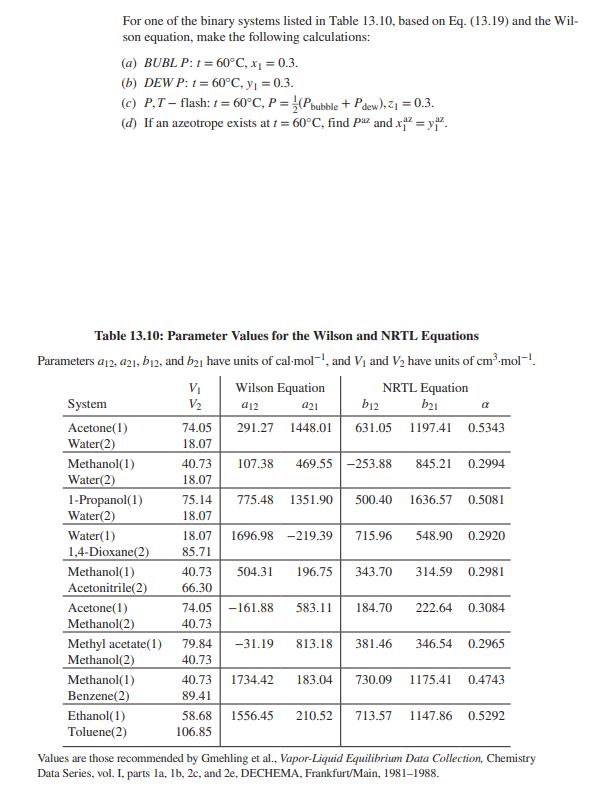
For one of the binary systems listed in Table 13.10, based on Eq. (13.19) and the...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
For one of the binary systems listed in Table 13.10, based on Eq. (13.19) and the Wil- son equation, make the following calculations: (a) BUBLP: 1 = 60°C, x₁ = 0.3. (b) DEW P: 1= 60°C, y₁ = 0.3. (c) P,T-flash: t=60°C, P = (Pbubble + Pdew), Z₁ = 0.3. (d) If an azeotrope exists at t=60°C, find Paz and x²=yiz. Table 13.10: Parameter Values for the Wilson and NRTL Equations Parameters a12, 421, b12, and b21 have units of cal-moll, and V₁ and V2 have units of cm³.mol-1. Wilson Equation System V₁ V2 a12 a21 b12 Acetone(1) 74.05 291.27 1448.01 631.05 NRTL Equation b21 1197.41 0.5343 α Water(2) 18.07 Methanol(1) 40.73 107.38 469.55 -253.88 845.21 0.2994 Water(2) 18.07 1-Propanol(1) Water(2) 75.14 775.48 1351.90 500.40 18.07 1636.57 0.5081 Water(1) 1,4-Dioxane(2) 18.07 1696.98 -219.39 85.71 715.96 548.90 0.2920 Methanol(1) Acetonitrile(2) 40.73 504.31 196.75 66.30 343.70 314.59 0.2981 Acetone(1) Methanol(2) 74.05 -161.88 583.11 184.70 40.73 222.64 0.3084 Methyl acetate(1) Methanol(2) 79.84 -31.19 813.18 40.73 381.46 346.54 0.2965 Methanol(1) Benzene(2) 40.73 1734.42 183.04 730.09 89.41 1175.41 0.4743 Ethanol(1) Toluene(2) 58.68 1556.45 210.52 106.85 713.57 1147.86 0.5292 Values are those recommended by Gmehling et al., Vapor-Liquid Equilibrium Data Collection, Chemistry Data Series, vol. I, parts la, lb, 2c, and 2e, DECHEMA, Frankfurt/Main, 1981-1988. For one of the binary systems listed in Table 13.10, based on Eq. (13.19) and the Wil- son equation, make the following calculations: (a) BUBLP: 1 = 60°C, x₁ = 0.3. (b) DEW P: 1= 60°C, y₁ = 0.3. (c) P,T-flash: t=60°C, P = (Pbubble + Pdew), Z₁ = 0.3. (d) If an azeotrope exists at t=60°C, find Paz and x²=yiz. Table 13.10: Parameter Values for the Wilson and NRTL Equations Parameters a12, 421, b12, and b21 have units of cal-moll, and V₁ and V2 have units of cm³.mol-1. Wilson Equation System V₁ V2 a12 a21 b12 Acetone(1) 74.05 291.27 1448.01 631.05 NRTL Equation b21 1197.41 0.5343 α Water(2) 18.07 Methanol(1) 40.73 107.38 469.55 -253.88 845.21 0.2994 Water(2) 18.07 1-Propanol(1) Water(2) 75.14 775.48 1351.90 500.40 18.07 1636.57 0.5081 Water(1) 1,4-Dioxane(2) 18.07 1696.98 -219.39 85.71 715.96 548.90 0.2920 Methanol(1) Acetonitrile(2) 40.73 504.31 196.75 66.30 343.70 314.59 0.2981 Acetone(1) Methanol(2) 74.05 -161.88 583.11 184.70 40.73 222.64 0.3084 Methyl acetate(1) Methanol(2) 79.84 -31.19 813.18 40.73 381.46 346.54 0.2965 Methanol(1) Benzene(2) 40.73 1734.42 183.04 730.09 89.41 1175.41 0.4743 Ethanol(1) Toluene(2) 58.68 1556.45 210.52 106.85 713.57 1147.86 0.5292 Values are those recommended by Gmehling et al., Vapor-Liquid Equilibrium Data Collection, Chemistry Data Series, vol. I, parts la, lb, 2c, and 2e, DECHEMA, Frankfurt/Main, 1981-1988.
Expert Answer:

