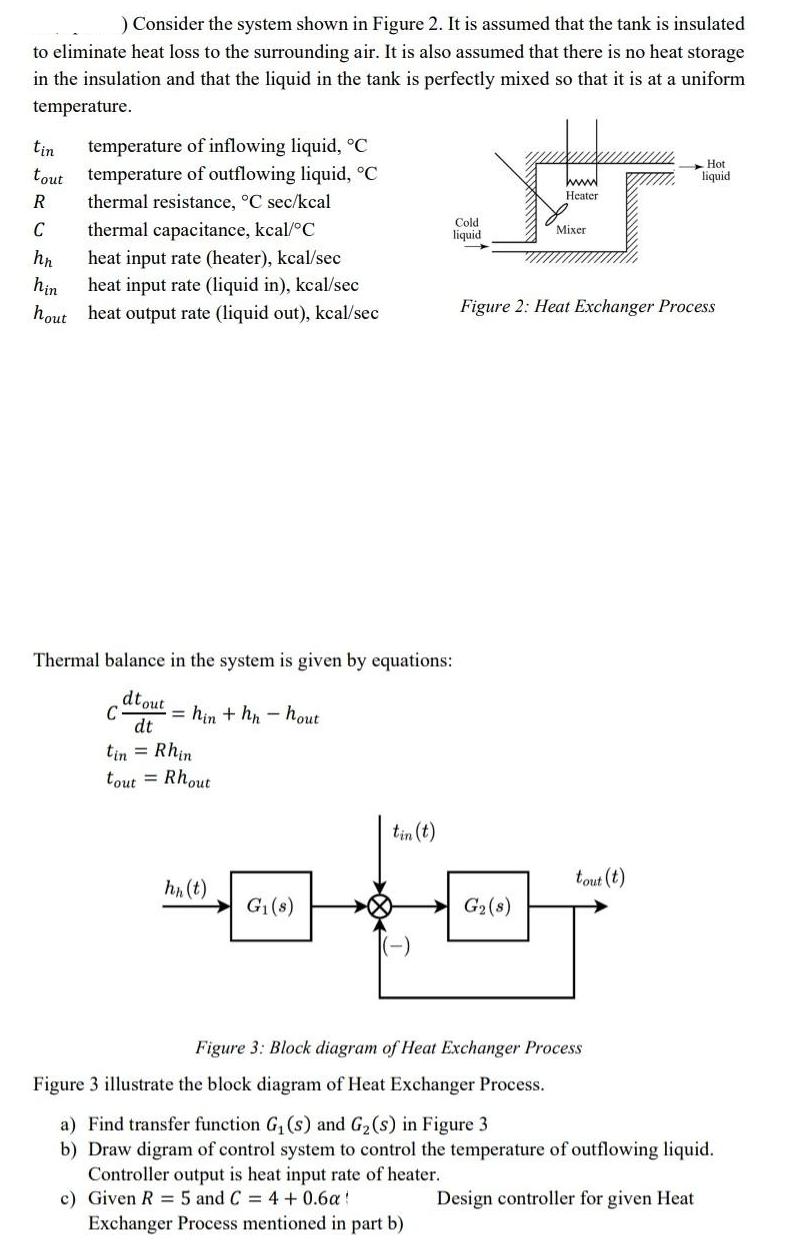
) Consider the system shown in Figure 2. It is assumed that the tank is insulated...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
) Consider the system shown in Figure 2. It is assumed that the tank is insulated to eliminate heat loss to the surrounding air. It is also assumed that there is no heat storage in the insulation and that the liquid in the tank is perfectly mixed so that it is at a uniform temperature. tin temperature of inflowing liquid, °C tout temperature of outflowing liquid, °C thermal resistance, °C sec/kcal R C thermal capacitance, kcal/°C hn hin hout heat input rate (heater), kcal/sec heat input rate (liquid in), kcal/sec heat output rate (liquid out), kcal/sec C Thermal balance in the system is given by equations: dtout dt =hin + hn-hout : Rhin tout Rhout hn (t) G₁(s) tin (t) LEC (-) Cold liquid www G₂ (8) Heater Mixer Figure 2: Heat Exchanger Process tout (t) Hot liquid Figure 3: Block diagram of Heat Exchanger Process Figure 3 illustrate the block diagram of Heat Exchanger Process. a) Find transfer function G₁ (s) and G₂ (s) in Figure 3 b) Draw digram of control system to control the temperature of outflowing liquid. Controller output is heat input rate of heater. Design controller for given Heat c) Given R = 5 and C = 4 + 0.6a Exchanger Process mentioned in part b) ) Consider the system shown in Figure 2. It is assumed that the tank is insulated to eliminate heat loss to the surrounding air. It is also assumed that there is no heat storage in the insulation and that the liquid in the tank is perfectly mixed so that it is at a uniform temperature. tin temperature of inflowing liquid, °C tout temperature of outflowing liquid, °C thermal resistance, °C sec/kcal R C thermal capacitance, kcal/°C hn hin hout heat input rate (heater), kcal/sec heat input rate (liquid in), kcal/sec heat output rate (liquid out), kcal/sec C Thermal balance in the system is given by equations: dtout dt =hin + hn-hout : Rhin tout= Rhout hn (t) G₁(s) tin (t) LEC (-) Cold liquid www G₂ (8) Heater Mixer Figure 2: Heat Exchanger Process tout (t) Hot liquid Figure 3: Block diagram of Heat Exchanger Process Figure 3 illustrate the block diagram of Heat Exchanger Process. a) Find transfer function G₁ (s) and G₂ (s) in Figure 3 b) Draw digram of control system to control the temperature of outflowing liquid. Controller output is heat input rate of heater. Design controller for given Heat c) Given R = 5 and C = 4 + 0.6a Exchanger Process mentioned in part b) ) Consider the system shown in Figure 2. It is assumed