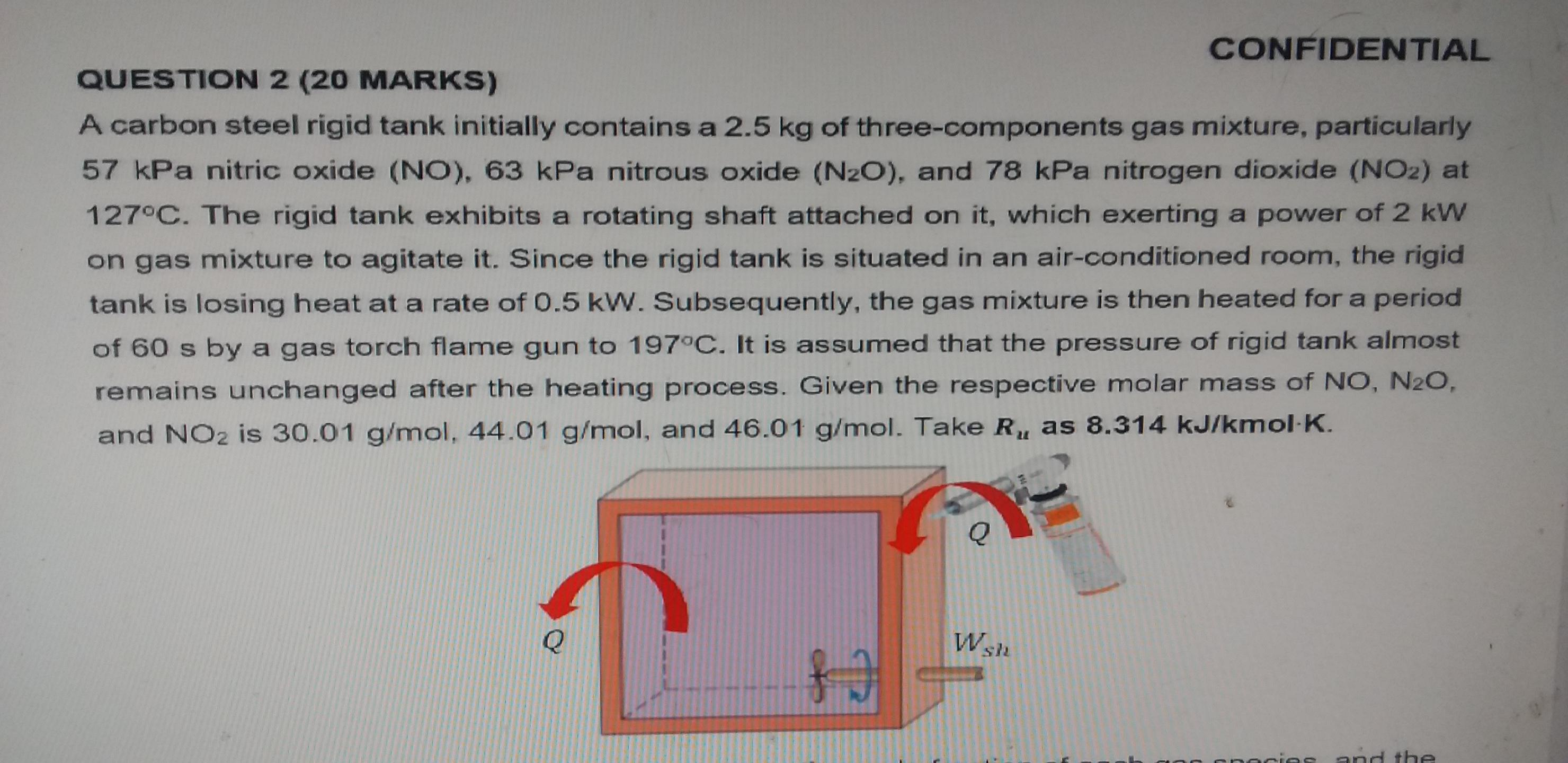
CONFIDENTIAL QUESTION 2 (20 MARKS) A carbon steel rigid tank initially contains a 2.5 kg of...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
CONFIDENTIAL QUESTION 2 (20 MARKS) A carbon steel rigid tank initially contains a 2.5 kg of three-components gas mixture, particularly 57 kPa nitric oxide (NO), 63 kPa nitrous oxide (N2O), and 78 kPa nitrogen dioxide (NO2) at 127°C. The rigid tank exhibits a rotating shaft attached on it, which exerting a power of 2 kW on gas mixture to agitate it. Since the rigid tank is situated in an air-conditioned room, the rigid tank is losing heat at a rate of 0.5 kW. Subsequently, the gas mixture is then heated for a period of 60 s by a gas torch flame gun to 197°C. It is assumed that the pressure of rigid tank almost remains unchanged after the heating process. Given the respective molar mass of NO, N2O, and NO2 is 30.01 g/mol, 44.01 g/mol, and 46.01 g/mol. Take R, as 8.314 kJ/kmol K. Q Wsh acies and the (a) Determine the total pressure of the system, the mole fraction of each gas species, and the molar mass of the gas mixture. (5 Marks) (b) Calculate the change of specific internal energy and internal energy of the gas mixture. (10 Marks) TABLE A-2 Ideal-gas specific heats of various common gases (Concluded) (c) As a function of temperature C,=a+ bT+cT² + dT (T in K, in kJ/kmol-K) Substance Formula a Nitric oxide NO -0.09395 x 10-2 5.8632 x 10-2 5.715 x 10-2 29.34 0.9747 x 10-5 -3.562 x 10-5 -3.52 x 10-s -4.187 x 10- 10.58 x 10-9 Nitrous oxide N,O 24.11 Nitrogen dioxide NO 22.9 7,87 x 10-9 (c) Determine the heat input of the gas torch flame gun to the gas mixture. (5 Marks) CONFIDENTIAL QUESTION 2 (20 MARKS) A carbon steel rigid tank initially contains a 2.5 kg of three-components