Determine whether the system of linear equations has one and only one solution, infinitely many solutions,...
Fantastic news! We've Found the answer you've been seeking!
Question:
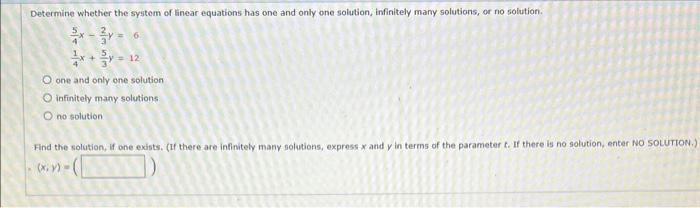
Transcribed Image Text:
Determine whether the system of linear equations has one and only one solution, infinitely many solutions, or no solution. 6 <= 12 O one and only one solution O infinitely many solutions O no solution. Find the solution, if one exists. (If there are infinitely many solutions, express x and y in terms of the parameter t. If there is no solution, enter NO SOLUTION.) (x, y) = Determine whether the system of linear equations has one and only one solution, infinitely many solutions, or no solution. 6 <= 12 O one and only one solution O infinitely many solutions O no solution. Find the solution, if one exists. (If there are infinitely many solutions, express x and y in terms of the parameter t. If there is no solution, enter NO SOLUTION.) (x, y) =
Expert Answer:
Answer rating: 100% (QA)
2x 4 Determine 5 X 6 X 1 1276 X 12 12 X 1x 1ex x 3 12 X 12 ... View the full answer

Related Book For 

Posted Date:
Students also viewed these finance questions
-
How do you perform chest compressions when providing high-quality CPR to a child victim? OA. By compressing the chest at least one third the depth of the chest, about 2 inches (5 cm) OB. By...
-
Read the following scenarios. For each scenario you need to decide which of the following four methods is appropriate. A. One sample t test (6 of them) B. Paired samples t test (5 of them) C....
-
Mark each statement True or False. Justify each answer. (If true, cite appropriate facts or theorems. If false, explain why or give a counterexample that shows why the statement is not true in every...
-
April Moon set a record in flight shooting. In 1981 in Utah, she sent an arrow a horizontal distance of 950 m. What was the speed of the arrow at the top of the flight if the arrow was launched at an...
-
The parallel-plate air capacitor in Fig consists of two horizontal conducting plates of equal area A. The bottom plate rests on a fixed support, and the top plate is suspended by four springs with...
-
Only the D sugars are shown in figure. Draw Fischer projections for the following L sugars: (a) L-Xylose (b) L-Galactose (c)L-Allose - - - D-Glyceraldehyde - R/L - - . 2R - - C- CH2 D-Erythrose...
-
Reactions of stakeholders who do not feel that accounting provides serviceable information include: resignation and loyalty; voice; and exit. 1 Why are exit and voice of particular importance as...
-
Prompt Parcel Service operates a fleet of delivery trucks in a large metropolitan area. A careful study by the companys cost analyst has determined that if a truck is driven 120,000 kilometres during...
-
Patton Dyes manufactures colorings, primarily for textiles. Information on the work in process follows: Beginning inventory, 150,000 partially complete liters. Ending inventory, 120,000 liters; units...
-
A woman wishes to sell a car within the next 4 weeks. She expects to receive one bid or offer each week from a prospective buyer. The weekly offer is a random variable, which has the following...
-
Pharoah Railroad Co. is about to issue $270,000 of 10-year bonds paying an 12% interest rate, with interest payable annually. The discount rate for such securities is 8%. Click here to view the...
-
Leaders must be clear about what drives them to authentically navigate the landscape and be able to see and understand other perspectives, as well as to be patient and persistent to be an influence....
-
Writing an executive summary should occur after the business plan is written. Think about a project or proposal that will either increase revenue or decrease expenses. Write a two-page concise...
-
What role does innovation play in disaster/emergency response planning? What is the role of the DNP-prepared nurse in disaster mitigation and response?
-
Indicate why you agree or disagree with the following statement: "Today, the proceeds from most original-issue, high-yield bonds are used for leveraged buyouts and recapitalizations."
-
Describe at least four of the features of the U.S. Department of Veterans Affairs Nursing Academy (VANA) that differentiate it from the other academicpractice partnerships described. a. Using one of...
-
a. What was the spot exchange rate of South Korea won for U.S. dollars (USD/KRW) on July 15, 2016? (Round your answer to 4 decimal places. (e.g., 32.1616)) b. What was the one-month forward exchange...
-
Nike manufactures shoes and sportswear. How has the Internet changed the way this company communicates with its suppliers and retail customers?
-
Find the adjugate of each of the following matrices. (a) (b) 3 1 0 0 -1 I 212 122
-
In each case, find all [a b c d]T m R4 such that the given set is orthogonal. (a) {[1 2 1 O]T, [1 -1 1 3]T, [2 -1 0 -1], [a b c d]T} (b) {[1 0 - 1]T, [2 1 1]T,[l -3 1 0]T, [a b c d}T}
-
Find a, b, and c so that the system x + ay + cz = 0 bx + cy - 3z = 1 ax: + 2y + bz = 5 has the solution x = 3,y = -1, z = 2.
-
Calculate the change in entropy for the conversion of \(1 \mathrm{~mol}\) of ice to liquid at \(273 \mathrm{~K}\) and \(1 \mathrm{~atm}\). The latent heat of fusion is \(6500 \mathrm{~J} /...
-
Calculate the change in entropy when 5 moles of an ideal gas expands from a volume of \(5 \mathrm{~L}\) to \(50 \mathrm{~L}\) at \(27^{\circ} \mathrm{C}\).
-
Calculate the entropy change when \(96 \mathrm{~g}\) of methane is heated from \(35^{\circ} \mathrm{C}\) to \(200^{\circ} \mathrm{C}\) at constant volume. Assume \(C_{V}=1.735 \mathrm{~kJ} /...

Study smarter with the SolutionInn App


