DETERMINATION OF IRON CONTENT IN A FERROUS AMMONIUM SULFATE UNKNOWN Obtain an unknown sample from your...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
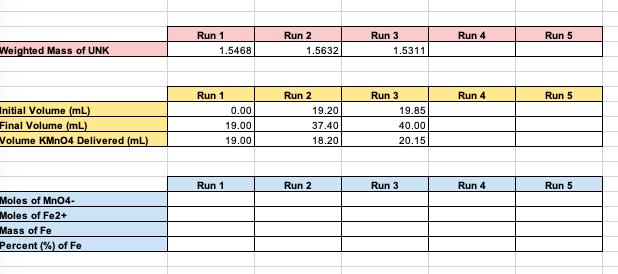
DETERMINATION OF IRON CONTENT IN A FERROUS AMMONIUM SULFATE UNKNOWN Obtain an unknown sample from your professor and record the unknown number in your notebook. Weigh out 3-4 samples of between 1.5000g and 2.0000g ferrous ammonium sulfate unknown and place in 250 mL flasks. Add 15 mL of distilled water and 1 mL of concentrated HCI to each sample. Swirl to dissolve sample completely. Gently warm on a hot plate at your bench. Wash down the sides of the flask with some distilled water. Add 0.2 M Potassium Permanganate solution (not your standardized solution) dropwise until a distinct yellow color forms. (should be about 2 drops) Carry out the following procedure individually on each sample. Add 25 mL of the Zimmermann-Reinhardt reagent and 300 mL of distilled water. Titrate immediately with your standardized KMnO4 solution to the first faint pink color that persists for 15 to 30 seconds. Do not titrate rapidly at any time. The pink color results from excess MnO4, indicating that all Fe²+ has been oxidized to Fe³+, Weighted Mass of UNK Initial Volume (mL) Final Volume (mL) Volume KMnO4 Delivered (mL) Moles of MnO4- Moles of Fe2+ Mass of Fe Percent (%) of Fe Run 1 1.5468 0.00 19.00 19.00 Run 1 Run 1 Run 2 1.5632 19.20 37.40 18.20 Run 2 Run 2 Run 3 1.5311 19.85 40.00 20.15 Run 3 Run 3 Run 4 Run 4 Run 4 Run 5 Run 5 Run 5 DETERMINATION OF IRON CONTENT IN A FERROUS AMMONIUM SULFATE UNKNOWN Obtain an unknown sample from your professor and record the unknown number in your notebook. Weigh out 3-4 samples of between 1.5000g and 2.0000g ferrous ammonium sulfate unknown and place in 250 mL flasks. Add 15 mL of distilled water and 1 mL of concentrated HCI to each sample. Swirl to dissolve sample completely. Gently warm on a hot plate at your bench. Wash down the sides of the flask with some distilled water. Add 0.2 M Potassium Permanganate solution (not your standardized solution) dropwise until a distinct yellow color forms. (should be about 2 drops) Carry out the following procedure individually on each sample. Add 25 mL of the Zimmermann-Reinhardt reagent and 300 mL of distilled water. Titrate immediately with your standardized KMnO4 solution to the first faint pink color that persists for 15 to 30 seconds. Do not titrate rapidly at any time. The pink color results from excess MnO4, indicating that all Fe²+ has been oxidized to Fe³+, Weighted Mass of UNK Initial Volume (mL) Final Volume (mL) Volume KMnO4 Delivered (mL) Moles of MnO4- Moles of Fe2+ Mass of Fe Percent (%) of Fe Run 1 1.5468 0.00 19.00 19.00 Run 1 Run 1 Run 2 1.5632 19.20 37.40 18.20 Run 2 Run 2 Run 3 1.5311 19.85 40.00 20.15 Run 3 Run 3 Run 4 Run 4 Run 4 Run 5 Run 5 Run 5
Expert Answer:
Answer rating: 100% (QA)
Molasity of Initial V ml 010 1920 1985 Final V ml 190 3740 4000 V 190 ... View the full answer