gas mixture, particularly 57 kPa nitric oxide (NO), 63 kPa nitrous oxide (N2O), and 78 kPa nitrogen dioxide (NO2) at 127°C. The rigid tank exhibits a rotating shaft attached on it, which exerting a power of 2 kW on gas mixture to agitate it. Since the rigid tank is situated in an air-conditioned room, the rigid tank is losing heat at a rate of 0.5 kW. Subsequently, the gas mixture is then heated for a period of 60 s by a gas torch flame gun to 197°C. It is assumed that the pressure of rigid tank almost remains unchanged after the heating process. Given the respective molar mass of NO, N2O, and NO2 is 30.01 g/mol, 44.01 g/mol, and 46.01 g/mol. Take R, as 8.314 kJ/kmol K. Q Wsh acies and the (a) Determine the total pressure of the system, the mole fraction of each gas species, and the molar mass of the gas mixture. (5 Marks) (b) Calculate the change of specific internal energy and internal energy of the gas mixture. (10 Marks) TABLE A-2 Ideal-gas specific heats of various common gases (Concluded) (c) As a function of temperature C,=a+ bT+cT² + dT (T in K, in kJ/kmol-K) Substance Formula a Nitric oxide NO -0.09395 x 10-2 5.8632 x 10-2 5.715 x 10-2 29.34 0.9747 x 10-5 -3.562 x 10-5 -3.52 x 10-s -4.187 x 10- 10.58 x 10-9 Nitrous oxide N,O 24.11 Nitrogen dioxide NO 22.9 7,87 x 10-9 (c) Determine the heat input of the gas torch flame gun to the gas mixture. (5 Marks)
Expert Answer:

Related Book For 

Thermodynamics An Interactive Approach
ISBN: 978-0130351173
1st edition
Authors: Subrata Bhattacharjee
Posted Date:
Students also viewed these chemical engineering questions
-
A 5-ft3 rigid tank initially contains refrigerant-134a at 60 psia and 100 percent quality. The tank is connected by a valve to a supply line that carries refrigerant-134a at 140 psia and 80F. The...
