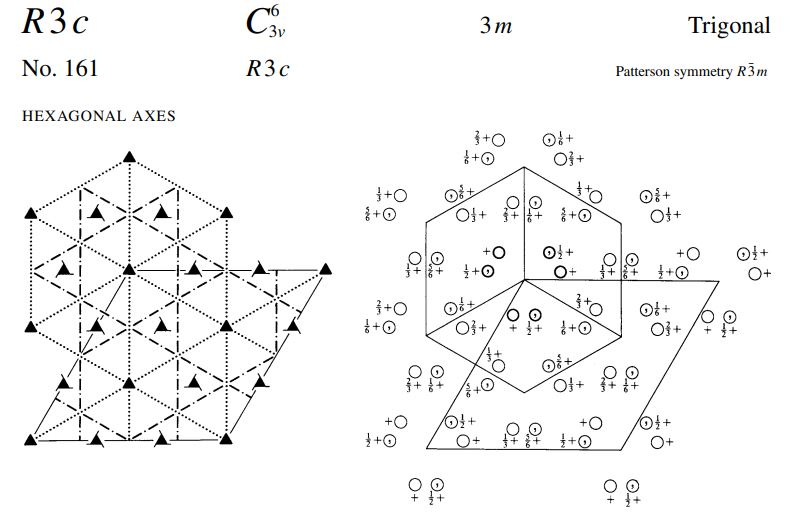
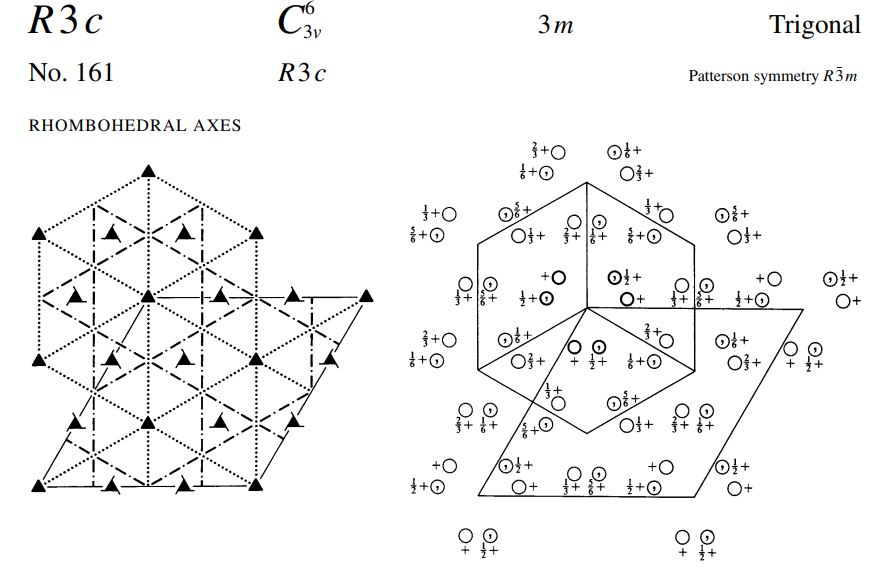
Crystals of Sodium Nitrate Na*(NO3) are trigonal with space group R-3c. When referred to a non...
Fantastic news! We've Found the answer you've been seeking!
