Using the volume of base at the equivalence point, its molarity, and the fact that you...
Fantastic news! We've Found the answer you've been seeking!
Question:



Transcribed Image Text:
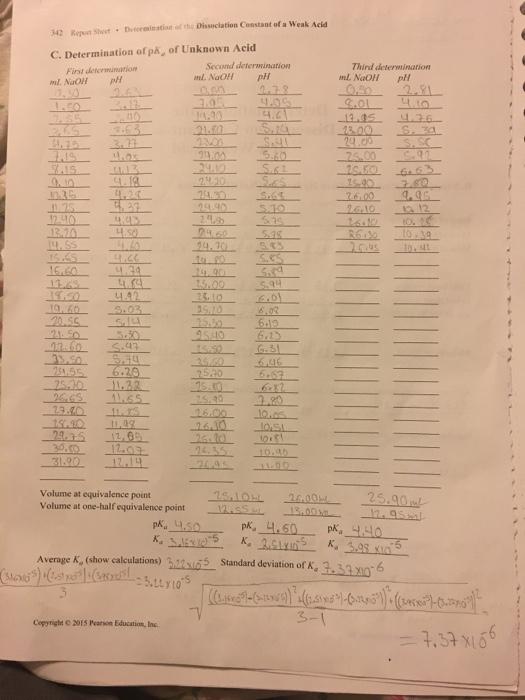
Using the volume of base at the equivalence point, its molarity, and the fact that you used 25.00 mL of acid, calculate the concentration of the unknown acid and record this on the report sheet. D. Concentration of Unknown Acid Volume of unknown acid Average molarity of NaOH from above mL of NaOH at equivalence point Molarity of unknown acid Average molarity (show calculations) Trial 1 Trial 2 Standard deviation Laboratory Experim Trial 3 B. Standardization of Sodium Hydroxide (NaOH) Solution Trial 1 Trial 2 Mass of bottle + KHP Mass of bottle Mass of KHP used 4139 14.09g Final buret reading 19.55 Initial buret reading 22,10 mL of NaOH used 13.30 17.55 Molarity of NaOH 117 M 114 M Average molarity (show calculations and standard deviation) 0.115M 117 +114 1.115 3 =115 M. Standard deviation (see Experiment 8) 0.0283M (147-15)²+(114-115)²+(115-115) ² -0.0283 M 3-1 =.117 M •114 M . =.115 M 43g Hip ARED met NOCH 204. 22gup 1 Cup 409g KHP mal x Imol NaOH Imor KHP 204.22g 419gxHPx mal Timel Noct 204.229 Avat kHP Copyright © 2015 Pearson Education, Inc. x 0200202 Mol ₂014 0.01730L 0.00200 Mag 0.01755 0.00205 met 0.01780 Trial 3 X x 419 38.25 20.45 17.80 -115M 342 Repen Sheet Dutcrmination of the Dissociation Constant of a Weak Acid C. Determination of på, of Unknown Acid Second First determination pH determination pH ml. NaOH ml. NaOH 10.50 nen 2.78 1.50 3.05 5135 - 4.6 21.40 5.14 3.77 23.00 349 4.0x 5.80 7.15 5.6.1 9.10 4.18 5.25 11.35 5.64 125 5.10 12.40 13.70 5.38 14.55 5.83 15.65 5.55 16.60 13.65 5.94 4.42 6.01 10.60 5.03 20.55 6.19 21.50 43.60 5.47 33.50 5.44 251.55 6.20 25.10 11.32 6.82 26,63 11.65 2.80 11. S 10.05 151.90 11.49 10:51 29.75 12.00 10.81 30.00 12.07 31.90 12.19 Volume at equivalence point Volume at one-half equivalence point 25.104 12.SS.L 26.00 13.00 pk, 4.50 pk. 4.60 K. X. 2.5LYD Average K., (show calculations) 32255 (SMONG") "(15) Mat (Garam Buttx 105 + Copyright © 2015 Pearson Education, Inc. 333 443 4.50 14.45 4.66 4.29 4.84 24.00 24.12 24.10 29.90 2.4.8 24.30 to 20 14.90 15.00 25.10 15:50 25.60 25.20 26.10 26.10 Third determination ml NaOH pH 2.81 8.01 4.10 13.05 4.36 13.00 S. 34 S.SC 25.00 26.50 6.63 15.90 4.95 26.10 26.10 10. 