Consider a system consisting of an ideal gas confined within a container, one wall of which...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
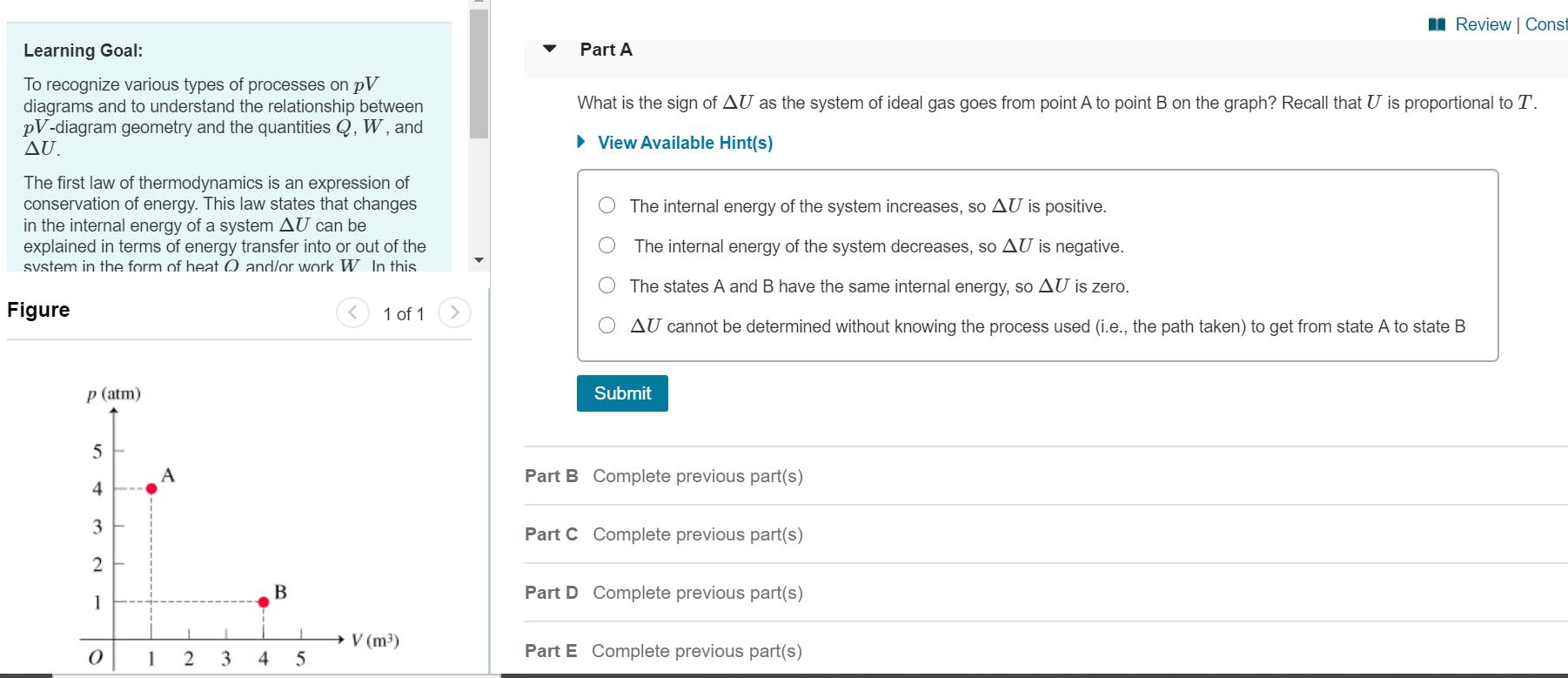
Consider a system consisting of an ideal gas confined within a container, one wall of which is a movable piston. Energy can be added to the gas in the form of heat by applying a flame to the outside of the container. Conversely, energy can also be removed from the gas in the form of heat by immersing the container in ice water. Energy can be added to the system in the form of work by pushing the piston in, thereby compressing the gas. Conversely, if the gas pushes the piston out, thereby pushing some atmosphere aside, the internal energy of the gas is reduced by the amount of work done. The internal energy of an ideal gas is directly proportional to its absolute temperature T. An ideal gas also obeys the ideal gas law pV = nRT, so the absolute temperature T is directly proportional to the product of the absolute pressure p and the volume V. Heren denotes the amount of gas in moles, which is a constant because the gas is confined, and R is the universal gas constant. ApV diagram (Figure 1) is a convenient way to track the pressure and volume of a system. Energy transfers by heat and/or work are associated with processes, which are lines or curves on the pV diagram taking the system from one state (i.e., one point on the diagram) to another. Work corresponds geometrically to the area under the curve on a pV diagram. If the volume increases (i.e., the system expands) the work will be classified as an energy output from the system. I Review | Const Learning Goal: Part A To recognize various types of processes on pV diagrams and to understand the relationship between pV-diagram geometry and the quantities Q, W, and AU. What is the sign of AU as the system of ideal gas goes from point A to point B on the graph? Recall that U is proportional to T. • View Available Hint(s) The first law of thermodynamics is an expression conservation of energy. This law states that changes in the internal energy of a system AU can be explained in terms of energy transfer into or out of the svstem in the form of heat 0 and/or work W. In this The internal energy of the system increases, so AU is positive. The internal energy of the system decreases, so AU is negative. The states A and B have the same internal energy, so AU is zero. Figure 1 of 1 AU cannot be determined without knowing the process used (i.e., the path taken) to get from state A to state B p (atm) Submit 5 A Part B Complete previous part(s) 4 3 Part C Complete previous part(s) В Part D Complete previous part(s) 1 V (m3) 1 3 4 Part E Complete previous part(s) Consider a system consisting of an ideal gas