Copyright At the beginning of the nineteenth century, chemists were searching for numerical relation- ships among...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
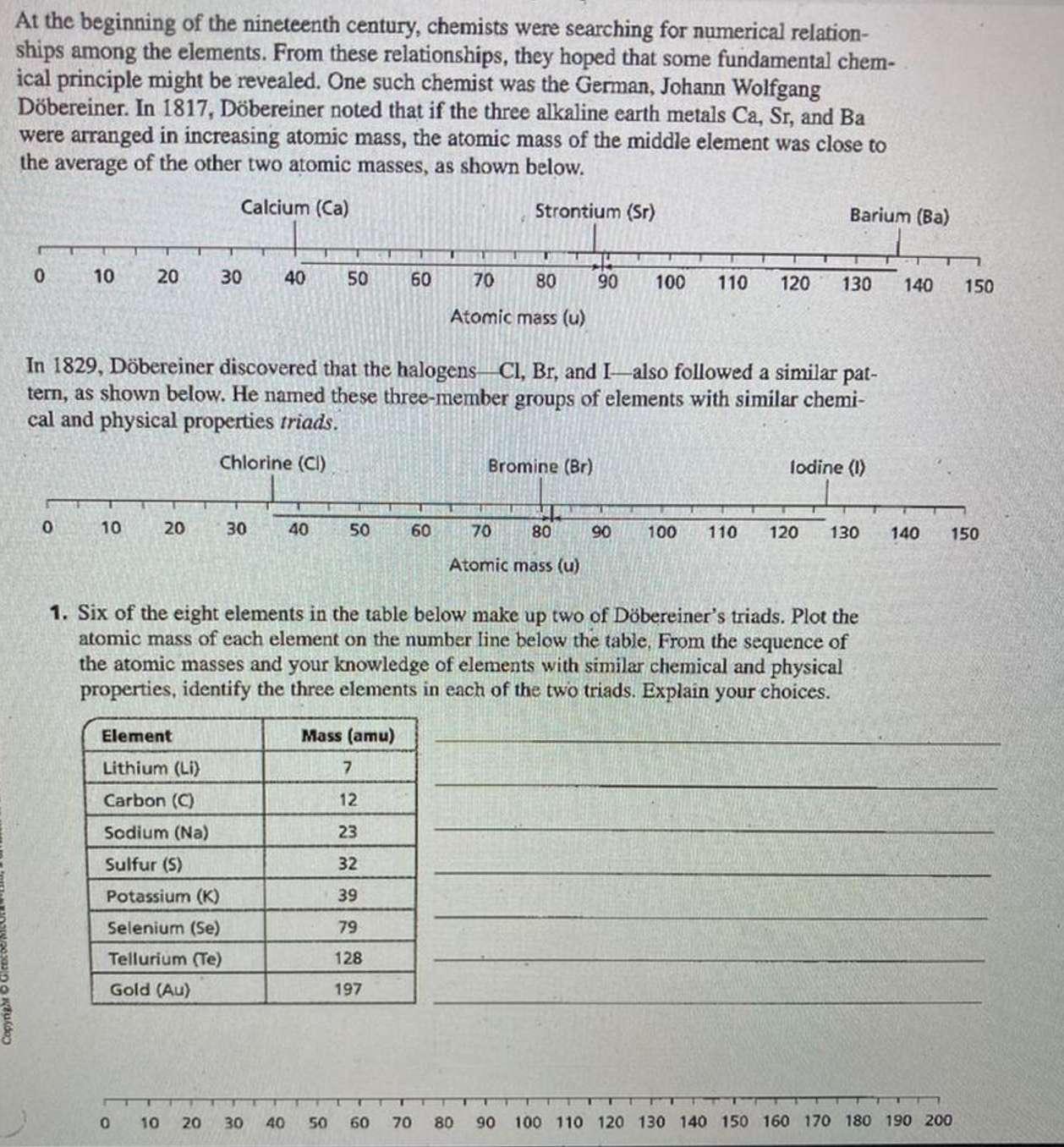
Copyright At the beginning of the nineteenth century, chemists were searching for numerical relation- ships among the elements. From these relationships, they hoped that some fundamental chem- ical principle might be revealed. One such chemist was the German, Johann Wolfgang Döbereiner. In 1817, Döbereiner noted that if the three alkaline earth metals Ca, Sr, and Ba were arranged in increasing atomic mass, the atomic mass of the middle element was close to the average of the other two atomic masses, as shown below. Calcium (Ca) Strontium (Sr) 0 10 0 20 10 20 30 Element Lithium (Li) Carbon (C) Sodium (Na) Sulfur (5) 40 30 Potassium (K) Selenium (Se) Tellurium (Te) Gold (Au) 50 In 1829, Döbereiner discovered that the halogens Cl, Br, and I also followed a similar pat- tern, as shown below. He named these three-member groups of elements with similar chemi- cal and physical properties triads. Chlorine (Cl) 40 0 10 20 30 40 50 60 Mass (amu) 7 12 23 32 39 79 128 197 60 T U 70 80 Atomic mass (u) 50 60 70 U Bromine (Br) 70 80 Atomic mass (u) 80 90 100 110 120 1. Six of the eight elements in the table below make up two of Döbereiner's triads. Plot the atomic mass of each element on the number line below the table, From the sequence of the atomic masses and your knowledge of elements with similar chemical and physical properties, identify the three elements in each of the two triads. Explain your choices. Barium (Ba) 90 130 lodine (1) 100 110 120 130 140 150 140 150 T 90 100 110 120 130 140 150 160 170 180 190 200 Copyright At the beginning of the nineteenth century, chemists were searching for numerical relation- ships among the elements. From these relationships, they hoped that some fundamental chem- ical principle might be revealed. One such chemist was the German, Johann Wolfgang Döbereiner. In 1817, Döbereiner noted that if the three alkaline earth metals Ca, Sr, and Ba were arranged in increasing atomic mass, the atomic mass of the middle element was close to the average of the other two atomic masses, as shown below. Calcium (Ca) Strontium (Sr) 0 10 0 20 10 20 30 Element Lithium (Li) Carbon (C) Sodium (Na) Sulfur (5) 40 30 Potassium (K) Selenium (Se) Tellurium (Te) Gold (Au) 50 In 1829, Döbereiner discovered that the halogens Cl, Br, and I also followed a similar pat- tern, as shown below. He named these three-member groups of elements with similar chemi- cal and physical properties triads. Chlorine (Cl) 40 0 10 20 30 40 50 60 Mass (amu) 7 12 23 32 39 79 128 197 60 T U 70 80 Atomic mass (u) 50 60 70 U Bromine (Br) 70 80 Atomic mass (u) 80 90 100 110 120 1. Six of the eight elements in the table below make up two of Döbereiner's triads. Plot the atomic mass of each element on the number line below the table, From the sequence of the atomic masses and your knowledge of elements with similar chemical and physical properties, identify the three elements in each of the two triads. Explain your choices. Barium (Ba) 90 130 lodine (1) 100 110 120 130 140 150 140 150 T 90 100 110 120 130 140 150 160 170 180 190 200
Expert Answer:
Answer rating: 100% (QA)
Dobereiners triads represent a classification system for elements that significantly contributed to ... View the full answer