10 10.41 25.90 12.95 pk. 4.40 K. 3.98 x 5 Standard deviation of K, 7.37 x6 (( - )" (2.5")-(³)) : (²-²) 3-1 = 7.37x166 Using the volume of base at the equivalence point, its molarity, and the fact that you used 25.00 mL of acid, calculate the concentration of the unknown acid and record this on the report sheet. D. Concentration of Unknown Acid Volume of unknown acid Average molarity of NaOH from above mL of NaOH at equivalence point Molarity of unknown acid Average molarity (show calculations) Trial 1 Trial 2 Standard deviation Laboratory Experim Trial 3 B. Standardization of Sodium Hydroxide (NaOH) Solution Trial 1 Trial 2 Mass of bottle + KHP Mass of bottle Mass of KHP used 4139 14.09g Final buret reading 19.55 Initial buret reading 22,10 mL of NaOH used 13.30 17.55 Molarity of NaOH 117 M 114 M Average molarity (show calculations and standard deviation) 0.115M 117 +114 1.115 3 =115 M. Standard deviation (see Experiment 8) 0.0283M (147-15)²+(114-115)²+(115-115) ² -0.0283 M 3-1 =.117 M •114 M . =.115 M 43g Hip ARED met NOCH 204. 22gup 1 Cup 409g KHP mal x Imol NaOH Imor KHP 204.22g 419gxHPx mal Timel Noct 204.229 Avat kHP Copyright © 2015 Pearson Education, Inc. x 0200202 Mol ₂014 0.01730L 0.00200 Mag 0.01755 0.00205 met 0.01780 Trial 3 X x 419 38.25 20.45 17.80 -115M 342 Repen Sheet Dutcrmination of the Dissociation Constant of a Weak Acid C. Determination of på, of Unknown Acid Second First determination pH determination pH ml. NaOH ml. NaOH 10.50 nen 2.78 1.50 3.05 5135 - 4.6 21.40 5.14 3.77 23.00 349 4.0x 5.80 7.15 5.6.1 9.10 4.18 5.25 11.35 5.64 125 5.10 12.40 13.70 5.38 14.55 5.83 15.65 5.55 16.60 13.65 5.94 4.42 6.01 10.60 5.03 20.55 6.19 21.50 43.60 5.47 33.50 5.44 251.55 6.20 25.10 11.32 6.82 26,63 11.65 2.80 11. S 10.05 151.90 11.49 10:51 29.75 12.00 10.81 30.00 12.07 31.90 12.19 Volume at equivalence point Volume at one-half equivalence point 25.104 12.SS.L 26.00 13.00 pk, 4.50 pk. 4.60 K. X. 2.5LYD Average K., (show calculations) 32255 (SMONG") "(15) Mat (Garam Buttx 105 + Copyright © 2015 Pearson Education, Inc. 333 443 4.50 14.45 4.66 4.29 4.84 24.00 24.12 24.10 29.90 2.4.8 24.30 to 20 14.90 15.00 25.10 15:50 25.60 25.20 26.10 26.10 Third determination ml NaOH pH 2.81 8.01 4.10 13.05 4.36 13.00 S. 34 S.SC 25.00 26.50 6.63 15.90 4.95 26.10 26.10 10. 10 10.41 25.90 12.95 pk. 4.40 K. 3.98 x 5 Standard deviation of K, 7.37 x6 (( - )" (2.5")-(³)) : (²-²) 3-1 = 7.37x166
Expert Answer:
Answer rating: 100% (QA)
This question is based on pH metric titration Initially 25mL of acid is taken Therefore the pH is ... View the full answer

Related Book For 

Probability & Statistics for Engineers & Scientists
ISBN: 978-0131877115
8th edition
Authors: Ronald E. Walpole, Raymond H. Myers, Sharon L. Myers, Keying Ye
Posted Date:
Students also viewed these mathematics questions
-
Complete the following table: Lab Notebook Trial 1 Trial 2 Trial 3 Volume of HCI 100 ml 100 mL 100 mL Initial temperature of HCI 25 C 25 C 24 C Volume of NaoH 100 ml 100 mL 100 mL Initial temperature...