-
A 100 m3 rigid tank initially contains atmospheric air at 100 kPa and 300 K is to be used as a storage vessel for compressed air at 2 MPa and 300 K. Compressed air is to be supplied by a compressor...
-
A 3-ft3 rigid tank initially contains saturated water vapor at 300F. The tank is connected by a valve to a supply line that carries steam at 200 psia and 400F. Now the valve is opened, and steam is...
-
Part A Light has both a wave and a particle nature. Particles have a wave nature as well, and therefore All particles decay with a certain characteristic lifetime. Their position can not be specified...
-
How do marketers turn marketing data into marketing knowledge?
-
Explain what HR management is and how it relates to the management process. Give examples of how HR management concepts and techniques can be of use to all managers. Explain with examples each of the...
-
A small block of wood of inertia \(m_{\text {block }}\) is released from rest a distance \(b\) above the ground, directly above your head. You decide to shoot it with your pellet gun, which fires a...
-
Elaine Jackson just had a visit from her cousin Phil. He wanted to apologize. Last year he had regaled her with stories about a small company he had discovered that had just invented a high-tech...
-
The Upton Company's most recent dividend was$1.60.Its dividend growth rate is expected to be constant at 25% for 3years, after which dividends are expected to grow at a constant rate of 10% forever....
-
A mechanical engineer at Anode Metals is considering five equivalent projects, some of which have different life expectations. Salvage value is nil for all alternatives. Assuming that the companys...
-
The function g is differentiable, and the tangent line to the graph of y= g(x) at x = 5 is y = 3x - 5. Let f(x) = -5g(x) + 5x +1. Give f'(5). 0-10 03 O-40 10 035 O None of the above
-
A cougar can run 25 miles per hour. A cheetah can run 55 miles per hour. If they both run for 3 hours at full speed, how much farther will the cheetah run?
-
29. Consider the following algorithm: for (i = 1; i < i = 1; i--) cout < < i; (a) What is the output when n = 2, n = 4, and n = 6? (b) What is the time complexity T(n)? You may assume that the input...
-
A movie club costs $30 per month, plus $15 per movie. In the store, movies cost $25 each with no monthly fee. On which movie purchase is money saved by joining the club?
-
8. Capital Records is considering several independent projects, all with positive net present values. However, a budget constraint of $1,550,000 has been placed on the capital budget for this year....
-
Mark weighs 74 pounds. Together, he and his sister weigh six pounds more than three times the weight of his sister. What is the weight, w, of Mark's sister?
-
Assume the following equation represents the value of a bond: 1000 80 (1.08) + 80 (1.08) + 80 1080 + (1.08)3 (1.08) What would be the yield to maturity for this bond?
-
Differentiate. y = ln(3x + 1) ln(5x + 1)
-
At steady state, the input shaft of a gearbox rotates at 2000 rpm while transmitting a torque of 0.2 kN-m. Due to friction, 1 kW of power is dissipated into heat and the rest is delivered to the...
-
Repeat problem 8-1-24 [OZW] assuming a regenerator with 80% effectiveness is added at the end of the last compressor. Determine (a) The thermal efficiency (th) of the cycle and (b) Back work ratio.
-
A closed system operating at steady state receives 5000 kW of heat from a source at 1500 K, produces 2000 kW of useful power and rejects the remaining heat into the atmosphere. Determine the rate at...
-
Interview three heads of household, each from a household representing a different stage of the life cycle orsocioeconomic status. Inquire about their financial planning process and their strategies...
-
Jimmy, an accountant, and Bethany just returned from their honeymoon in the Bahamas. They celebrated their marriage and the completion of Bethany's M.B.A. program. They have been encouraged by their...
-
Review the six financial accomplishments that may result from studying personal finance. In your opinion, which three are most important? Why?

Study smarter with the SolutionInn App


