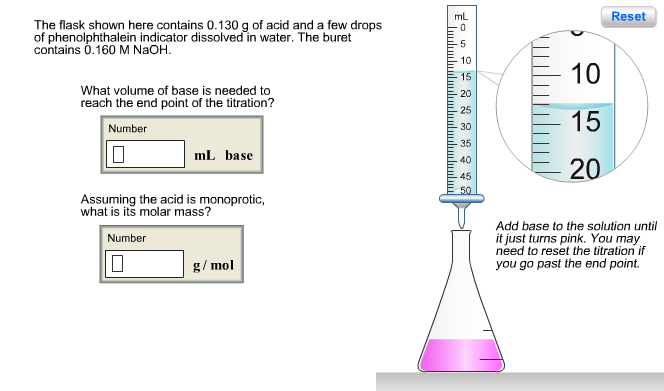
The flask shown here contains 0.130 g of acid and a few drops of phenolphthalein indicator...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
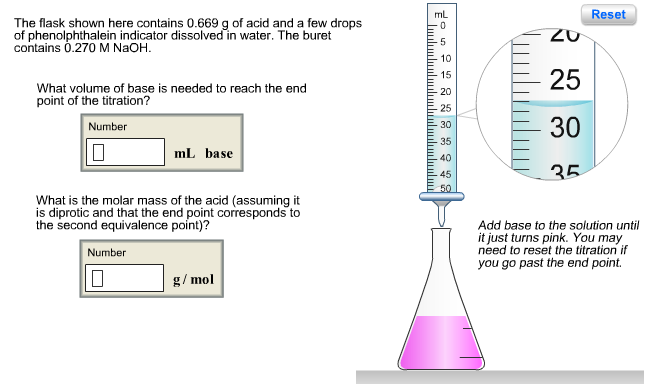
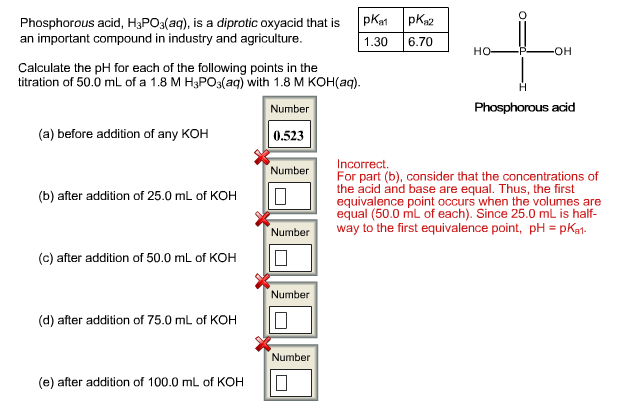
The flask shown here contains 0.130 g of acid and a few drops of phenolphthalein indicator dissolved in water. The buret contains 0.160 M NaOH. What volume of base is needed to reach the end point of the titration? Number 0 mL base Assuming the acid is monoprotic, what is its molar mass? Number 0 g/mol mL ساسا 5 10 15 -20 25 30 35 40 45 50 10 15 20 Reset Add base to the solution until it just turns pink. You may need to reset the titration if you go past the end point. The flask shown here contains 0.669 g of acid and a few drops of phenolphthalein indicator dissolved in water. The buret contains 0.270 M NaOH. What volume of base is needed to reach the end point of the titration? Number 10 mL base What is the molar mass of the acid (assuming it is diprotic and that the end point corresponds to the second equivalence point)? Number 0 g/mol mL 10 15 -20 -25 30 -35 40 -45 50 ZU 25 30 35 Reset Add base to the solution until it just turns pink. You may need to reset the titration if you go past the end point. Phosphorous acid, H3PO3(aq), is a diprotic oxyacid that is an important compound in industry and agriculture. Calculate the pH for each of the following points in the titration of 50.0 mL of a 1.8 M H3PO3(aq) with 1.8 M KOH(aq). Number 0.523 (a) before addition of any KOH (b) after addition of 25.0 mL of KOH (c) after addition of 50.0 mL of KOH (d) after addition of 75.0 mL of KOH (e) after addition of 100.0 mL of KOH Number 10 Number 10 Number Number pka1 pka2 1.30 6.70 HỌ -OH Phosphorous acid Incorrect. For part (b), consider that the concentrations of the acid and base are equal. Thus, the first equivalence point occurs when the volumes are equal (50.0 mL of each). Since 25.0 mL is half- way to the first equivalence point, pH = pka1- The flask shown here contains 0.130 g of acid and a few drops of phenolphthalein indicator dissolved in water. The buret contains 0.160 M NaOH. What volume of base is needed to reach the end point of the titration? Number 0 mL base Assuming the acid is monoprotic, what is its molar mass? Number 0 g/mol mL ساسا 5 10 15 -20 25 30 35 40 45 50 10 15 20 Reset Add base to the solution until it just turns pink. You may need to reset the titration if you go past the end point. The flask shown here contains 0.669 g of acid and a few drops of phenolphthalein indicator dissolved in water. The buret contains 0.270 M NaOH. What volume of base is needed to reach the end point of the titration? Number 10 mL base What is the molar mass of the acid (assuming it is diprotic and that the end point corresponds to the second equivalence point)? Number 0 g/mol mL 10 15 -20 -25 30 -35 40 -45 50 ZU 25 30 35 Reset Add base to the solution until it just turns pink. You may need to reset the titration if you go past the end point. Phosphorous acid, H3PO3(aq), is a diprotic oxyacid that is an important compound in industry and agriculture. Calculate the pH for each of the following points in the titration of 50.0 mL of a 1.8 M H3PO3(aq) with 1.8 M KOH(aq). Number 0.523 (a) before addition of any KOH (b) after addition of 25.0 mL of KOH (c) after addition of 50.0 mL of KOH (d) after addition of 75.0 mL of KOH (e) after addition of 100.0 mL of KOH Number 10 Number 10 Number Number pka1 pka2 1.30 6.70 HỌ -OH Phosphorous acid Incorrect. For part (b), consider that the concentrations of the acid and base are equal. Thus, the first equivalence point occurs when the volumes are equal (50.0 mL of each). Since 25.0 mL is half- way to the first equivalence point, pH = pka1-
Expert Answer:

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
Please answer these questions in SQL statements. The logical model is provided, please answer with SQL statements based on this logical model. Q1 List all endorsements held by pilots where the...
