u t. Show that there must...
-
Ernst & Ernst argued that the mail rule was not relevant to its audits of First Securities since that rule only involved personal transactions of Nay and the escrow investors. Do you agree? Why or...
-
Auditing standards dont specifically discuss the audit procedures that should be applied to a clients pension-related financial statement amounts. Identify five audit procedures that would be...
-
Under what circumstances is an inventory rollback or roll-forward typically performed? How valid is the evidence yielded by this audit procedure? Explain.
-
Use lists to find the LCM of 12 and 15.
-
Using prime factorization, determine the greatest common divisor of 120 and 200.
-
There are three gym classes. The number of students in the classes is 21,35 , and 28 . What is the largest team size that can be formed if teams from every class must have the same number of students?

Study smarter with the SolutionInn App