Question:
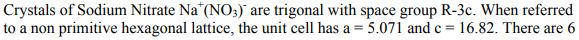
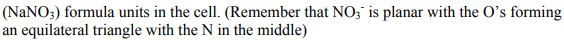





Transcribed Image Text:
Crystals of Sodium Nitrate Na*(NO3) are trigonal with space group R-3c. When referred to a non primitive hexagonal lattice, the unit cell has a = 5.071 and c= 16.82. There are 6 (NaNO3) formula units in the cell. (Remember that NO3 is planar with the O's forming an equilateral triangle with the N in the middle) a) Use the International Tables to determine the crystal structure as completely as possible. b) What point symmetry is allowed for the NO, anion? c) What atomic positional parameters are not determined by the space group symmetry? R3c No. 161 HEXAGONAL AXES R3c O+f {+O O+ { O + f O+ O+F 3m 0+ { O+ Of+ 0}+ + ²/1/ + 10/ O+ +10 0+ +10 O+ O}+ Off +0 O+ toi 3+0 +O Of+ O+ O+f Patterson symmetry R3m Of+ Of+ O+F +10 Trigonal 0}+ Ⓒ++ O+ O+ Origin on 3 c Asymmetric unit Vertices 0≤x≤ 0≤y≤t: 0≤z≤ x ≤ (1+y)/2; y ≤min(1-x, (1+x)/2) 0,0,0,0,0,0.0 0,0,0,* * 0 Symmetry operations For (0,0,0) + set (1) 1 (4) c x,x,z For (+)+ set (1) *(4,4) (4) g() x+xz For (..)+ set (1) +(,,) (4) g() x+xz (2) 3+ 0,0,z (5) c x, 2x, z 0.4.0 (2) 3 (0,0,4) 4.4.2 (5) g() x+1,2x, z (2) 3 (0,0,4) 0,,z (5) g(..) x,2x, z (3) 3- 0,0,z (6) c 2x,x,z (3) 3 (0,0,1),0,z (6) g(,) 2x, x,z (3) 3 (0,0,1) 1.1.2 (6) g() 2x-1.x,z Positions Multiplicity. Wyckoff letter, Site symmetry 18 bl 6 a 3. 0,0,z Symmetry of special projections Along [001] p31m a = (2a+b) b = (-a+b) Origin at 0, 0, z (1) x,y,z (4) y,x,z++ IIa IIb none (0,0,0)+ (..)+ (..)+ [2] R31 (R3, 146) [3] R1c (Cc, 9) [3] RIc (Cc, 9) [3] R1c (Cc, 9) [3] P3c1 (158) Coordinates I II Maximal non-isomorphic subgroups I (1; 2; 3) + (1; 4)+ (1; 5)+ (1; 6)+ 1; 2; 3; 4; 5; 6 (2) y,x-y,z (5) x+y,y,z + + 0,0,z++ Minimal non-isomorphic supergroups (3) x+y, x, z (6) x,x-y,z++ Along [100] pl a' = (2a + 4b+c) Origin at x, 0,0 Maximal isomorphic subgroups of lowest index IIc [4] R3 c (a = -2a, b = -2b) (161); [5] R3c (a' = -a, b' = -b, c = 5c) (161) b=(-a-2b+c) Reflection conditions [2] R3c (167); [4] P43n (218); [4] F43c (219); [4] 143d (220) [2] R3 m (a = a, b = -b, c = c) (160); [3] P31c (a' = (2a+b), b = (-a+b), c = c) (159) General: hkil : -h+k+1=3n hki0 -h+k= 3n hh2hl: 1 = 3n hhol 0001 1= 6n hh00: h=3n Special: as above, plus hkil : 1 = 2n h+1=3n, 1 = 2n Along [210] plgl a = b b = c Origin at x, x,0 R3c No. 161 RHOMBOHEDRAL AXES 3v R3 c By +0 +O }+O +O +O +0 +O ++O O 3m 3+0 +O O+ Of+ +0 0}+ +0 Ot+ O+ Ⓒto +O 0}+ O+ +O Of+ + /O+ 00+ ++ + + +0 O+ Patterson symmetry R3m Of+ Of+ Ot+ Trigonal +0 +O 0}+ +O /Ⓒ++ O+ O+ 0+. Origin on 3 c Asymmetric unit Vertices 0≤x≤1; 0≤y≤1; 0≤z≤1; y≤x; z≤y 0,0,0 1,0,0 