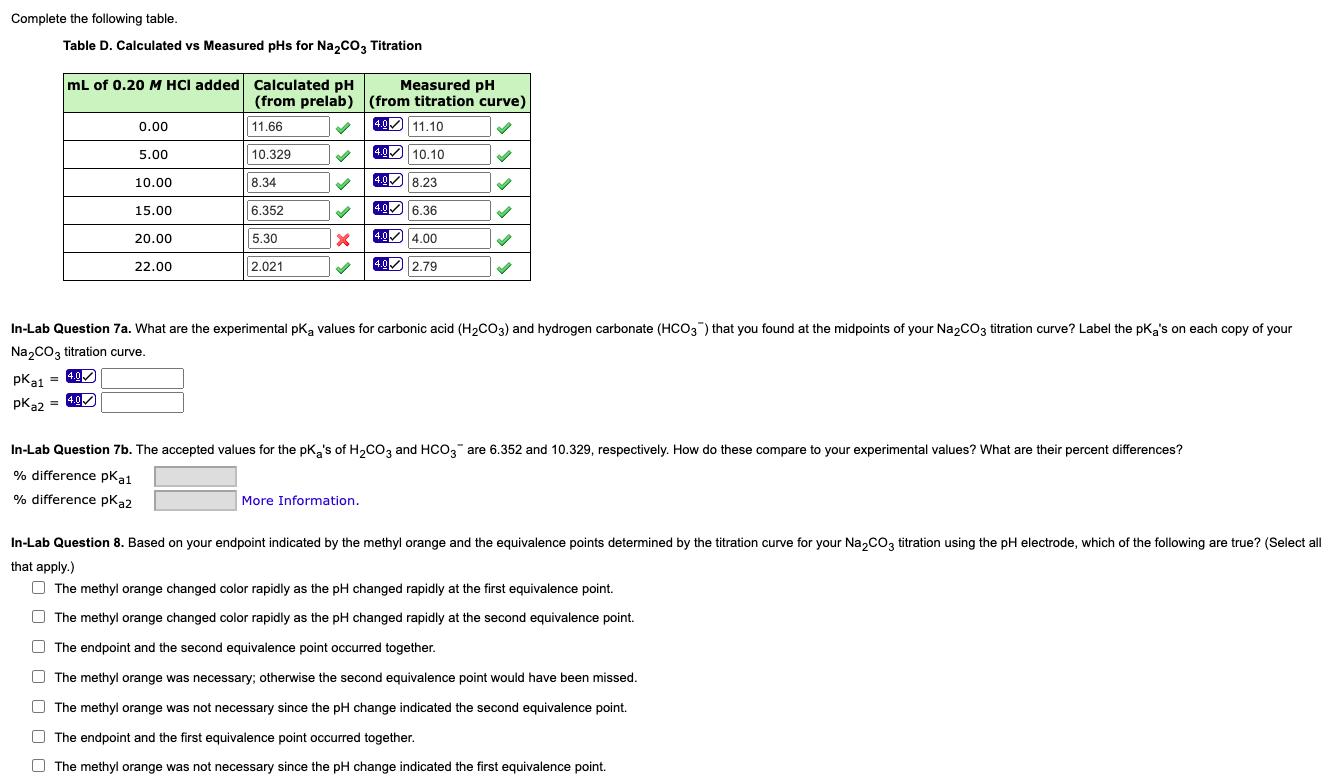
Complete the following table. Table D. Calculated vs Measured pHs for Na,co3 Titration mL of 0.20...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Complete the following table. Table D. Calculated vs Measured pHs for Na,co3 Titration mL of 0.20 M HCI added Calculated pH Measured pH (from prelab) (from titration curve) 49 11.10 0.00 11.66 4.0 5.00 10.329 4.0 10.10 8.34 40 8.23 10.00 40 6.36 15.00 6.352 5.30 494.00 20.00 2.021 40 2.79 22.00 In-Lab Question 7a. What are the experimental pką values for carbonic acid (H2CO3) and hydrogen carbonate (HCO3) that you found at the midpoints of your Na2CO3 titration curve? Label the pka's on each copy of your Na2CO3 titration curve. 4.0 pka1 = 40 pka2 = 4.0 In-Lab Question 7b. The accepted values for the pK,'s of H,Co, and HCO, are 6.352 and 10.329, respectively. How do these compare to your experimental values? What are their percent differences? % difference pKat % difference pka2 More Information. In-Lab Question 8. Based on your endpoint indicated by the methyl orange and the equivalence points determined by the titration curve for your Na,COz titration using the pH electrode, which of the following are true? (Select all that apply.) O The methyl orange changed color rapidly as the pH changed rapidly at the first equivalence point. O The methyl orange changed color rapidly as the pH changed rapidly at the second equivalence point. O The endpoint and the second equivalence point occurred together. O The methyl orange was necessary; otherwise the second equivalence point would have been missed. O The methyl orange was not necessary since the pH change indicated the second equivalence point. O The endpoint and the first equivalence point occurred together. O The methyl orange was not necessary since the pH change indicated the first equivalence point. Complete the following table. Table D. Calculated vs Measured pHs for Na,co3 Titration mL of 0.20 M HCI added Calculated pH Measured pH (from prelab) (from titration curve) 49 11.10 0.00 11.66 4.0 5.00 10.329 4.0 10.10 8.34 40 8.23 10.00 40 6.36 15.00 6.352 5.30 494.00 20.00 2.021 40 2.79 22.00 In-Lab Question 7a. What are the experimental pką values for carbonic acid (H2CO3) and hydrogen carbonate (HCO3) that you found at the midpoints of your Na2CO3 titration curve? Label the pka's on each copy of your Na2CO3 titration curve. 4.0 pka1 = 40 pka2 = 4.0 In-Lab Question 7b. The accepted values for the pK,'s of H,Co, and HCO, are 6.352 and 10.329, respectively. How do these compare to your experimental values? What are their percent differences? % difference pKat % difference pka2 More Information. In-Lab Question 8. Based on your endpoint indicated by the methyl orange and the equivalence points determined by the titration curve for your Na,COz titration using the pH electrode, which of the following are true? (Select all that apply.) O The methyl orange changed color rapidly as the pH changed rapidly at the first equivalence point. O The methyl orange changed color rapidly as the pH changed rapidly at the second equivalence point. O The endpoint and the second equivalence point occurred together. O The methyl orange was necessary; otherwise the second equivalence point would have been missed. O The methyl orange was not necessary since the pH change indicated the second equivalence point. O The endpoint and the first equivalence point occurred together. O The methyl orange was not necessary since the pH change indicated the first equivalence point.
