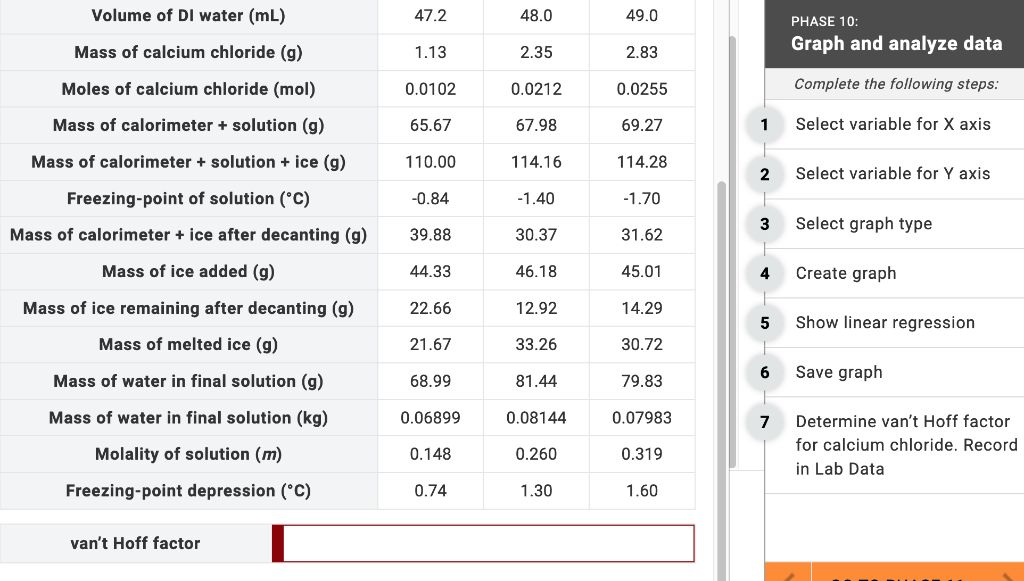
Determine van't Hoff factor for calcium chloride. Record in LabData. Volume of DI water (mL) Mass of
Fantastic news! We've Found the answer you've been seeking!
Question:
Determine van't Hoff factor for calcium chloride. Record in LabData.
Transcribed Image Text:
Volume of DI water (mL) Mass of calcium chloride (g) Moles of calcium chloride (mol) Mass of calorimeter + solution (g) Mass of calorimeter + solution + ice (g) Freezing-point of solution (°C) Mass of calorimeter + ice after decanting (g) Mass of ice added (g) Mass of ice remaining after decanting (g) Mass of melted ice (g) Mass of water in final solution (g) Mass of water in final solution (kg) Molality of solution (m) Freezing-point depression (°C) van't Hoff factor 47.2 1.13 0.0102 65.67 110.00 -0.84 39.88 44.33 22.66 21.67 68.99 0.06899 0.148 0.74 48.0 2.35 0.0212 67.98 114.16 -1.40 30.37 46.18 12.92 33.26 81.44 0.08144 0.260 1.30 49.0 2.83 0.0255 69.27 114.28 -1.70 31.62 45.01 14.29 30.72 79.83 0.07983 0.319 1.60 1 2 3 5 7 PHASE 10: Graph and analyze data Complete the following steps: Select variable for X axis Select variable for Y axis Select graph type Create graph Show linear regression. Save graph Determine van't Hoff factor for calcium chloride. Record in Lab Data Volume of DI water (mL) Mass of calcium chloride (g) Moles of calcium chloride (mol) Mass of calorimeter + solution (g) Mass of calorimeter + solution + ice (g) Freezing-point of solution (°C) Mass of calorimeter + ice after decanting (g) Mass of ice added (g) Mass of ice remaining after decanting (g) Mass of melted ice (g) Mass of water in final solution (g) Mass of water in final solution (kg) Molality of solution (m) Freezing-point depression (°C) van't Hoff factor 47.2 1.13 0.0102 65.67 110.00 -0.84 39.88 44.33 22.66 21.67 68.99 0.06899 0.148 0.74 48.0 2.35 0.0212 67.98 114.16 -1.40 30.37 46.18 12.92 33.26 81.44 0.08144 0.260 1.30 49.0 2.83 0.0255 69.27 114.28 -1.70 31.62 45.01 14.29 30.72 79.83 0.07983 0.319 1.60 1 2 3 5 7 PHASE 10: Graph and analyze data Complete the following steps: Select variable for X axis Select variable for Y axis Select graph type Create graph Show linear regression. Save graph Determine van't Hoff factor for calcium chloride. Record in Lab Data
Expert Answer:
Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-1119498759
4th edition
Authors: Richard M. Felder, ? Ronald W. Rousseau, ? Lisa G. Bullard
Posted Date:
Students also viewed these chemical engineering questions
-
Determine the vant Hoff factor for the following ionic solute dissolved in water. 823918
-
Calcium chloride is a salt used in a number of food and medicinal applications and in brine for refrigeration systems. Its most distinctive property is its affinity for water: in its anhydrous form...