Related Book For 

Posted Date:
Students also viewed these mechanical engineering questions
-
A dilute solution of potassium permanganate in water at 25C was prepared. The solution was in a horizontal tube of length 10 cm, and at first there was a linear gradation of intensity of the purple...
-
Potassium sulfate solution reacts with barium bromide solution to produce a precipitate of barium sulfate and a solution of potassium bromide. Write the molecular equation for this reaction. Then...
-
A 35.2-mL, 1.66 M KMnO4 solution is mixed with 16.7mL of 0.892 M KMnO4 solution. Calculate the concentration of the final solution.
-
Susan recently quit working for a local firm and has yet to find a new job. She knows she can maintain her health insurance from her old employer due to COBRA. How much will it likely cost her for...
-
Suppose that the government imposes a proportional income tax on the representative consumer's wage income. That is, the consumer's wage income is w(1 t)(h l) where t is the tax rate. What effect...
-
Obtain automobile industry sales and sales of major automobile manufacturers for the last five years from secondary sources.
-
Describe how workers and firms marry each other in the labor market when there are many types of jobs offering various levels of risk to their workers.
-
The G. Company's financing plans for next year include the sale of long-term bonds with a 12 percent coupon. The company believes it can sell the bonds at a price that will give a yield to maturity...
-
The Refining Department of SweetBeet, Inc. had 72,000 tons of sugar to account for in July. Of the 72,000 tons, 40,000 tons were completed and transferred to the Boiling Department, and the remaining...
-
A steel company is producing steel for a new contract. The contract specifies the information in the following table for the steel. The steel company mixes batches of eight different available...
-
Consider the three bail scenarios. James, a teenager, was accused of stealing a bus pass. After police arrested him, the court set a $1,500 bail that neither he nor his family could afford. He...
-
JDBC is standard Java Database Connectivity, and the JDBC API can be considered the Java Database Connectivity Application Programming Interface.(True/False)
-
When establishing a database connection, the DriverManager.getConnection() method is a better method than the Driver.connect() method. (True/False)
-
Prepare a three-minute presentation, using an outline, about one strategy for managing speech anxiety. You may include sources in the speaking tool or from your own research. Before delivering the...
-
To prepare for an upcoming presentation, create a storyboard. Use software, a PowerPoint template, or paper and sticky notes. Draw pictures and write a few words to capture what you would create on...
-
When using a CallableStatement object to run a stored procedure, you need to register the __________ parameters by using the _____________ method. a. IN/OUT, getParameters() b. IN,...
-
A chemist fills a reaction vessel with 4.43 atm methane (CH) gas, 9.19 atm oxygen (O) gas, 7.38 atm carbon dioxide (CO) gas, and 4.79 atm hydrogen (H) gas at a temperature of 25.0C. Under these...
-
Use of the contraceptive Depo Provera appears to triple women's risk of infection with chlamydia and gonorrhea , a study reports today. An estimated 20 million to 30 million women worldwide use Depo...
-
An acidic solution containing 0.010 M La3+ is treated with NaOH until La(OH)3 precipitates. At what pH does this occur?
-
Sulfide ion was determined by indirect titration with EDTA. To a solution containing 25.00 mL of 0.04332 M Cu(ClO4)2 plus 15 mL of 1M acetate buffer (pH 4.5) were added 25.00 mL of unknown sulfide...
-
What are the general steps in developing an isocratic separation for reversed-phase chromatography with one organic solvent and temperature as variables?
-
Apple stock is selling for \($120\) per share. Call options with a \($117\) exercise price are priced at \($12.\) What is the intrinsic value of the option, and what is the time value?
-
Ibrahim bought 200 shares of a stock trading in the Abu Dhabi Securities Exchange at AED 12 (United Arab Emirates dirham) per share. Over time, the price of the stock increased to AED 18 per share....
-
Twitter is trading at \($34.50.\) Call options with a strike price of \($35\) are priced at \($2.30\) . What is the intrinsic value of the option, and what is the time value?

Study smarter with the SolutionInn App