that the tank is insulated to eliminate heat loss to the surrounding air. It is also assumed that there is no heat storage in the insulation and that the liquid in the tank is perfectly mixed so that it is at a uniform temperature. tin temperature of inflowing liquid, °C tout temperature of outflowing liquid, °C thermal resistance, °C sec/kcal R C thermal capacitance, kcal/°C hn hin hout heat input rate (heater), kcal/sec heat input rate (liquid in), kcal/sec heat output rate (liquid out), kcal/sec C Thermal balance in the system is given by equations: dtout dt =hin + hn-hout : Rhin tout Rhout hn (t) G₁(s) tin (t) LEC (-) Cold liquid www G₂ (8) Heater Mixer Figure 2: Heat Exchanger Process tout (t) Hot liquid Figure 3: Block diagram of Heat Exchanger Process Figure 3 illustrate the block diagram of Heat Exchanger Process. a) Find transfer function G₁ (s) and G₂ (s) in Figure 3 b) Draw digram of control system to control the temperature of outflowing liquid. Controller output is heat input rate of heater. Design controller for given Heat c) Given R = 5 and C = 4 + 0.6a Exchanger Process mentioned in part b) ) Consider the system shown in Figure 2. It is assumed that the tank is insulated to eliminate heat loss to the surrounding air. It is also assumed that there is no heat storage in the insulation and that the liquid in the tank is perfectly mixed so that it is at a uniform temperature. tin temperature of inflowing liquid, °C tout temperature of outflowing liquid, °C thermal resistance, °C sec/kcal R C thermal capacitance, kcal/°C hn hin hout heat input rate (heater), kcal/sec heat input rate (liquid in), kcal/sec heat output rate (liquid out), kcal/sec C Thermal balance in the system is given by equations: dtout dt =hin + hn-hout : Rhin tout= Rhout hn (t) G₁(s) tin (t) LEC (-) Cold liquid www G₂ (8) Heater Mixer Figure 2: Heat Exchanger Process tout (t) Hot liquid Figure 3: Block diagram of Heat Exchanger Process Figure 3 illustrate the block diagram of Heat Exchanger Process. a) Find transfer function G₁ (s) and G₂ (s) in Figure 3 b) Draw digram of control system to control the temperature of outflowing liquid. Controller output is heat input rate of heater. Design controller for given Heat c) Given R = 5 and C = 4 + 0.6a Exchanger Process mentioned in part b)
Expert Answer:

Related Book For 

Fundamentals of Heat and Mass Transfer
ISBN: 978-0471457282
6th Edition
Authors: Incropera, Dewitt, Bergman, Lavine
Posted Date:
Students also viewed these finance questions
-
If you were coming into newton for your first day, what would you be excited for?
