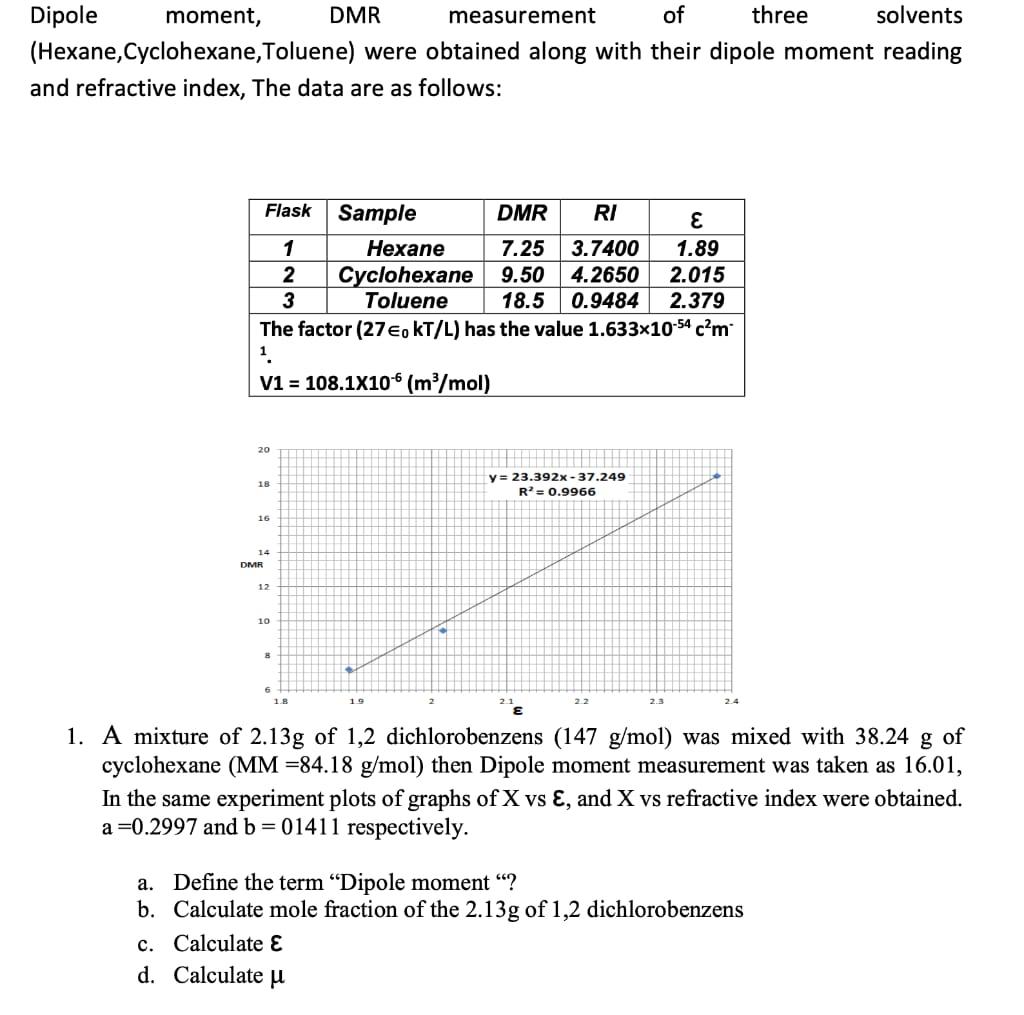
Dipole moment, (Hexane,Cyclohexane,Toluene) and refractive index, The data are as follows: Flask 1 2 3 20...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Dipole moment, (Hexane,Cyclohexane,Toluene) and refractive index, The data are as follows: Flask 1 2 3 20 18 દ 1.89 2.015 18.5 0.9484 2.379 The factor (27€, KT/L) has the value 1.633x10-54 c²m 1. V1 = 108.1X106 (m³/mol) 16 14 DMR 12 10 8 6 DMR measurement of three solvents were obtained along with their dipole moment reading 1.8 DMR RI Hexane 7.25 3.7400 Cyclohexane 9.50 4.2650 Toluene Sample c. Calculate E d. Calculate μ 19 y= 23.392x-37.249 R² = 0.9966 2.2 2.3 1. A mixture of 2.13g of 1,2 dichlorobenzens (147 g/mol) was mixed with 38.24 g of cyclohexane (MM -84.18 g/mol) then Dipole moment measurement was taken as 16.01, In the same experiment plots of graphs of X vs E, and X vs refractive index were obtained. a=0.2997 and b = 01411 respectively. a. Define the term "Dipole moment "? b. Calculate mole fraction of the 2.13g of 1,2 dichlorobenzens Dipole moment, (Hexane,Cyclohexane,Toluene) and refractive index, The data are as follows: Flask 1 2 3 20 18 દ 1.89 2.015 18.5 0.9484 2.379 The factor (27€, KT/L) has the value 1.633x10-54 c²m 1. V1 = 108.1X106 (m³/mol) 16 14 DMR 12 10 8 6 DMR measurement of three solvents were obtained along with their dipole moment reading 1.8 DMR RI Hexane 7.25 3.7400 Cyclohexane 9.50 4.2650 Toluene Sample c. Calculate E d. Calculate μ 19 y= 23.392x-37.249 R² = 0.9966 2.2 2.3 1. A mixture of 2.13g of 1,2 dichlorobenzens (147 g/mol) was mixed with 38.24 g of cyclohexane (MM -84.18 g/mol) then Dipole moment measurement was taken as 16.01, In the same experiment plots of graphs of X vs E, and X vs refractive index were obtained. a=0.2997 and b = 01411 respectively. a. Define the