1,1,0 1,1,1 Symmetry operations (1) 1 (4) n() x,y,x Heights refer to hexagonal axes (2) 3 x, x,x (5) n() x,x, Z (3) 3 x, x,x (6) n() x,y,y Generators selected (1); t(1,0,0); t(0,1,0); t(0,0,1); (2); (4) Positions Multiplicity, Wyckoff letter, Site symmetry 6 b1 (1) x, y, z (4) z+y+x+ 2 a 3. X, X, X Symmetry of special projections Along [111] p31m a = (2a-b-c) b = (-a+2b-c) Origin at x, x,x Coordinates Maximal non-isomorphic subgroups I 1; 2; 3 1; 4 1; 5 1; 6 [2] R31 (R3, 146) [[3] R1c (Cc, 9) [3] R1c (Cc, 9) [[3] R1c (Cc, 9) I x+x+x+! II (2) z,x,y (5) y+x+z+! Ila none IIb [3] P3c1 (aa-b₁b-b-c,c=a+b+c) (158) Minimal non-isomorphic supergroups Along [110] pl a = + (a+b-2c) Origin at x.*,0 (3) y, z, x (6) x+z+y+ b = c Reflection conditions [2] R3c (167); [4] P43n (218); [4] F43c (219); [4] 143d (220) [2] R3m (a = (-a+b+c), b = (a-b+c), c = (a+b-c)) (160); [3] P31c (a=(2a-b-c), b=(-a+2b-c), c = (a+b+c)) (159) General: hhl: 1 = 2n hhh: h= 2n Maximal isomorphic subgroups of lowest index IIc [4] R3c (a' = -a+b+c,b=a-b+c,c=a+b-c) (161); [5] R3 c (a=a+2b+2c, b = 2a+b+2c, c = 2a+2b+c) (161) Special: as above, plus hkl h+k+1= 2n Along [211] p1g1 a = -(b-c) Origin at 2x, .*,* b = + (a+b+c) Crystals of Sodium Nitrate Na*(NO3) are trigonal with space group R-3c. When referred to a non primitive hexagonal lattice, the unit cell has a = 5.071 and c= 16.82. There are 6 (NaNO3) formula units in the cell. (Remember that NO3 is planar with the O's forming an equilateral triangle with the N in the middle) a) Use the International Tables to determine the crystal structure as completely as possible. b) What point symmetry is allowed for the NO, anion? c) What atomic positional parameters are not determined by the space group symmetry? R3c No. 161 HEXAGONAL AXES R3c O+f {+O O+ { O + f O+ O+F 3m 0+ { O+ Of+ 0}+ + ²/1/ + 10/ O+ +10 0+ +10 O+ O}+ Off +0 O+ toi 3+0 +O Of+ O+ O+f Patterson symmetry R3m Of+ Of+ O+F +10 Trigonal 0}+ Ⓒ++ O+ O+ Origin on 3 c Asymmetric unit Vertices 0≤x≤ 0≤y≤t: 0≤z≤ x ≤ (1+y)/2; y ≤min(1-x, (1+x)/2) 0,0,0,0,0,0.0 0,0,0,* * 0 Symmetry operations For (0,0,0) + set (1) 1 (4) c x,x,z For (+)+ set (1) *(4,4) (4) g() x+xz For (..)+ set (1) +(,,) (4) g() x+xz (2) 3+ 0,0,z (5) c x, 2x, z 0.4.0 (2) 3 (0,0,4) 4.4.2 (5) g() x+1,2x, z (2) 3 (0,0,4) 0,,z (5) g(..) x,2x, z (3) 3- 0,0,z (6) c 2x,x,z (3) 3 (0,0,1),0,z (6) g(,) 2x, x,z (3) 3 (0,0,1) 1.1.2 (6) g() 2x-1.x,z Positions Multiplicity. Wyckoff letter, Site symmetry 18 bl 6 a 3. 0,0,z Symmetry of special projections Along [001] p31m a = (2a+b) b = (-a+b) Origin at 0, 0, z (1) x,y,z (4) y,x,z++ IIa IIb none (0,0,0)+ (..)+ (..)