confined within a container, one wall of which is a movable piston. Energy can be added to the gas in the form of heat by applying a flame to the outside of the container. Conversely, energy can also be removed from the gas in the form of heat by immersing the container in ice water. Energy can be added to the system in the form of work by pushing the piston in, thereby compressing the gas. Conversely, if the gas pushes the piston out, thereby pushing some atmosphere aside, the internal energy of the gas is reduced by the amount of work done. The internal energy of an ideal gas is directly proportional to its absolute temperature T. An ideal gas also obeys the ideal gas law pV = nRT, so the absolute temperature T is directly proportional to the product of the absolute pressure p and the volume V. Heren denotes the amount of gas in moles, which is a constant because the gas is confined, and R is the universal gas constant. ApV diagram (Figure 1) is a convenient way to track the pressure and volume of a system. Energy transfers by heat and/or work are associated with processes, which are lines or curves on the pV diagram taking the system from one state (i.e., one point on the diagram) to another. Work corresponds geometrically to the area under the curve on a pV diagram. If the volume increases (i.e., the system expands) the work will be classified as an energy output from the system. I Review | Const Learning Goal: Part A To recognize various types of processes on pV diagrams and to understand the relationship between pV-diagram geometry and the quantities Q, W, and AU. What is the sign of AU as the system of ideal gas goes from point A to point B on the graph? Recall that U is proportional to T. • View Available Hint(s) The first law of thermodynamics is an expression conservation of energy. This law states that changes in the internal energy of a system AU can be explained in terms of energy transfer into or out of the svstem in the form of heat 0 and/or work W. In this The internal energy of the system increases, so AU is positive. The internal energy of the system decreases, so AU is negative. The states A and B have the same internal energy, so AU is zero. Figure 1 of 1 AU cannot be determined without knowing the process used (i.e., the path taken) to get from state A to state B p (atm) Submit 5 A Part B Complete previous part(s) 4 3 Part C Complete previous part(s) В Part D Complete previous part(s) 1 V (m3) 1 3 4 Part E Complete previous part(s)
Expert Answer:
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these operating system questions
-
Consider a system consisting of an ice cube. (a) Under what conditions can the ice cube melt reversibly? (b) If the ice cube melts reversibly, E is zero for the process? Explain.
