please show all work Reaction A: Acid-base neutralization reaction between phosphoric acid and sodium hydroxide: H3PO4(aq) +
Question:
please show all work
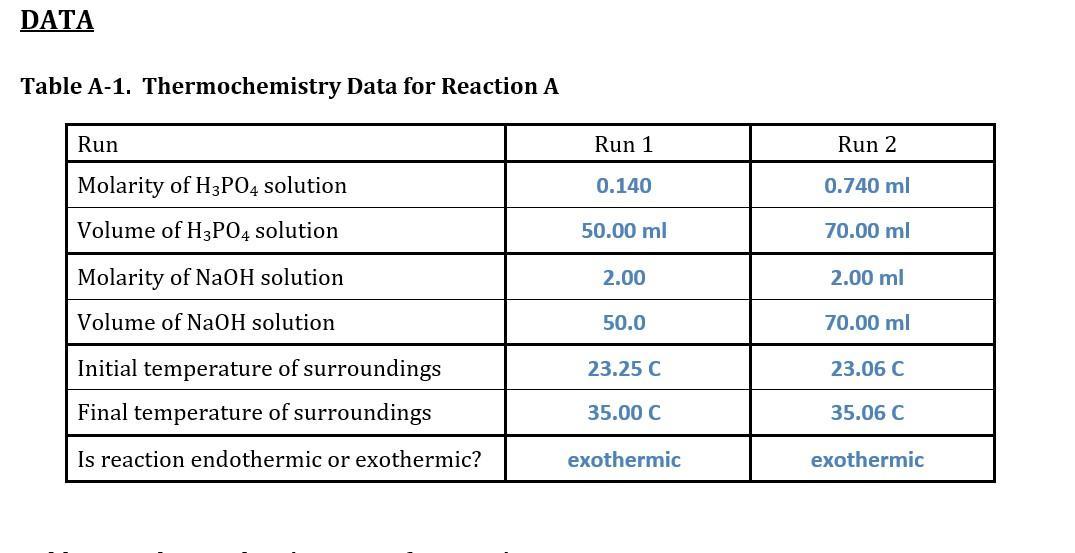
Reaction A: Acid-base neutralization reaction between phosphoric acid and sodium hydroxide:
H3PO4(aq) + 3 NaOH(aq) ⟶ 3 H2O(l) + 3 Na+(aq) + PO4^3–(aq)
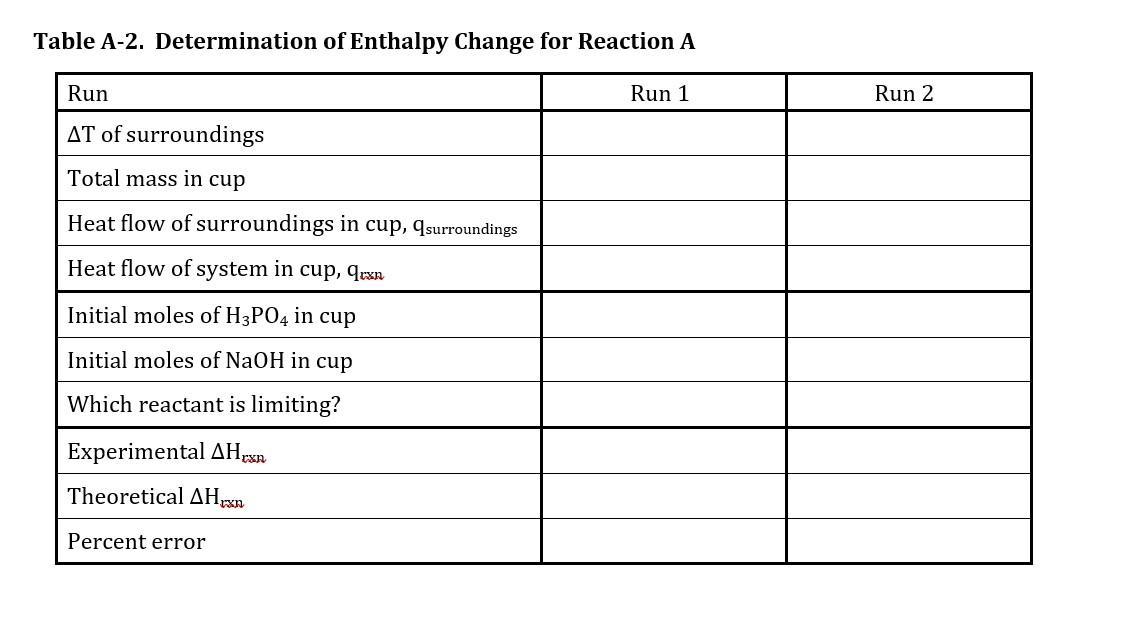
TABLES A-2 and B-2
For each of the four runs, calculate the quantities listed in Tables A-2 and B-2 by performing the steps
described in the following sections.
Initial Moles of Reactants in Cup
This is not the first quantity listed in either table but will need to be calculated along the way. For
the solid, use molar mass. For the aqueous solutions, use the measured volume and molarity to
find this. In a later step, you will find the mass of the entire solution. Here you want the mass of
only the solute.
Experimental Hrxn
There are three steps to calculate the enthalpy change using a coffee cup calorimeter. A worked-
out example is provided in Appendix A of this document.
STEP 1: Determine the heat produced or absorbed during the reaction in the coffee cup (q).
Since heat flows from the system (reactants and products) to the surroundings (water,
thermometer, stirrer, etc.) or vice versa, the temperature of the surroundings changes, and that
is what is measured. How much the temperature changes depends on the amount of heat released
or absorbed by the system (reaction), and the total mass and specific heat of the material in the
cup. Specific heat is the energy required to raise the temperature of one gram of matter by one
degree Celsius.
The equation for heat flow of the surroundings, qsurroundings which is how much heat the
surroundings gains or loses during the reaction is:
qsurroundings = total mass specific heat Tsurroundings
• The total mass is the sum of the masses of all reactants and solvents put into the cup.
− For solids, mass is generally measured with a balance.
− For pure liquids and solutions, mass is generally calculated using density and the
measured volume.
− For a dilute aqueous solution, it is assumed that the density of the solution is the same
as that of water, therefore the mass is calculated using this density and the measured
volume of the solution. This assumption is reasonable since such solutions are
generally about 95% water.
For this experiment, use the density of water at 25 C to five significant figures, which
is 0.99705 g/mL. This many significant figures are needed because the volume in some
of the runs has five significant figures.

Accounting
ISBN: 978-1337899451
27th edition
Authors: Carl S. Warren, James M. Reeve, Jonathan Duchac