Expert Answer:
Answer rating: 100% (QA)
InLab Question 7a pka1 40 pka2 49 InLab Question 7b The accepted values for ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Complete the following table describing the short-run daily costs of the Attractive Magnet Co. for2009. Total Total Costs Total Costs Fixed Costs Costs Total Costs Costs S 30 Average Product Total...
-
Complete the following table for a perfectly competitive firm, and indicate its profit-maximizingoutput. Total Marginal Marginal Quantity Price Revenue Revenue Total Cost Cos Total Profit $10 $30 35...
-
Complete the following table with either a yes or no regarding the attributes of a proprietorship, partnership and corporation. Proprietorship Attribute Present I. Business taxed. 2. Business entity...
-
Qualitative data can be graphically represented by using a : Line graph Frequency polygon Bar diagram Ogives
-
What is the difference between a name server and a resolver in the DNS?
-
The following table shows UK nominal GDP for the years 1990, 1992, 2008, 2010, 2019 and 2021. It also shows the GDP deflator, where 2019 = 100. 1. Work out the real GDP for each year based on 2019...
-
The random variable x represents the number of misprints in a page of a book. Determine whether the distribution is a probability distribution. If it is not a probability distribution, explain why. x...
-
Seth Fitch owns a small retail ice cream parlor. He is considering expanding the business and has identified two attractive alternatives. One involves purchasing a machine that would enable Mr. Fitch...
-
A baseball is thrown from the top of a building at a speed of 29 m / s with 54 of launch angle. The launch height is 20 m. Assume that air resistance is negligible. Find the baseball's final speed...
-
Zev enters it a contract with Aerin to buy Aerin's boat for $2,500. Zev tells Aerin he plans to use the boat to charge people to travel around the harbor to look at the Christmas lights. Zev has done...
-
Review the following website relevant to ssae-16 (soc1) at101 (soc2/3) Clarified Statements on Standards for Attestation Engagements and discuss how this would be relevant to cloud providers, GLBA,...
-
What ethical considerations arise in collaborative endeavors involving sensitive data sharing and knowledge exchange, and what governance mechanisms should be implemented to safeguard privacy and...
-
Marketing 1. How will we market coffee shop business? be detailed 2. Describe the marketing mix ( the four Ps of marketing ). BE DETAILED WITH EXAMPLES FROM A COFFEE SHOP. Product: price: point of...
-
What is the present value of the following future amount? $311,205 to be received 15 years from now, discounted back to the present at 5 percent, compounded annually.
-
The use of Clinical Decision Support Systems to facilitate the decision-making process carries with it some concerns. Some fear the use of Clinical Decision Support Systems can create dependency on...
-
Simplify. 74 y3 12n 3 21x y 3mn
-
Facial tissue Company % share 3. The following table gives US market share data in percentages for three paper product markets in 1994. Toilet paper Company % share Paper towels Company % share...
-
For all of the following words, if you move the first letter to the end of the word, and then spell the result backwards, you will get the original word: banana dresser grammar potato revive uneven...
-
Multiple Choice Questions: 1. Assume that the opportunity cost of producing a pair of pants in the United States is 2 pounds of rice, while in China, it is 5 pounds of rice. As a result, a. The...
-
The 20002001 California energy crisis produced brownouts, utility company bankruptcies, and worries about high prices. The California electric power regulatory program imposed price ceilings on...
-
Multiple Choice Questions 1. The law of diminishing marginal utility implies that the more of a commodity you consume, the a. More you value additional units of output. b. Less you value additional...
-
Because we cannot see any obvious difference between the ends of a bar magnet, could it be that like poles attract each other and unlike poles repel each other?
-
If you are constructing a parallel-plate capacitor, having air and paper as the dielectric medium between the plates, which dielectric material within the plates will make the capacitor act better?...
-
(a) Is the interaction between a charged object and an electrically neutral object always attractive? Why or why not? (b) In Figure 27.4, which type of magnetic pole is induced at the top of each...

Study smarter with the SolutionInn App


