Determine the polarity and molecular geometry of the following compounds based on electronegativity differences. Molecular Polarity...
Fantastic news! We've Found the answer you've been seeking!
Question:
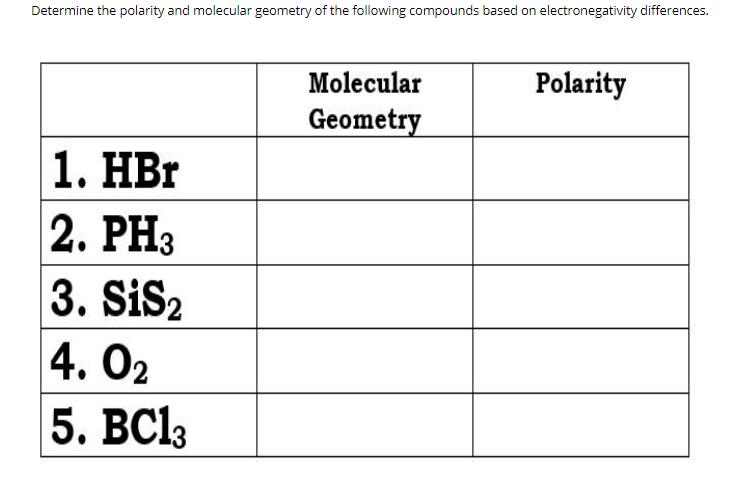
Transcribed Image Text:
Determine the polarity and molecular geometry of the following compounds based on electronegativity differences. Molecular Polarity Geometry 1. HBr 2. PH3 3. SiS2 4. О2 5. ВС(3 Determine the polarity and molecular geometry of the following compounds based on electronegativity differences. Molecular Polarity Geometry 1. HBr 2. PH3 3. SiS2 4. О2 5. ВС(3
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Determine the polarity and molecular geometry of the following compounds based on electronegativity differences. Molecular Polarity Geometry 1. HBr 2. PH 3. Sis2 4. O2 5. i
-
The following compounds have the same molecular formulas (C4H10). Which one would you expect to have a higher boiling point?
-
Compounds L and M have the molecular formula C7H14, Compounds L and M are optically inactive, are nonresolvable, and are diastereomers of each other. Catalytic hydrogenation of either L or M yields...
-
This bar chart displays the demographics (age group and gender) of a Business Analysis class Business Analysts Students 23-33 3410 M lem How many male students are in the class? 65 80 130 50
-
Your friend says, Id rather be a big fish in a small pond than a small fish in a big pond. What concept does this comment illustrate?
-
The adjusted trial balance for Tybalt Construction as of December 31, 2017, follows. O. Tybalt invested $5,000 cash in the business during year 2017 (the December 31, 2016, credit balance of the O....
-
Some acacia trees have evolved a special relationship with certain species of ants. The trees provide food and nesting sites (in the form of hollow thorns) for the ants. The ants attack insects and...
-
Use the model in the File C12 to work this problem. a. Refer back to Problem. Now assume that the debt ratio is increased to 65 percent, causing all interest rates to rise by 1 percentage point, to...
-
A licensed practical/vocational nurse (LP/VN) suffered a back injury and is working while taking prescription narcotic analgesics. A coworker noticed that the impaired nurse is risking clients' care...
-
Lombard Ltd has been offered a contract for which there is available production capacity. The contract is for 20,000 identical items, manufactured by an intricate assembly operation, to be produced...
-
Consider the following series. For the work that follows, the root or ratio test will likely be convenient! n=1 (a) Determine the value(s) of r (if any) for which the series converges absolutely. (b)...
-
Good Time Clothing has sales of $25 million and variable costs of $10 million. Its output is 1 million pairs of shorts. Its fixed costs are equal to $5 million. Using above information, complete the...
-
1. Cost of goods sold included: Attempted: 0/1 Attempted: 0/1 Moolan Enterprise, owned by Moolan, is in the business of trading in beauty equipment, with an accounting year end of 31 December. Most...
-
Statement of Comprehensive Income for the year ended 31 December 2022 Sales (all credit) Cost of sales Opening inventory Purchases (all credit) Closing inventory Gross profit Statement of Financial...
-
(1) Life on a medieval estate centered on the castle, the residence of the lords family and refuge for the estates population in time of war. (2) After Christian crusaders had studied Byzantine and...
-
Explain why empowerment is so important to supervisors, employees, and organizations, individually and as a whole. Please share/provide some firsthand examples from your work or social experiences....
-
Wetzel, Inc. has 10,000 shares of cumulative preferred stock outstanding, with annual dividends paid at a rate of $4 per share. Wetzel, Inc. also has 40,000 shares of common stock outstanding....
-
Portal Manufacturing has total fixed costs of $520,000. A unit of product sells for $15 and variable costs per unit are $11. a). Prepare a contribution margin income statement showing predicted net...
-
Name the kinds of attractive forces that must be overcome in order to (a) Boil liquid ammonia, (b) Melt solid phosphorus (P4), (c) Dissolve CsI in liquid HF, (d) Melt potassium metal.
-
W rite the formulas and names of the hydrides of the following second-period elements: Li, C, N, O, F. Predict their reactions with water.
-
Draw the Lewis structure for the BeCl42- ion. Predict its geometry and describe the hybridization state of the Be atom?
-
(a) The longitudinal data set "v4c" is in the vertical format; transform it into the horizontal format. (b) Transform the data set you obtained in part (a) back into the vertical format.
-
Perform some exploratory analysis on the DTS study described above. (a) Compute the mean and standard deviation of the HamD scores for the two treatment groups at each time point. (b) Treat repeated...
-
For the DTS study, use subjects with all five assessments in HamD scores in the CAU group for this question. The intraclass correlation coefficient among the repeated measures in Ham-D scores can be...

Study smarter with the SolutionInn App



