Consider an ideal gas enclosed in a 1.00 L container at an internal pressure of 10.0 atm.
Fantastic news! We've Found the answer you've been seeking!
Question:
Consider an ideal gas enclosed in a 1.00 L container at an internal pressure of 10.0 atm.
Transcribed Image Text:
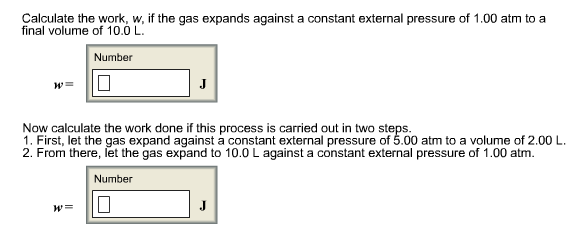
Calculate the work, w, if the gas expands against a constant external pressure of 1.00 atm to a final volume of 10.0 L. Number W= J Now calculate the work done if this process is carried out in two steps. 1. First, let the gas expand against a constant external pressure of 5.00 atm to a volume of 2.00 L. 2. From there, let the gas expand to 10.0 L against a constant external pressure of 1.00 atm. Number 0 W= J Calculate the work, w, if the gas expands against a constant external pressure of 1.00 atm to a final volume of 10.0 L. Number W= J Now calculate the work done if this process is carried out in two steps. 1. First, let the gas expand against a constant external pressure of 5.00 atm to a volume of 2.00 L. 2. From there, let the gas expand to 10.0 L against a constant external pressure of 1.00 atm. Number 0 W= J
Expert Answer:
Answer rating: 100% (QA)
The problem is based on the concept of thermodynamics Thermodynamics deals with the change with ener... View the full answer

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these chemistry questions
-
When 20g of CACO3 were put into 10 litre flask and heated to 800 C, 35% CACO, remained unreacted at equilibrium, Kp for decomposition of CACO3 is (A) 1.145 atm (B) 0.145 atm (C) 2.145 atm (D) 3.145...
-
Consider an ideal gas at 300 K and 100 kPa. As a result of some disturbance, the conditions of the gas change to 305 K and 96 kPa. Estimate the change in the specific volume of the gas using (a) Eq....
-
Consider an ideal gas refrigeration cycle using helium as the working fluid. Helium enters the compressor at 100 kPa and -10C and compressed to 250 kPa. Helium is then cooled to 20C before it enters...
-
A heat engine operates between two reservoirs at 800 and 20C. One-half of the work output of the heat engine is used to drive a Carnot heat pump that removes heat from the cold surroundings at 2C and...
-
Shareholders of General Electric Company have asked the board of directors to include several shareholder proposals in its annual proxy statement. One proposal is that GE's articles of incorporation...
-
Key figures for Polaris and Arctic Cat follow. Required 1. Compute the recent two years cash flow on total assets ratios for Polaris and Arctic Cat. 2. What does the cash flow on total assets ratio...
-
The following data are for Marvin Department Store. The account balances (in thousands) are for 2017. 1. Compute (a) the cost of goods purchased and (b) the cost of goods sold. 2. Prepare the income...
-
Zeigler Manufacturing Company purchased a robot for $720,000 at the beginning of year 1. The robot has an estimated useful life of four years and an estimated residual value of $60,000. The robot,...
-
. Conduct research and review the pricing models for Amazon Web Services and Microsoft's Azure. . How are they similar and how are they different?
-
Consolidation related simulation example: Millennium Capital Management, Inc., (MCM) acquired a 90% interest in NextGen, Inc. MCM's Financial Manager, Matthew Steven, has prepared a draft memo to the...
-
a.) Suppose y1=t^2y1+4y2+sec(t) y2=sin(t)y1+ty26. This system of linear differential equations can be put in the form y'= P(t) y + g (t). Determine P(t) and g (t) b.) Suppose (t+3)y1=5ty1+9y2...
-
List three of Zandvliet and Anderson's principles of effective community engagement and participation. . A BI i 80
-
To best use Fred's pension, Carlos suggested that his client's Fred and Ethel use pension maximization. This would entail the sale of what type of insurance? Explain.
-
One person has estimated that with 500,000 dolars per year, he will be able to live worthily when he gets older and the first thing he did was to look for some unconventional alternative to deposit...
-
Discuss the atomic instruments and physiological importance of electivejoining in eukaryotic quality expression, with a center on its part in producing protein differences and its suggestions for...
-
The static budget for Practical Company is based on expected sales of 20,000 units for the upcoming period. At this sales volume, its budgeted income is as follows: Total Sales $1,000,000 Less...
-
Recently, Jamie and Jake each bought new cars. Both received a loan from a local bank with a rate of 10% where payments are made at the end of each month, and they both pay the same monthly payment....
-
The following table shows the rates of total return in successive years from 2004 to 2008 for the Sprott Canadian Equity Fund and for the benchmark Toronto Stock Exchange S&P/TSX Composite Index. By...
-
Repeat Prob. 14-67 for a heat removal rate of 950kJ/min. Prob. 14-67 Air enters a 30-cm-diameter cooling section at 1 atm, 35oC, and 45 percent relative humidity at 18 m/s. Heat is removed from the...
-
Argon gas enters a turbine at 1000 psia and 1000 R with a velocity of 300 ft/s and leaves at 150 psia and 500 R with a velocity of 450 ft/s at a rate of 12 lbm/s. Heat is being lost to the...
-
Air enters a turbojet engine at 320 m/s at a rate of 30 kg/s, and exits at 650 m/s relative to the aircraft. The thrust developed by the engine is (a) 5 kN (b) 10 kN (c) 15 kN (d) 20 kN (e) 26 kN
-
Enter up a columnar purchases day book with columns for the various expenses for J. Still for the month from the following information on credit items. 2016 January f 1 Bought goods from H. Graham...
-
A Enter up a columnar purchases day book with columns for the various expenses for F. Graham for the month from the following information on credit items. 2016 June Bought goods from J. Syme 4 Bought...
-
Enter up the relevant accounts in the purchases and general ledgers from the columnar purchases day book you completed for Review Question 20.4A. Data From Review Question 20.4A 20.4A Enter up a...

Study smarter with the SolutionInn App