+ [2] R31 (R3, 146) [3] R1c (Cc, 9) [3] RIc (Cc, 9) [3] R1c (Cc, 9) [3] P3c1 (158) Coordinates I II Maximal non-isomorphic subgroups I (1; 2; 3) + (1; 4)+ (1; 5)+ (1; 6)+ 1; 2; 3; 4; 5; 6 (2) y,x-y,z (5) x+y,y,z + + 0,0,z++ Minimal non-isomorphic supergroups (3) x+y, x, z (6) x,x-y,z++ Along [100] pl a' = (2a + 4b+c) Origin at x, 0,0 Maximal isomorphic subgroups of lowest index IIc [4] R3 c (a = -2a, b = -2b) (161); [5] R3c (a' = -a, b' = -b, c = 5c) (161) b=(-a-2b+c) Reflection conditions [2] R3c (167); [4] P43n (218); [4] F43c (219); [4] 143d (220) [2] R3 m (a = a, b = -b, c = c) (160); [3] P31c (a' = (2a+b), b = (-a+b), c = c) (159) General: hkil : -h+k+1=3n hki0 -h+k= 3n hh2hl: 1 = 3n hhol 0001 1= 6n hh00: h=3n Special: as above, plus hkil : 1 = 2n h+1=3n, 1 = 2n Along [210] plgl a = b b = c Origin at x, x,0 R3c No. 161 RHOMBOHEDRAL AXES 3v R3 c By +0 +O }+O +O +O +0 +O ++O O 3m 3+0 +O O+ Of+ +0 0}+ +0 Ot+ O+ Ⓒto +O 0}+ O+ +O Of+ + /O+ 00+ ++ + + +0 O+ Patterson symmetry R3m Of+ Of+ Ot+ Trigonal +0 +O 0}+ +O /Ⓒ++ O+ O+ 0+. Origin on 3 c Asymmetric unit Vertices 0≤x≤1; 0≤y≤1; 0≤z≤1; y≤x; z≤y 0,0,0 1,0,0 1,1,0 1,1,1 Symmetry operations (1) 1 (4) n() x,y,x Heights refer to hexagonal axes (2) 3 x, x,x (5) n() x,x, Z (3) 3 x, x,x (6) n() x,y,y Generators selected (1); t(1,0,0); t(0,1,0); t(0,0,1); (2); (4) Positions Multiplicity, Wyckoff letter, Site symmetry 6 b1 (1) x, y, z (4) z+y+x+ 2 a 3. X, X, X Symmetry of special projections Along [111] p31m a = (2a-b-c) b = (-a+2b-c) Origin at x, x,x Coordinates Maximal non-isomorphic subgroups I 1; 2; 3 1; 4 1; 5 1; 6 [2] R31 (R3, 146) [[3] R1c (Cc, 9) [3] R1c (Cc, 9) [[3] R1c (Cc, 9) I x+x+x+! II (2) z,x,y (5) y+x+z+! Ila none IIb [3] P3c1 (aa-b₁b-b-c,c=a+b+c) (158) Minimal non-isomorphic supergroups Along [110] pl a = + (a+b-2c) Origin at x.*,0 (3) y, z, x (6) x+z+y+ b = c Reflection conditions [2] R3c (167); [4] P43n (218); [4] F43c (219); [4] 143d (220) [2] R3m (a = (-a+b+c), b = (a-b+c), c = (a+b-c)) (160); [3] P31c (a=(2a-b-c), b=(-a+2b-c), c = (a+b+c)) (159) General: hhl: 1 = 2n hhh: h= 2n Maximal isomorphic subgroups of lowest index IIc [4] R3c (a' = -a+b+c,b=a-b+c,c=a+b-c) (161); [5] R3 c (a=a+2b+2c, b = 2a+b+2c, c = 2a+2b+c) (161) Special: as above, plus hkl h+k+1= 2n Along [211] p1g1 a = -(b-c) Origin at 2x, .*,* b = + (a+b+c)
Expert Answer:
Answer rating: 100% (QA)
The skin friction coefficient Cf for a laminar boundary ... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemical engineering questions
-
The unit cell for Cr2O3 has hexagonal symmetry with lattice parameters a = 0.4961 nm and c = 1.360 nm. If the density of this material is 5.22 g/cm3, calculate its atomic packing factor. For this...