-
A point mass m is located a distance D from the nearest end of a thin rod of mass M and length L along the axis of the rod. Find the gravitational force exerted on the point mass by the rod.
-
A rod of weight W and uniform cross section is bent into a quarter circle and is supported as shown. Determine the bending moment at point J when θ = 30°. B
-
Write the shear and momentfunctions and draw shear and moment diagrams for the following frames: (a) Support A is a roller, B and C are fixed and support C is a pin.
-
Why should a local appliance store designate control units, even though this may be time consuming?
-
Repeat Example 7-20 for the input waveforms given in Figure 7-116. Data from Example 7-20 A shift register is often used as a way to delay a digital signal by an integral number of clock cycles. The...
-
The wall shear stress in a fully developed flow portion of a 12 -in.-diameter pipe carrying water is \(1.85 \mathrm{lb} / \mathrm{ft}^{2}\). Determine the pressure gradient, \(\partial p / \partial...
-
From the following list of performance measures, label each one as Financial, Customer, Internal Business Processes, or Learning and Growth: Percentage of on-time deliveries Employee turnover ratio...
-
Two 1.5-cm-diameter disks face each other, 2.5 mm apart. They are charged to 19 nC. A proton is shot from the negative disk toward the positive disk. What launch speed must the proton have to just...
-
A survey indicates that the mean annual wages for forensic science technicians working for local and state governments are $55,950 and $51,100, respectively. The survey includes a randomly selected...
-
Takealot's core business model operates off an e-commerce platform. Discuss e commerce and argue the importance of e-commerce in warehouse management in relation to Takealot There are 3 steps to...
-
Research the Triple Bottom Line, otherwise known as the Triple P's, introduced by John Elkington. How does this theory impact the definition of business success in today's organizations?
-
2 What is the best estimate of the number of atoms in a piece of metal of volume 50cm ? A5 x 105 B5x105 C5x10 D) 5 x 101
-
References: Valacich, J. S., Schneider, C., & Hashim, M. (2022). Information Systems Today: Managing in the Digital World (9th ed.). Pearson. 9780136735854 2.6. What is the freemium model, and how...
-
Example: Fit a polynomial of degree two and find the approximate value of f(0.8). Find the relative error occured at the point x = 0.8 where f(x) = 2 In(x + 1). Xi Yi 0.5 0.446287 0.6 0.614969 0.9...
-
A company.compiled the following information for the current year. (Assume that all raw, materials used were direct materials.) Raw materials inventory, January 1 Raw materials inventory, December 31...
-
Spotlighting opportunities for business in Africa and strategies to succeed in the world's next big growth market Acha Leke @achaleke - Senior Partner and Chairman of Africa Region, McKinsey &...
-
a) Calculate the goodwill that was paid by Major Ltd on the acquisition of Minor Ltd. [10 marks] b) Prepare the consolidated statement of financial position for Major Ltd at 31 July 20X8. [30 marks]...
-
A random sample of size n1 = 25 taken from a normal population with a standard deviation < 1 = 5 has a mean 1 = 80. A second random sample of size n2 = 36, taken from a different normal population...
-
A new cure has been developed for a certain type of cement that results in a compressive strength of 5000 kilograms per square centimeter and a standard deviation of 120. To test the hypothesis that...
-
Rework Exercise 16,6 by using the signed-rank test, In Exercise 16,6 Two types of instruments for measuring the amount of sulfur monoxide in the atmosphere are beting compared in an air-pollution...
-
Motor vehicles are necessarily non-current (fixed) assets when: A. They are intended for continuing use in the business. B. They are more than one year old. C. They are parked with the handbrake on....
-
Explain the meaning of capital maintenance.
-
The four conditions of IAS 11 [see Section 8.4.4] provide entirely adequate safeguards for the use of the percentage-of-completion method for long-term contracts. When these requirements are met,...

Study smarter with the SolutionInn App