-
Please answer these questions with Computer Sciencebelow ? 1- a pharmaceutical company is using blockchain to manage theirsupply chain. some of their drugs must be stored at a lowertemperature...
-
Could you please answer the "Reflection" part link here: https://drive.google.com/file/d/112_IS9IvDCcm9opRGiefB8T9R9-rbOqb/view?usp=sharing
-
Sandra, a sole proprietor, is an interior designer and has thefollowing account balances on December 31, 20X0: Cash $2,500,Inventory $9,000, Equipment $25,000, Accumulated depreciation$5,000, Liabi 2...
-
For each of the following accounts, indicate whether we use a debit or a credit to increase the balance of the account. a. Cash. b. Salaries Payable. c. Utilities Expense. d. Service Revenue.
-
Pick a company that you are familiar with and describe its operations strategy and how it relates to winning customers. Describe specific activities used by the company that support the strategy.
-
10.A company contracted with a marketing firm to construct software and create a business website. A quote was requested and accepted. Sometime later, the business asked for updates and revisions but...
-
Purchases Recorded Gross and Net some of the transactions of Torres Company during August are listed below. Torres uses the periodic inventory method. August 10 Purchased merchandise on account,...
-
While many parties were interested in acquiring MCI, the major players included Verizon and Qwest. U.S.-based Qwest is an integrated communications company that provides data, multimedia, and...
-
1. Mr. Albert has a piece of property he has used in his business for 25 years. He is moving his business across the country and needs to get rid of the real estate he currently owns, as he won't be...
-
Susan Company commenced business with the investment of $1,372,000 in its operating assets on 1 January 2021 to produce a single product a handmade barbecue grill. The company planned to produce and...
-
5,000 new cases of E.Coli (a bacteria) infection happened in a city with a population of 500,000 over a 5-year period. What is the crude incidence rate?
-
Firearms have been in the news again as calls for increased gun control measures resurface. But the gun industry has been going through it own ups and downs just last week when firearms giant...
-
What do you think about all the earning management done at GE? Do you think users would eventually respond to the fact that GE has been a "relatively aggressive practitioner of earnings management"?
-
11. Evaluate each of the following expressions: a. 4*3/6-4+ Math.pow(7,2) b. (3+4)*7-3 c. 9*2/4+5%3+3 d. 55==55
-
John is a software engineer and contributes 10% of his salary to his 401(k). His employer makes matching contribution of 3% of salary. Last year, John earned $50,000, but he received a raise and will...
-
Find the market equilibrium point for the following demand and supply functions. Demand: 2p = - q + 56 Supply: 3p - q = 34
-
Although HCl and H2SO4 have very different properties as pure substances, their aqueous solutions possess many common properties. List some general properties of these solutions, and explain their...
-
The hypochlorite ion, ClO-, acts as a weak base. (a) Is ClO- a stronger or weaker base than hydroxylamine? (b) When ClO- acts as a base, which atom, Cl or O, acts as the proton acceptor? (c) Can you...
-
Among the elementary subatomic particles of physics is the muon, which decays within a few nanoseconds after formation. The muon has a rest mass 206.8 times that of an electron. Calculate the de...
-
One end of a wire is tied to a point A on the ceiling and the other end is tied to a point on a ring of radius \(R\) and negligible mass. The ring therefore hangs from the wire in a vertical plane...
-
A particle moves in a cylindrically symmetric potential \(U(ho, z)\). Use cylindrical coordinates \(ho, \varphi\), and \(z\) to parameterize the space. (a) Write the Lagrangian for an unconstrained...
-
A particle of mass \(m\) slides inside a smooth paraboloid of revolution whose axis of symmetry \(z\) is vertical. The surface is defined by the equation \(z=\alpha ho^{2}\), where \(z\) and \(ho\)...

Study smarter with the SolutionInn App