-
An equilateral triangle has a point charge + q at each of the three vertices (A, B, C). Another point charge Q is placed at D, the midpoint of the side BC. Solve for Q if the total electric force on...
-
In a beehive, each cell is a regular hexagonal prism, open at one end with a trihedral angle at the other end. It is believed that bees form their cells in such a way as to minimize the surface area...
-
Consider X[k] be the N-point DFT of an N-point sequence x[n]. (20 %) x [n] = {1, -2, 1, 3} x [n] = {0, 2, -1,0,0,4} (a) Determine the linear convolution x [n] * x [n] (5%) (b) Determine the...
-
Before Parriss lawsuit against Railroad had settled, he left town and closed out his account with Bank. Railroad then issued a check to him which somehow came to be in Eddys possession. Eddy indorsed...
-
Example 2.6 on page 51 contains a two-way table showing preferences for an award (Academy Award, Nobel Prize, Olympic gold medal) by gender for the students sampled in StudentSurvey. The data are...
-
What is the purpose of a declaration?
-
In his 1865 science fiction novel From the Earth to the Moon, Jules Verne described how three astronauts are shot to the Moon by means of a huge gun. According to Verne, the aluminum capsule...
-
3. (24 Points) For the following problem, no justification is required. Let f(x) be a polynomial whose second derivative f"(x) is graphed below: "(x) A B D Answer the following question about the...
-
Frankel Ltd., a British merchandising company, is the exclusive distributor of a product that is gaining rapid market acceptance. The companys revenues and expenses (in British pounds) for the last...
-
Assume the following data for AAA Corporation: Market price/share = 19 Book value/share = 14 Number of shares outstanding = 104785782 Market price/bond = 926 Face value/bond = 1000 Number of bonds...
-
Given the long term capital requirements, propose a capital structure of Andreas Pelekanos (Contemporary Epiplo Design) label, advising Petra Marble on the number of shares you propose to issue. You...
-
An object has four forces acting on it, which balance in pairs. How will the object behave? A 3.0 kg object moves on a horizontal plane. A 9.0 N force acts on the object to the right and a 4.0 N...
-
Question 9 Which trees are full and complete? U U C E G E H B H 1 pts
-
On December 1, Jasmin Ernst organized Ernst Consulting. On December 3, the owner contributed $85,050 in assets in exchange for its common stock to launch the business. On December 31, the company's...
-
Train Accident. A train carrying freight is slowly moving down the tracks at a constant speed of 14.0 m/s. A passenger train ends up on the same tracks behind the freight train, travelling 35.0 m/s...
-
"Convergence"in economic growth refers to the idea that a. less developed economies experience higher growth rates,thus catching up with more developed countries b. labor and capital tend to come...
-
In Problem 8.43, determine the smallest value of for which the rod will not fall out of the pipe. IA -3 in.-
-
Identify the Lewis acid and Lewis base among the reactants in each of the following reactions: (lq denotes liquid ammonia as solvent)
-
The specific heat of octane, C8H18(l), is 2.22 J g-K. (a) How many J of heat are needed to raise the temperature of 80.0 g of octane from 10.0 C to 25.0 C? (b) Which will require more heat,...
-
Acetonitrile (CH3CN) is a polar organic solvent that dissolves a wide range of solutes, including many salts. The density of a 1.80 M LiBr solution in acetonitrile is 0.826 g/cm3. Calculate the...
-
Consider the general orientation of three strain gages at a point as shown. Write a computer program that can be used to determine the principal in-plane strains and the maximum in-plane shear strain...
-
For the case of plane stress, show that Hooke's law can be written as \[\sigma_{x}=\frac{E}{\left(1-u^{2} ight)}\left(\epsilon_{x}+u \epsilon_{y} ight), \quad \sigma_{y}=\frac{E}{\left(1-u^{2}...
-
A bar of copper alloy is loaded in a tension machine and it is determined that \(\epsilon_{x}=940\left(10^{-6} ight)\) and \(\sigma_{x}=14 \mathrm{ksi}, \sigma_{y}=0\), \(\sigma_{z}=0\). Determine...

Study smarter with the SolutionInn App


