Composition of gas and properties: C (0.875) C2 (0.083) C3 (0.021) i-C4 (0.006) -4(0.002) i-C5 (0.003)...
Fantastic news! We've Found the answer you've been seeking!
Question:

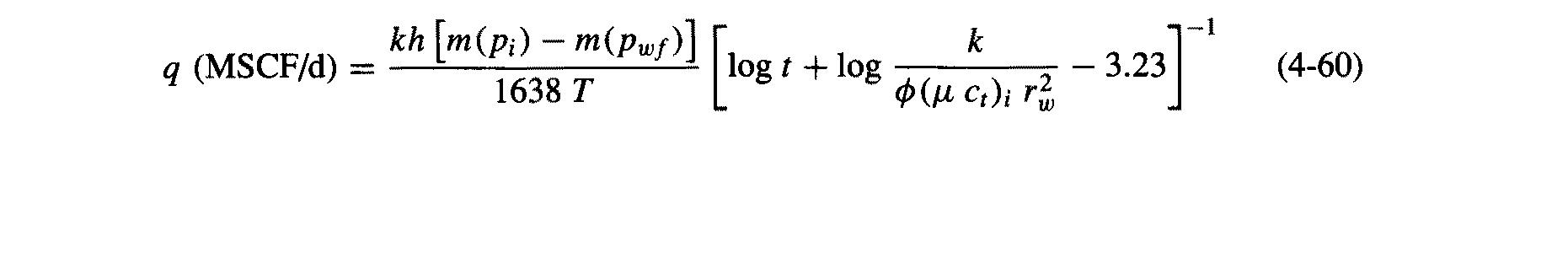

Transcribed Image Text:
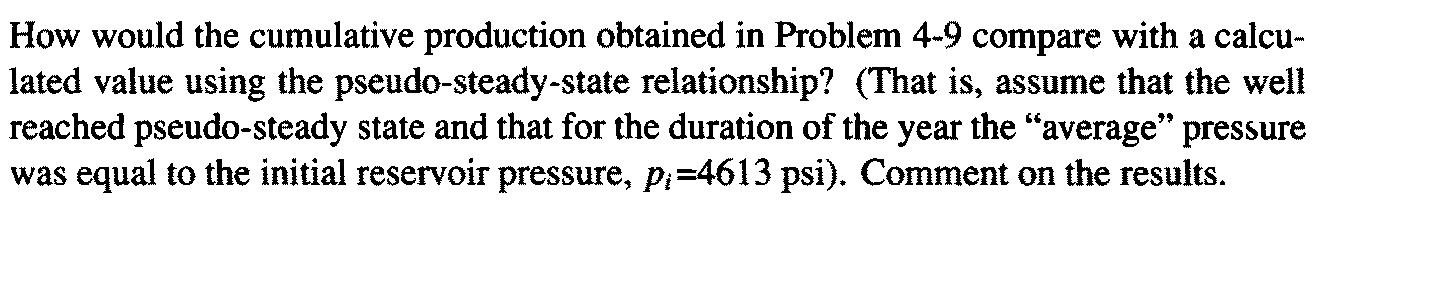
Composition of gas and properties: C (0.875) C2 (0.083) C3 (0.021) i-C4 (0.006) п-С4(0.002) i-C5 (0.003) п-Cs(0.008) п-Сo(0.001) C7+(0.001) Tpc = 378°R Ppe = 671 psi Yg = 0.65 Well and Reservoir Variables T = 180°F = 640°R Pi = 4613 psi Z; = 0.945 Hi = 0.0244 cp h = 78 ft = 0.27 0.73 $ = 0.14 rw = 0.328 ft (7 7/8-in. well) k = 0.17 md kh [m{p;) – m(Puf)] k q (MSCF/d) log t + log - 3.23 (4-60) 1638 T O (u ci); r Calculate a cumulative production curve for the gas well in Appendix C, assuming infinite-acting behavior [Eq. (4-60)] for 1 year. Bottomhole pressure is 2000 psi. What fraction of the original gas-in-place will be produced if A=4000 acres? How would the cumulative production obtained in Problem 4-9 compare with a calcu- lated value using the pseudo-steady-state relationship? (That is, assume that the well reached pseudo-steady state and that for the duration of the year the "average" pressure was equal to the initial reservoir pressure, p;=4613 psi). Comment on the results. Composition of gas and properties: C (0.875) C2 (0.083) C3 (0.021) i-C4 (0.006) п-С4(0.002) i-C5 (0.003) п-Cs(0.008) п-Сo(0.001) C7+(0.001) Tpc = 378°R Ppe = 671 psi Yg = 0.65 Well and Reservoir Variables T = 180°F = 640°R Pi = 4613 psi Z; = 0.945 Hi = 0.0244 cp h = 78 ft = 0.27 0.73 $ = 0.14 rw = 0.328 ft (7 7/8-in. well) k = 0.17 md kh [m{p;) – m(Puf)] k q (MSCF/d) log t + log - 3.23 (4-60) 1638 T O (u ci); r Calculate a cumulative production curve for the gas well in Appendix C, assuming infinite-acting behavior [Eq. (4-60)] for 1 year. Bottomhole pressure is 2000 psi. What fraction of the original gas-in-place will be produced if A=4000 acres? How would the cumulative production obtained in Problem 4-9 compare with a calcu- lated value using the pseudo-steady-state relationship? (That is, assume that the well reached pseudo-steady state and that for the duration of the year the "average" pressure was equal to the initial reservoir pressure, p;=4613 psi). Comment on the results.
Expert Answer:
Answer rating: 100% (QA)
43 Given Mole trachion of Methane 0875 Mole traction of P... View the full answer