Related Book For 

Posted Date:
Students also viewed these mathematics questions
-
It's October 10, 2021, and you, CPA, work as an audit senior at Chow and Co. LLP. One of the firm's long-time clients, Tyler Wallace, owns a group of companies, and recently added a new addition to...
-
Write a literature review for your study. See below for an example of a literature review. Your literature review should provide both analysis and synthesis of previous studies as related to the...
-
Capital Inc. has prepared the operating budget for the first quarter of 2015. They forecast sales of $50,000 in January, $60,000 in February, and $70,000 in March. Variable and fixed expenses are as...
-
Soon after December 31, 2013, the auditor requested a depreciation schedule for trucks of Jarrett Trucking Company, showing the additions, retirements, depreciation, and other data affecting the...
-
calculate the the total laibilities and stockholders equity B C 0 1 2 MARTA COMMUNICATIONS, INC. Statement of Retained Earnings For the Month Ended March 31, 20X1 3 Retained Earnings, March 1, 20X1...
-
Locate questions that can be used during a debtors examination once a judgment is obtained. Try to find questions specific to California, as many states have limits on the types of questions that can...
-
Listed are the equity sections of balance sheets for years 2011 and 2012 as reported by Mountain Air Ski Resorts, Inc. The overall value of stockholders equity has risen from $2,000,000 to...
-
Considering the competitive and payor landscape in Charlotte, NC, propose a growth strategy and provide a breakeven analysis for this new market. Assume that you are the Center Director and tailor...
-
The balance sheet provides a snapshot of the financial condition of a company. Investors and analysts use the information given on the balance sheet and other financial statements to make several...
-
Let A = 8x + 5 B = -3x + 3y C = 1x+-9 Complete each vector sum. A+B+C= A-B+C= 4 + + A+B-C= A A-B-C= + +
-
The purpose of this assignment is to enhance learners' ability to discuss the widespread use of information technology in the banking industry. REQUIREMENT: The advancement of technology within the...
-
Describe the overall process for effectively using technology within an organization. Consider such aspects as strategic alignment, planning, centralized systems, data governance, cybersecurity, the...
-
Garfield Corporation expects to sell 1,400 units of its pet beds in March and 700 units in April. Each unit sells for $140. Garfield's ending inventory policy is 30 percent of the following month's...
-
Modern physics rocket is moving from the Moon to Mars (a distance of about 8.64x10^7 km) at 0.93c. What distance would an observer on the rocket measure, and what distance would an observer on the...
-
Q1. What types of searches are performed in prisons and why are they important? Q2. After viewing the Prison Searches Training Video under lesson reviews, what did you learn and why was this...
-
3 7 Use Case Diagram 2- extend, include, generalize, systems Purpose: describe relationships between use cases and subtypes of use cases or actors. DO NOT USE to show progress from one task to...
-
Outline a general process applicable to most control situations. Using this, explain how you would develop a system to control home delivery staff at a local pizza shop.
-
The chief financial officer of a firm presented the CEO with a set of financial statements showing $2,234 million in after-tax operating income. This number yielded a return on beginning-of-period...
-
Xerox Corporation is a long-established company whose very name has been lent to the process of copying documents. The firm develops copying technology through an extensive research program and...
-
Comment on the following statement. The challenge in measuring the required return for investing is to measure the size of the risk premium over the risk-free rate, but the capital asset pricing...
-
Consider two components of U.S. monthly industrial production index from December 1963 to December 2012. The two components are (a) nondurable consumer goods and (b) materials. The data are in...
-
Consider the bivariate VMA(1) model \[ \boldsymbol{z}_{t}=\boldsymbol{a}_{t}-\left[\begin{array}{rr} -0.3 & 0.2 \\1.1 & 0.6 \end{array} ight] \boldsymbol{a}_{t-1} \] where \(a_{t}\) is a Gaussian...
-
Consider two components of U.S. monthly industrial production index from December 1963 to December 2012. The two components are (a) business equipments and (b) materials. The data are in columns 5...

Study smarter with the SolutionInn App


