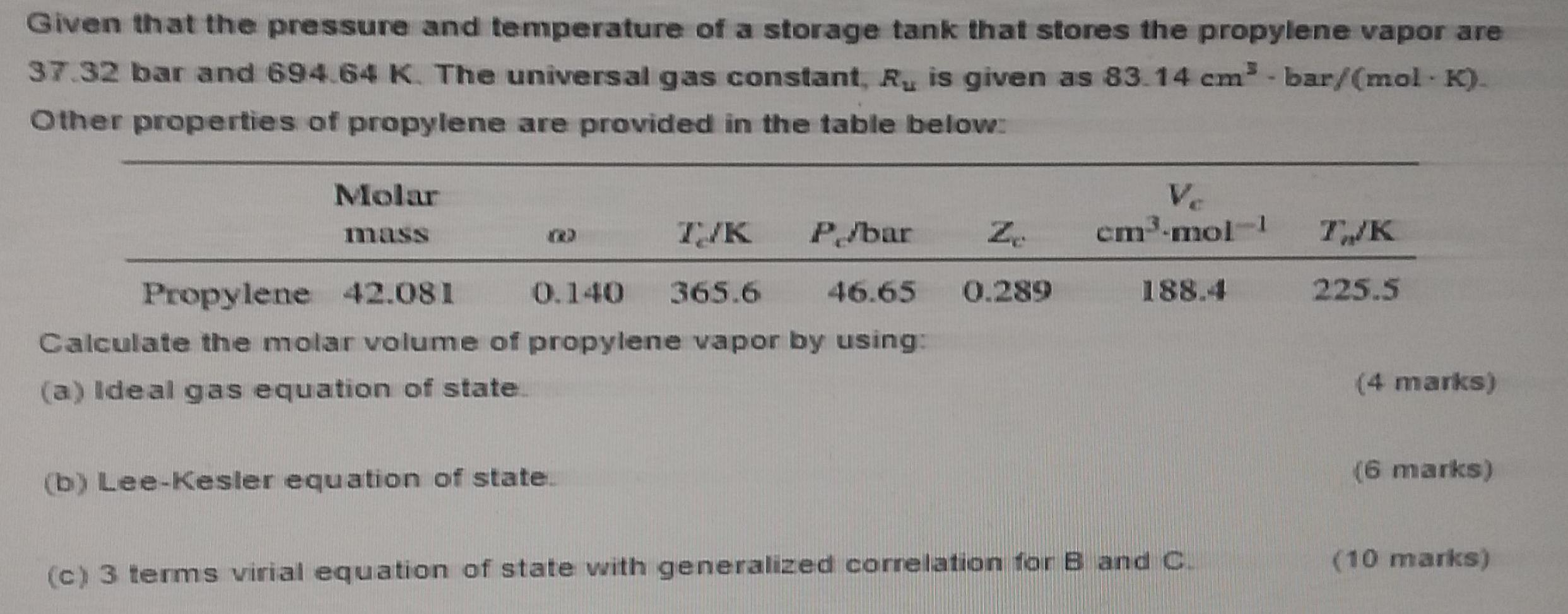
Given that the pressure and temperature of a storage tank that stores the propylene vapor are...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Given that the pressure and temperature of a storage tank that stores the propylene vapor are 37.32 bar and 694.64 K. The universal gas constant, R, is given as 83.14 cm- bar/(mol K). Other properties of propylene are provided in the table below: Molar Ve cm3-mol- mass T/K P/bar Z. TWK Propylene 42.081 0.140 365.6 46.65 0.289 188.4 225.5 Calculate the molar volume of propylene vapor by using: (a) Ideal gas equation of state (4 marks) (6 marks) (b) Lee-Kesler equation of state. (10 marks) (c) 3 terms virial equation of state with generalized correlation for B and C. Given that the pressure and temperature of a storage tank that stores the propylene vapor are 37.32 bar and 694.64 K. The universal gas constant, R, is given as 83.14 cm- bar/(mol K). Other properties of propylene are provided in the table below: Molar Ve cm3-mol- mass T/K P/bar Z. TWK Propylene 42.081 0.140 365.6 46.65 0.289 188.4 225.5 Calculate the molar volume of propylene vapor by using: (a) Ideal gas equation of state (4 marks) (6 marks) (b) Lee-Kesler equation of state. (10 marks) (c) 3 terms virial equation of state with generalized correlation for B and C.