Posted Date:
Students also viewed these physics questions
-
Composition of gas and properties: C (0.875) i-C4 (0.006) n-C5 (0.008) Tpc www.am : 378R Well and Reservoir Variables T = 180F 640R Pi = 4613 psi Zi 0.945 0.0244 cp 78 ft Mi = h Sw Sg $ C (0.083)...
-
Consider the following data set: -0.02 -0.05 -0.03 0.01 0.13 25th 50th percentile 75th percentile 0.12 0.12 Click here for the Excel Data File a-1. Calculate the 25th, 50th, and 75th percentiles....
-
In Figure V = 12 V, C1 = C4 = 2.0F, C2 = 4.0F, and C3 = 1.0F. What is the charge on capacitor 4? C2
-
The dehydration butanol of alumina is carried out over a silica-alumina catalyst at 680K. CH3CH2CH2CH20H------->cat CH3CH=CHCH3 + H2O The rate law is -r Bu = KPBU/(1+KBuPBul with k= 0.054...
-
Madison took his Porsche to the Downtown Repair Shop for some major repairs that would take three days. The repair order form prepared by the repair shop that he signed contained the following...
-
Assume that a start-up manufacturing company raises capital through a series of equity issues. a. Using the financial statement template below, summarize the financial statement effects of the...
-
If people pay for good weather, who ultimately receives the good-weather payment?
-
Prior to the distribution of cash to the partners, the accounts in the VUP Company are: Cash $24,000; Vogel, Capital (Cr.) $17,000; Utech, Capital (Cr.) $15,000; and Pena, Capital (Dr.) $8,000. The...
-
Please provide the answers and formulas, preferably through excel. Question 1 Consider the following three stocks: a) Stock A is expected to provide a dividend of $10 a share forever (starting next...
-
Two identical particles, each having charge #q, are fixed in space and separated by a distance d. A third point charge -Q is free to move and lies initially at rest on the perpendicular bisector of...
-
Dominique, age 70, has just retired. She is not interested in the stock market and explains that she would like to structure a stable income stream for herself in retirement. However, a high priority...
-
Do you ever hear someone say, Life isnt fair? Equity sensitivity is an individual difference that affects how different people react to inequity. Individuals fall along a continuum as either...
-
Marco, the Italian COO of a technology company in Mumbai, cant motivate his Indian employees. Anat, an Israeli management consultant working in the United States, struggles to give American style...
-
You are listening to music from a loudspeaker. Then a second speaker is turned on. Is it possible that the music you now hear is quieter than it was with only the first speaker playing? Explain.
-
In terms of containing healthcare costs and improving healthcare quality, do you think healthcare consumers and professionals need even more restrictions than are currently used in managed care? Are...
-
Amazon has reinvented itself again and again and thrived on organizational change. Since the company went online in 1995, the e-commerce giant has evolved under the leadership of one personJeff...
-
2. Simplify the following functions based on the given forms: a. f(x,h,y,k)=m(0,2,4,6,11,13,15) in POS form b. fa(z,m,p)=M(1,3,5,7) in SOP form
-
One hundred pounds of water at atmospheric pressure are heated from 60F to 200F. What is the enthalpy change? The internal energy change? Why is the difference between the internal energy change and...
-
At what temperature does the temperature in kelvin have the same numerical value as the temperature in \({ }^{\circ} \mathrm{F}\) ?
-
A heat engine does \(200 \mathrm{~J}\) of work while exhausting \(600 \mathrm{~J}\) of heat to the cold reservoir. What is the engine's efficiency?
-
The record time for a Tour de France cyclist to ascend the famed 1100-m-high Alpe d'Huez was \(37.5 \mathrm{~min}\), set by Marco Pantani in 1997. Pantani and his bike had a mass of \(65...

Study smarter with the SolutionInn App

