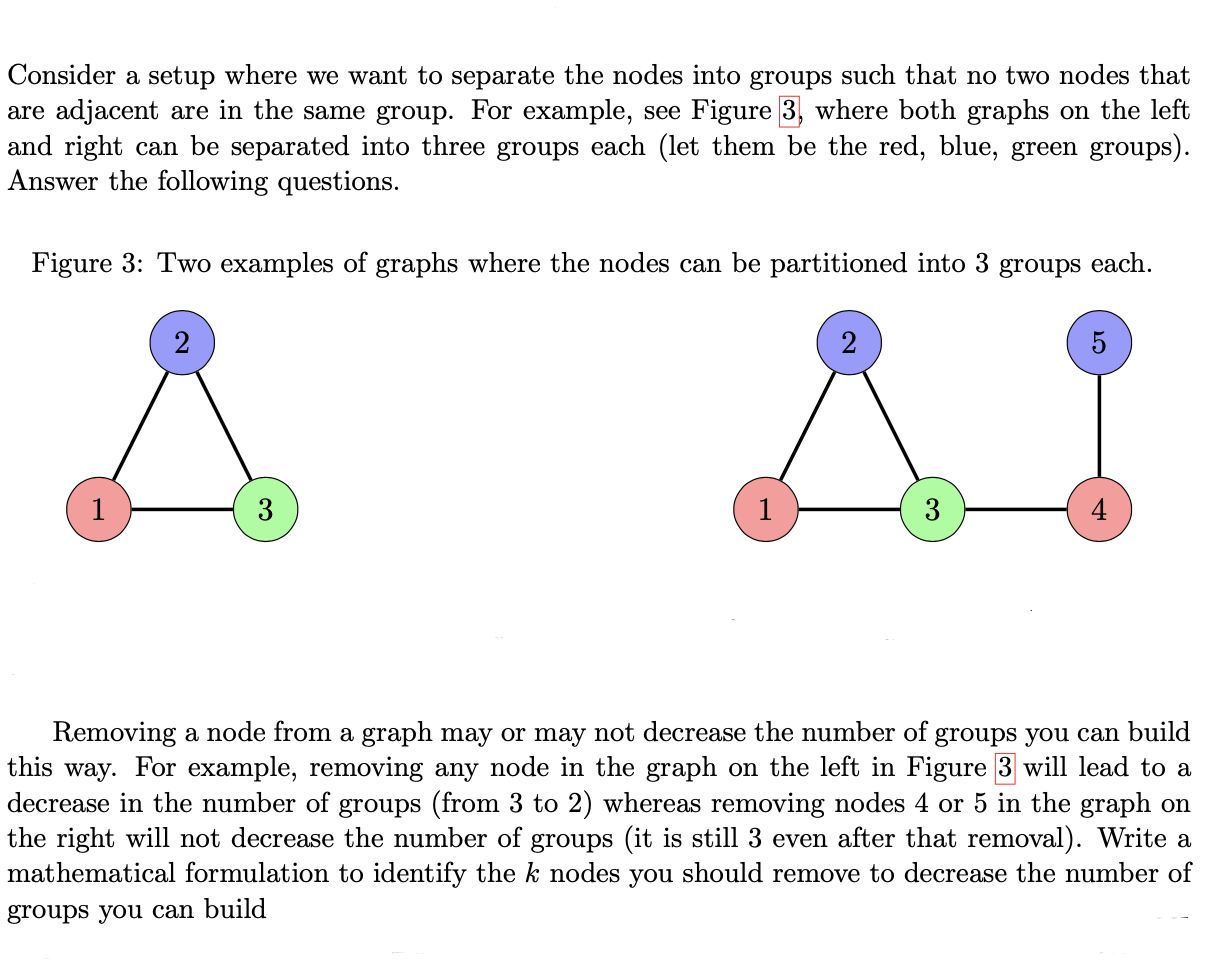
Consider a setup where we want to separate the nodes into groups such that no two...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Consider a setup where we want to separate the nodes into groups such that no two nodes that are adjacent are in the same group. For example, see Figure 3, where both graphs on the left and right can be separated into three groups each (let them be the red, blue, green groups). Answer the following questions. Figure 3: Two examples of graphs where the nodes can be partitioned into 3 groups 3 3 each. Cr 5 Removing a node from a graph may or may not decrease the number of groups you can build this way. For example, removing any node in the graph on the left in Figure 3 will lead to a decrease in the number of groups (from 3 to 2) whereas removing nodes 4 or 5 in the graph on the right will not decrease the number of groups (it is still 3 even after that removal). Write a mathematical formulation to identify the k nodes you should remove to decrease the number of groups you can build Consider a setup where we want to separate the nodes into groups such that no two nodes that are adjacent are in the same group. For example, see Figure 3, where both graphs on the left and right can be separated into three groups each (let them be the red, blue, green groups). Answer the following questions. Figure 3: Two examples of graphs where the nodes can be partitioned into 3 groups 3 3 each. Cr 5 Removing a node from a graph may or may not decrease the number of groups you can build this way. For example, removing any node in the graph on the left in Figure 3 will lead to a decrease in the number of groups (from 3 to 2) whereas removing nodes 4 or 5 in the graph on the right will not decrease the number of groups (it is still 3 even after that removal). Write a mathematical formulation to identify the k nodes you should remove to decrease the number of groups you can build
Expert Answer:

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these computer network questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
You and your spouse have earned salary and wages of $41,750. In addition you have municipal bond interest income of $600 and savings account and certificate of deposit interest income of $800. You...