Expert Answer:
Answer rating: 100% (QA)
a Ideal gas equation of state PV nRT P absolute pressure V volum... View the full answer

Posted Date:
Students also viewed these chemical engineering questions
-
question 3 20 marks z plc is considering a project which will necessitate the acquisition of a new machine to neutralize th
-
The pressure and temperature are measured in a tank containing air to be 60 kPa and 2 80 8 C, respectively. Determine the specific volume using the: i) Ideal-gas law ii) Van der Waals equation iii) Z...
-
What happens to the pressure and temperature of an air parcel aloft as it descends in the atmosphere?
-
Compare the ACA passed in 2010 with the Massachusetts health plan adopted in 2006 (use the Internet to obtain more details than we have provided in this chapter). Be sure to note the similarities and...
-
Explain the term value-added.
-
In a recent article in the New York Times, Jeff ZuckerCEO of NBC-Universaldescribed the digital age as one trading analog dollars for digital pennies.13 Discuss this comment from the viewpoint of...
-
What are the different sources of paid-in capital?
-
The separate incomes (which do not include investment income) of Pic Corporation and Sil Corporation, its 80 percentowned subsidiary, for 2011 were determined as follows (in thousands): During 2011,...
-
More and more, pilotless cargo aircraft are being considered and even been used by companies like FedEx and UPS. During the current supply chain crisis, where do you think the concept of probotics...
-
Journal Entry 14 15 17 19 SESSION DATE - APRIL 14, 2023 Memo #4 Dated April 8/23 From Owner: Cheque #5033 for $2 435.60 from Grande Pointe Towers was postdated and the discount period had expired. It...
-
12.5g of Iron reacts with 9.50g of Hydrogen chloride according to the equation below: Fe + 2HCl FeCl 2 + H 2 What mass of EXCESS reagent is expected to be left behind once the reaction is finished?
-
4. Determine the possible pairs of values (a, b) which make the function below continuous everywhere. 4x2 x < a f(x)=3b 4x+3x
-
Please use current tax rates, exemptions, and laws. Be sure to show ALL of your work so that I can give you partial credit. Use 2 0 2 3 tax law. Use these instructions: 2 0 2 3 Instruction 1 0 4 0 (...
-
Bradley Airport north of Hartford recently opened a new "transportation hub" next to the terminal to house car rental companies. Suppose Hertz and Avis are planning to open rental locations where...
-
Several businesses on campus - including Starbucks in the library, Einstein Brothers in the Morgan University Center, and Elements Caf in the Sunquist Building -- sell a cup of coffee. There are...
-
Jose is married filing a joint return. In 2 0 2 3 , he earned $ 1 6 0 , 0 0 0 of profit from his sole proprietorship ( after allowable deductions other than the QBI deduction ) , which operates a...
-
Consider the following distribution of prior arrests of a population of probationers. a) What measure of central tendency should we use to determine the most common number of prior arrests among the...
-
In Exercises evaluate the limit, using LHpitals Rule if necessary. lim 07x cos x X
-
Describe what a badly run stock exchange would be like and explain how society would be poorer as a result.
-
Many countries are encouraging small investors to buy quoted shares. Why might they be doing this? Write an essay to include a discussion of the societal benefits of a stock exchange.
-
Frame-up plc is considering a flotation on the Main Market of the London Stock Exchange. The managing director has asked you to produce a 1,000-word report explaining the advantages of such a move.

Study smarter with the SolutionInn App