Related Book For 

Corporate Finance Core Principles And Applications
ISBN: 9781260571127
6th Edition
Authors: Stephen Ross, Randolph Westerfield, Jeffrey Jaffe, Bradford Jordan
Posted Date:
Students also viewed these finance questions
-
= - Let f(0) 2 sine for 0 0 2. Part A: At what value of 0 does f(e) reach a maximum? (10 points) Part B: Determine the equation of a line tangent to f(0) at 0 = (10 points) Leave your answer as an...
-
Masterson, Inc., has 4 million shares of common stock outstanding. The current share price is $70, and the book value per share is $9. The company also has two bond issues outstanding. The first bond...
-
Dinklage Corp. has 8 million shares of common stock outstanding. The current share price is $74, and the book value per share is $7. The company also has two bond issues outstanding. The first bond...
-
Date 1 July 2019 1 June 2020 30 June 2020 1 July 2020 1 July 2020 30 June 2021 1 July 2021 Particulars (???) (???) (To record acquisition of delivery truck) (???) (???) (???) (To record minor repair...
-
Janet Chao, accountant of Seaward Electronics Ltd., learned that Seaward Electronics's $24 million cost of inventory at the end of last year was overstated by $3.0 million. She notified the company...
-
You have a sample of gas in a container with a movable piston, such as the one in the drawing. (a) Redraw the container to show what it might look like if the temperature of the gas is increased from...
-
An auditor's primary concern when performing tests of controls over purchasing is to determine whether: a. Purchases are properly authorized. b. Purchases are properly recorded. c. Purchase orders...
-
Using the data in Problem 4-10, test to see if there is a statistically significant relationship between sales and YouTube views at the 0.05 level of significance. Use the formulas in this chapter...
-
4. Calvin's Cleaning has employed you to investigate whether any accrual entries are needed in the business. On completion of your investigation on 30 June, you have discovered that the following...
-
1) Individually complete the Career Motivation test (click on tests/quizzes/polls) from the website http://queendom.com. There are a number of different tests you can complete if you wish. Review the...
-
You have collected the following data: If you place these five numbers in a bag and randomly select one, what is the probability the number (X) will be ... a. equal to 6 ? b. less than 11 ? c....
-
You go out to the best restaurant in town and order a steak dinner for 40. After eating half of the steak, you realize that you are quite full. Your partner wants you to finish your dinner, because...
-
For each of the following situations, calculate the population standard error of the mean \(\sigma \mathrm{X}^{-}\). a. \(\sigma=18 ; N=36\) b. \(\sigma=9.42 ; N=49\) c. \(\sigma=1.87 ; N=60\) d....
-
A lottery contains 500 tickets. In this lottery, there are 25 prizes of \(\$ 1,10\) prizes of \(\$ 5\), and 5 prizes of \(\$ 25\). What is the probability of \(\ldots\) a. winning nothing \((\$ 0)\)...
-
For each of the following situations, calculate the \(z\)-statistic \((z)\), make a decision about the null hypothesis (reject, do not reject), and indicate the level of significance \((p>.05, p
-
Do you believe that clients in drug treatment programs should be drug tested? Why or why not? (Problems of Substance Abuse and Addiction)
-
In the previous problem, suppose the fixed asset actually qualifies for 100 percent bonus depreciation. All the other facts are the same. What is the new NPV? Data from Previous Problem Coris Meats...
-
In the previous problem, if the SEC filing fee and associated administrative expenses of the offering are $1.3 million, how many shares need to be sold? Data from Previous Problem The Trafford...
-
Suppose the spot exchange rate for the Canadian dollar is Can$1.29 and the six-month forward rate is Can$1.34. a. Which is worth more, a U.S. dollar or a Canadian dollar? b. Assuming absolute PPP...
-
Determine the amplitudes of motion of the three masses in Fig. 6.40 when a harmonic force \(F(t)=F_{0} \sin \omega t\) is applied to the lower left mass with \(m=1 \mathrm{~kg}, k=1000 \mathrm{~N} /...
-
(a) Determine the natural frequencies and mode shapes of the torsional system shown in Fig. 6.11 for \(k_{t 1}=k_{t 2}=k_{t 3}=k_{t}\) and \(J_{1}=J_{2}=J_{3}=J_{0}\). (b) If a torque \(M_{t 3}(t)=\)...
-
Using the results of Problems 6.24 and 6.56, determine the modal matrix \([X]\) of the system shown in Fig. 6.29 and derive the uncoupled equations of motion. Data From Problem 6.24:- Find the...

Study smarter with the SolutionInn App