-
1. Ten students in a physical education class are to be divided into five-member teams for a basketball game. In how many ways can the two teams be selected? 2. In how many ways can 12 sweaters be...
-
In Raffles v. Wichelhaus, 2 Hurl. & C. 906, 159 Eng. Rep. 375 (Ex. 1864), the plaintiff sold the defendants 125 bales of cotton to arrive ex Peerless from Bombay, that is, by way of the ship Peerless...
-
John J. Williams began working at Delaware Elevator as a branch sales manager in late 2004. The company manufactures components for elevators and installs and repairs elevators. It employs 175 people...
-
Cornucopia Inc. is planning to invest in new manufacturing equipment to make a new garden tool. The new garden tool is expected to generate additional annual sales of 4,000 units at $ 68 each. The...
-
7. 8. The work done by a gas molecule in an isolated system is given by W=ae KT, where x is the displacement, k is the Boltzmann constant and T is the temperature. and are constants. Then the...
-
George Pharmacy is a pharmaceutical salesman who has been very successful at his job in the last few years. Unfortunately, his family life has not been very happy. Three years ago, his only child,...
-
The Witness test for feline leukemia has a 92.9% sensitivity and 96.5% specificity. Assume for this question that 2% of cats in a population actually have the disease. a) Draw a tree diagram...
-
You have been asked to value Oneida Steel, a midsize steel company. The firm reported $80 million in net income, $50 million in capital expenditures, and $20 million in depreciation in the...
-
The benefits of joining the Texas nurse's Association include all of the following except- The membership is free Exclusive resources and access to publications Discounts through other partnerships...
-
When the parent and subsidiary have differing fiscal periods, the fiscal period of the subsidiary can be changed or the financial statements of the subsidiary can be adjusted each period. Current...
-
Which campaign finance loophole was exploited by the energy company Enron, leading to the passage of the Bipartisan Campaign Finance Reform Act of 2002? Multiple choice question. contributions by...
-
A retail company is considering investing in a new IT system for online sales. This would involve investing 10 million in servers and computer software that would have an expected useful life of 3...
-
Restructuring the liabilities might positively influence the ROE, but will negatively influence the service level of the company agree or disagree?
-
Explain the operation of the dividends received deduction.
-
Ken (age 31) and Amy (age 28) Booth have brought you the following information regarding their income, expenses, and withholding for the year. They are unsure which of these items must be used to...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family. The Incisors own a rental beach house in Hawaii. The beach house was rented for the full year during 2012...
-
During 2012, William purchases the following capital assets for use in his catering business: New passenger automobile (September 30)........................$21,500 Baking equipment (June 30)...
-
When you exhale, all of the air in your lungs must exit through the trachea. If you exhale through your nose, this air subsequently leaves through your nostrils. The area of your nostrils is less...
-
A balloon is launched at sea level, where the air pressure is \(100 \mathrm{kPa}\). The helium has a volume of \(1000 \mathrm{~m}^{3}\) at this altitude. What is the volume of the helium when the...
-
A balloon is launched at sea level, where the air pressure is \(100 \mathrm{kPa}\). The density in the hot-air chamber is \(1.0 \mathrm{~kg} / \mathrm{m}^{3}\). What is the density of the air when...

Study smarter with the SolutionInn App