-
Consider a system consisting of two oppositely charged spheres hanging by strings and separated by a distance r1, as shown in the accompanying illustration. Suppose they are separated to a larger...
-
Consider a system consisting of four resources of the same type that are shared by three processes, each of which needs at most two resources. Show that the system is deadlock free.
-
The following information has been extracted from the trial balance of M/s Randhir Transport Corporation. Adjustments 1. Closing stock for the year was Rs. 35,500. 2. Depreciation charged on plant...
-
What industries are more likely to hire contingent workers?
-
Is proximity to known outbreaks of WNV a factor in its spread? Looking at the map in the following figure , describe geographic factors that seem to influence the spread of WNV. Explain. Year...
-
What are the four dimensions of ITIL4 ?
-
Assume that you have an after-tax cost of capital of 10 percent. Compute the net present value of each of the five projects listed in the following table. Rank the above projects from 1 (best for...
-
What does Athens have to do with Jerusalem?" That's the question asked by the early Church FatherTertullianin his rejection of rhetoric and non-Christian learning.But like many of the other early...
-
Better Fitness, Inc. (BFI) manufactures exercise equipment at its plant in Freeport, Long Island. It recently designed two universal weight machines for the home exercise market. Both machines use...
-
In a certain railroad layout of the subgrade, it was decided to connect a - 5% grade with + 3% grade by a 140 m vertical parabolic sag curve. In the actual construction work, it was found out that...
-
Before you can use a Servlet such as FacesServlet on the server side from a Web browser, you need to map the FacesServlet to a path in your deployment descriptor file . a. Web pages b. WEB INF file...
-
Use the Java CallableStatement method to develop a data query from the Student and StudentCourse Tables with the StudentFrame class in the OracleSelectFaculty project (the project file can be found...
-
Navigation from one page to another can be done in two ways. One is directly to use code by writing a JSP tag such as or an HTML hyperlink in the JSF file. Another way that is provided by JSF is to...
-
UsePL-SQL to create a package, Student_Course, in the Oracle SQL Developer. The package contains a stored procedure namedSelectStudentCourse() with two positional parameters, a. The input student...
-
When using the JFileChooser object to select an image, the returned image is a Java File object, and one needs to convert it to a Byte[] array to be inserted into a column in a database Table....
-
Starting from rest, a ball rolls down a ramp with uniform acceleration. It travels 2 m in 1 seconds. (a) What is its average velocity over this time? V x m/s (b) How far does it travel after 2...
-
The Higher the time period of the financial security the higher the. ............... risk. O a. Maturity O b. Default and Maturity Oc. Default O d. Liquidity
-
(a) What does the term electromotive force mean? (b) What is the definition of the volt? (c) What does the term cell potential mean?
-
Calculate the concentration of an aqueous solution of NaOH that has a pH of 11.50.
-
(a) What is the meaning of the term coordination number as it applies to metal complexes? (b) Give an example of a ligand that is neutral and one that is negatively charged. (c) Would you expect...
-
S1 Ltd and S2 Ltd belong to the same capital gains group. In May 2020, S1 Ltd transferred a chargeable asset to S2 Ltd. The original cost of this asset to S1 Ltd was 10,000 and its market value in...
-
The ordinary share capital of W Ltd (which is a trading company) is owned 30% by X Ltd, 25% by Y Ltd and 45% by Z Ltd. All of these companies are UK resident and they prepare accounts to 31 March....
-
E Ltd has taxable total profits of 800,000 for the year to 31 March 2024 and receives no dividends. For many years, the company has owned 65% of the ordinary shares of F Ltd and 30% of the ordinary...

Study smarter with the SolutionInn App