-
Why is it advantageous for candidates to stay in contact with prospective employers after an interview? Provide at least three reasons please with more details explanation and recent references
-
Consider the system shown in Figure 8.21, in which an RC high pass filter is followed by an ideal low pass filter having bandwidth W. Assume that the input to the system is A cos(2Ïf c t), where...
-
The accounting records for The Skate Shed, Inc., reflected the following amounts at the end of January 2018: Prepare The Skate Shed?s multistep income statement for the fiscal year ended January 31,...
-
Explain why GE might have been willing to accept subordinated debt and preferred stock for such a large part of the purchase price? Why might Comcast want to use debt and preferred equity as part of...
-
Assume Ellina earns a 10 percent after-tax rate of return, and that she owes a friend $1,200. Would she prefer to pay the friend $1,200 today or $1,750 in four years?
-
Indicate whether each of the following statements constitutes a potential advantage (A), disadvantage (D), or neither (N) of using transfer prices for support department costs. a. Requires additional...
-
Summit Corp.s stock is currently selling at $32 per share. There are 1 million shares outstanding. The firm is planning to raise $2 million to finance a new project. What are the ex-rights stock...
-
A 5.85 percent coupon bond with 14 years left to maturity can be called in nine years. The call premium is one year of coupon payments. It is offered for sale at $1,167.50. What is the yield to call...
-
Medical Testing Labs, Inc., provides routine testing services for blood banks in the Los Angeles area. Tests are supervised by skilled technicians using equipment produced by two leading competitors...
-
How does the structure of a type 3 secretion system enable proteins to be delivered directly into a eukaryotic cell?"
-
Cast Iron Grills, Incorporated, manufactures premium gas barbecue grills. The company reports inventory and cost of goods sold based on calculations from a LIFO periodic inventory system. Cast Iron's...
-
Taveras Corporation is currently operating at 5 0 % of its available manufacturing capacity. It uses a job - order costing system with a plantwide predetermined overhead rate based on machine -...
-
The following question was designed to test your knowledge of closing entries. If the account is Column "A" should be closed indicate in Column "B" whether the accounts in Column "A" should be...
-
Please prepare a Adjusting entries for the following transactions: a) Wages unpaid at the end of the month were $300. b) JS Started the period with $800 of supplies on hand. At the end of the period...
-
Transactions for the Whispering Winds Company for the month of June are presented as follows. June 1 Whispering Winds invests $4,500 cash in a small welding business of which he is the sole...
-
There are many registered in the management information systems major, some of whom will graduate this year and some of whom are about to graduate. Our student Ahmed is one of the students who has...
-
Factor and simplify, if possible. Check your result using a graphing calculator. 3 cot 2 + 6 cot + 3
-
Estimate values of the mass diffusivity DAB for binary mixtures of the following gases at 350K and 1 atm: ammonia-air and hydrogen-air.
-
A pan is used to boil water by placing it on a stove, from which heat is transferred at a fixed rate qo. There are two stages to the process. In Stage I, the water is taken from its initial (room)...
-
Consider a thermal storage system in which the phase change material (paraffin) is housed in a large container whose bottom, horizontal surface is maintained at T s = 50C by warm water delivered from...
-
Murphy Corporation has 50,000 shares of \(\$ 10\) par value common stock outstanding. The company has \(\$ 450,000\) of retained earnings. At year-end, the company declares a cash dividend of \(\$...
-
Torey Corporation was organized on April 1, with an authorization of 25,000 shares of six percent, \(\$ 50\) par value preferred stock and 200,000 shares of \(\$ 5\) par value common stock. During...
-
Ryan Corporation began business on March 1, 2016. At that time, it issued 20,000 shares of \(\$ 60\) par value, seven percent cumulative preferred stock and 100,000 shares of \(\$ 5\) par value...

Study smarter with the SolutionInn App


