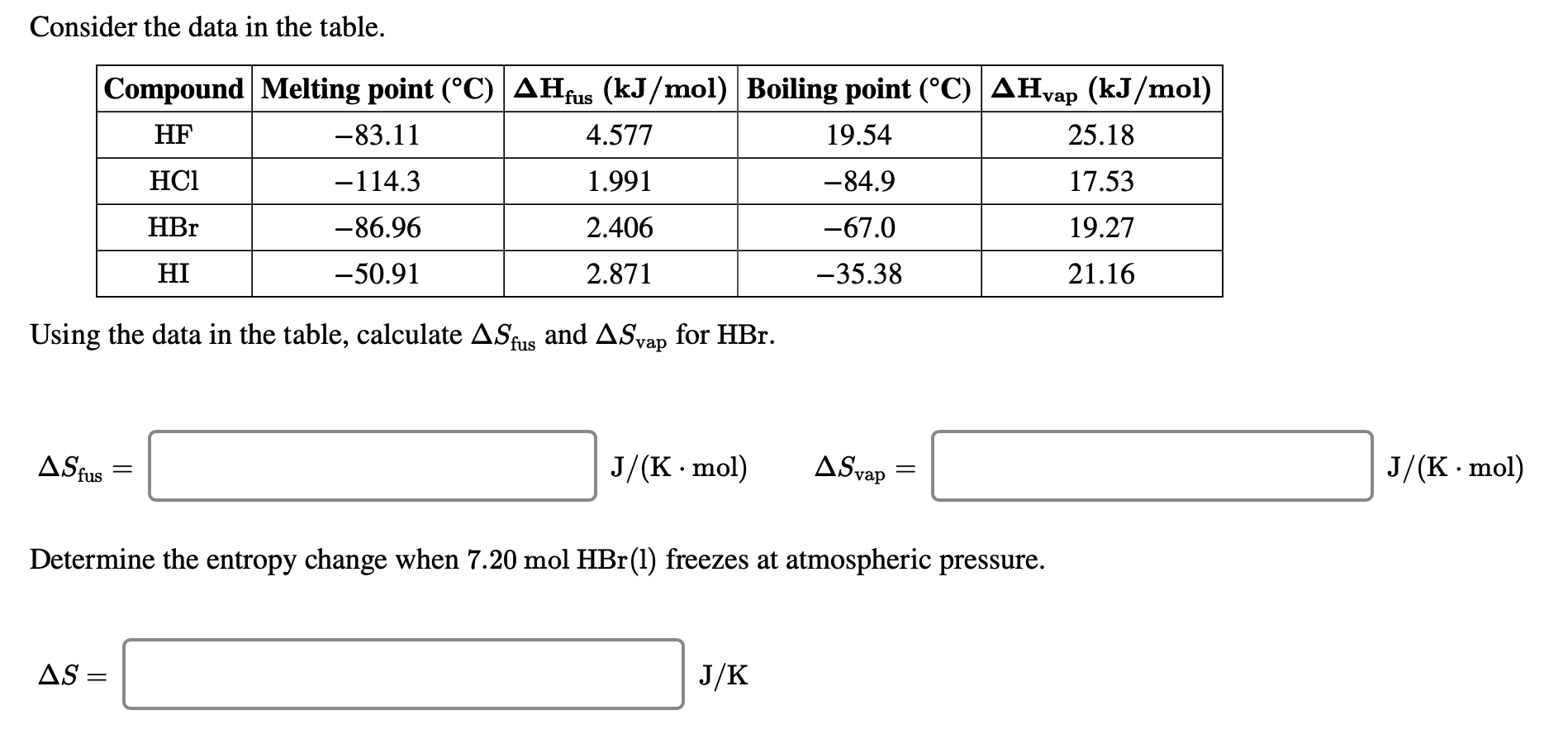
Consider the data in the table. Compound Melting point (C) AHfus (kJ/mol) Boiling point (C) AHvap...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Consider the data in the table. Compound Melting point (°C) AHfus (kJ/mol) Boiling point (°C) AHvap (kJ/mol) HF -83.11 19.54 HC1 -114.3 -84.9 HBr -86.96 -67.0 HI -50.91 -35.38 4.577 1.991 2.406 2.871 Using the data in the table, calculate ASfus and ASvap for HBr. AS fus = AS = J/(K. mol) AS vap Determine the entropy change when 7.20 mol HBr (1) freezes at atmospheric pressure. J/K = 25.18 17.53 19.27 21.16 J/(K.mol) Consider the data in the table. Compound Melting point (°C) AHfus (kJ/mol) Boiling point (°C) AHvap (kJ/mol) HF -83.11 19.54 HC1 -114.3 -84.9 HBr -86.96 -67.0 HI -50.91 -35.38 4.577 1.991 2.406 2.871 Using the data in the table, calculate ASfus and ASvap for HBr. AS fus = AS = J/(K. mol) AS vap Determine the entropy change when 7.20 mol HBr (1) freezes at atmospheric pressure. J/K = 25.18 17.53 19.27 21.16 J/(K.mol)
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
sauiod sz9 Save Ar A ball of mass 0.25 kg is thrown towards a wall with a speed of 12 m/s. It is observed that the ball bounces back from the wall in a straight line having a speed of 8.0 m/s. If the...