-
For each of the following independent scenarios, determine whether Van Allen Corporation is a principal or an agent for purposes of applying the 5-step revenue recognition model. 1. Van Allen is a...
-
Compare the ACA passed in 2010 with the Massachusetts health plan adopted in 2006 (use the Internet to obtain more details than we have provided in this chapter). Be sure to note the similarities and...
-
What accounts are typically affected by transactions in the revenue cycle? Identify the relationships among them.
-
A student siphons water over a 8.5-m-high wall at sea level. She then climbs to the summit of Mount Shasta (elevation 4390 m, P atm = 58.5 kPa) and attempts the same experiment. Comment on her...
-
The following transactions occurred during April 2010, for Sandy Salon Products, Inc.: Requirement 1. Journalize the April transactions for Sandy Salon Products, Inc. Omit explanations. Apr 3...
-
R.T. Jackson started Jackson Company on January 1, 2013. The company experienced the following events during its first year of operation: 1. Earned $3,000 of cash revenue for performing services. 2....
-
Please solve correctly Explain the basic differences between. the Marketing Mix described by Kotler in his book and the Film Marketing Mix described by Finola Keriggan, Explain the meaning of the two...
-
In December, Northern Kiteboarding magazine collected $80,000 for subscriptions for the next calendar year. The company collects cash in advance and then downloads the magazines to subscribers each...
-
A pipeline consists of 4 stages, as shown in the Figure below. Each stage performs the operation yout= yin+axx Determine the overall computation performed.
-
Calculate the Net income/Loss from milestone three income statement. Milestone Three - Income Statement Revenue: Collars Leashes Harnesses Total Revenue: Cost of goods sold $ 13,200 12,320 12,500 $...
-
Coffee Maker Supreme (CMS) has been in the business of manufacturing coffee makers for three generations. During the first and second genera- tions the growth in demand was modestly positive, and...
-
a. What is the yield to maturity of this bond as of 08/25/2015? b. What are the risk factors the yield you determined in part a offers rewards based on the information above? Explain. c. On...
-
1) What factors contribute to follower growth? 2) How hashtag affect the performance of TikTok content?
-
Identify two digital marketing methods that can be used for brand awareness and support with reasons. Define short-term and long-term goals for brand awareness in the digital marketing strategy. How...
-
Suppose the natural gas industry exhibited the the following market condition Total Market Sales 43,100 Company A sales 6,800 Company B sales 5,000 Company C sales 3,780 Company D sales 3,500 What's...
-
Find the equations of the ellipses satisfying the given conditions. The center of each is at the origin. Passes through (2, 2) and (1, 4)
-
Estimate the heat of vaporization (kJ/mol) of benzene at 25C, using each of the following correlations and data: (a) The heat of vaporization at the normal boiling point and Watsons correlation. (b)...
-
Vapor?liquid equilibrium calculations can sometimes be simplified through the use of a quantity called the relative volatility, which may be defined to terms of the following depiction of vapor and...
-
Using Raoults law or Henrys law for each substance (whichever one you think appropriate), calculate the pressure and gas-phase composition (mole fractions) in a system containing a liquid that is 0.3...
-
A cylindrical tank of methanol has a mass of \(60 \mathrm{~kg}\) and a volume of \(75 \mathrm{~L}\). Determine the methanol's weight, density, and specific gravity. Take the gravitational...
-
A mass of \(0.5-\mathrm{kg}\) of argon is maintained at \(1400 \mathrm{kPa}\) and \(40^{\circ} \mathrm{C}\) in a tank. What is the volume of the tank?
-
The air in an automobile tire with a volume of \(0.015 \mathrm{~m}^{3}\) is at \(30^{\circ} \mathrm{C}\) and \(140 \mathrm{kPa}\) (gage). Determine the amount of air that must be added to raise the...

Study smarter with the SolutionInn App