-
Consider the data in Table 12.1. Construct a bounded adjustment chart using ï¬ = 0.3 and L = 10. Compare the performance of this chart to the one in Table 12.1 and Figure 12.12. TABLE 12.1...
-
Consider the data in Table 12.1. Construct a bounded adjustment chart using = 0.4 and L = 10. Compare the performance of this chart to the one in Table 12.1 and Fig. 12.12.
-
1. Estimate the model Debt = 30 + 1lnc + 32Unemp + . Is unemployment significant at the 5% level? 2. You are told that the unemployment rate might have a quadratic influence on monthly debt payments....
-
On January 22, 2002, as a result of a real estate transaction, First Service Title Agency issued three checks drawn on its account with Key Bank. The first check was for $850 and was payable to the...
-
If a company that uses IFRS discovers an error but determines that it is impracticable to restate its financial statements for all prior periods, what alternatives does IFRS allow? How does this...
-
Refer to the information in Exercise 16-12. Prepare a process cost summary using the FIFO method. (Round cost per equivalent unit calculations to two decimal places.) Data From Exercise 16-12 The...
-
Kerns Book Warehouse distributes hardcover books to retail stores and extends credit terms of 2/10, n/30 to all of its customers. At the end of May, Kerns inventory consisted of books purchased for...
-
How can the relationship between Online Analytical Processing (OLAP) systems and Customer Relationship Management (CRM) bolster an organization's marketing efforts?
-
Use a dot plot to display the data. The data represent the systolic blood pressures (in millimeters of mercury) of 30 patients at a doctor's office. Organize the data using the indicated type of...
-
Daniel Fox (he/him/his) and his business partner, Dylan (them/they), run a company called Facts Pty Ltd together. Dylan is the creative brain behind Facts Pty Ltd. The business has taken out a life...
-
"What are the implications of empowerment for talent management and human resource development? Furthermore, what strategies can be implemented to empower employees through training, mentoring, and...
-
Discuss the challenges and opportunities associated with cross-cultural delegation, considering factors such as cultural values, communication styles, and power distance dimensions in shaping...
-
7. You want to buy a house and will need to borrow $280,000. The interest rate on your loan is 6.19 percent compounded monthly and the loan is for 20 years. What are your monthly mortgage payments?
-
Claire Gerber wants to buy 400 shares of Google, which is selling in the market for $534.14 a share. Rather than liquidate all her savings, she decides to borrow through her broker at 5 percent a...
-
An employee is arriving late to a meeting and she gets to her computer so fast that her boss s 2 8 2 . 3 Hz tenor voice is shifted 2 . 3 Hz higher. How fast was she moving?
-
The Internal and external chameker of the impeller of a centrifugal pump are 200mm and 400mm respectively. The qump is running at $1200 rpm. The vone ongles of the impeller at Inlet and outlet are 20...
-
Outline a general process applicable to most control situations. Using this, explain how you would develop a system to control home delivery staff at a local pizza shop.
-
Describe the bonding in the first excited state of N2 (the one closest in energy to the ground state) using the MO model. What differences do you expect in the properties of the molecule in the...
-
Consider the reaction H2(g) + Br2(g) 2HBr(g) where Ho = 2103.8 kJ. In a particular experiment, 1.00 atm of H2(g) and 1.00 atm of Br2(g) were mixed in a 1.00-L flask at 25oC and allowed to reach...
-
Elements with very large ionization energies also tend to have highly exothermic electron affinities. Explain. Which group of elements would you expect to be an exception to this statement?
-
A very simple version of the normative model described in the text involves a simple economic growth process converging to a steady-state, where values do not change over time. \({ }^{14}\) A simple...
-
Go to the library or the Internet and, for a particular year, put together a data set of profits in agriculture in different geographical units of your state or country (e.g., counties of a U.S....
-
For each of the following social choice methods, which of Arrow's axioms are violated, and why: a. the Pareto criterion b. plurality-rule voting (of several choices, the one with the most votes wins)...

Study smarter with the SolutionInn App