term "Dipole moment "? b. Calculate mole fraction of the 2.13g of 1,2 dichlorobenzens Dipole moment, (Hexane,Cyclohexane,Toluene) and refractive index, The data are as follows: Flask 1 2 3 20 18 દ 1.89 2.015 18.5 0.9484 2.379 The factor (27€, KT/L) has the value 1.633x10-54 c²m 1. V1 = 108.1X106 (m³/mol) 16 14 DMR 12 10 8 6 DMR measurement of three solvents were obtained along with their dipole moment reading 1.8 DMR RI Hexane 7.25 3.7400 Cyclohexane 9.50 4.2650 Toluene Sample c. Calculate E d. Calculate μ 19 y= 23.392x-37.249 R² = 0.9966 2.2 2.3 1. A mixture of 2.13g of 1,2 dichlorobenzens (147 g/mol) was mixed with 38.24 g of cyclohexane (MM -84.18 g/mol) then Dipole moment measurement was taken as 16.01, In the same experiment plots of graphs of X vs E, and X vs refractive index were obtained. a=0.2997 and b = 01411 respectively. a. Define the term "Dipole moment "? b. Calculate mole fraction of the 2.13g of 1,2 dichlorobenzens Dipole moment, (Hexane,Cyclohexane,Toluene) and refractive index, The data are as follows: Flask 1 2 3 20 18 દ 1.89 2.015 18.5 0.9484 2.379 The factor (27€, KT/L) has the value 1.633x10-54 c²m 1. V1 = 108.1X106 (m³/mol) 16 14 DMR 12 10 8 6 DMR measurement of three solvents were obtained along with their dipole moment reading 1.8 DMR RI Hexane 7.25 3.7400 Cyclohexane 9.50 4.2650 Toluene Sample c. Calculate E d. Calculate μ 19 y= 23.392x-37.249 R² = 0.9966 2.2 2.3 1. A mixture of 2.13g of 1,2 dichlorobenzens (147 g/mol) was mixed with 38.24 g of cyclohexane (MM -84.18 g/mol) then Dipole moment measurement was taken as 16.01, In the same experiment plots of graphs of X vs E, and X vs refractive index were obtained. a=0.2997 and b = 01411 respectively. a. Define the term "Dipole moment "? b. Calculate mole fraction of the 2.13g of 1,2 dichlorobenzens
Expert Answer:
Answer rating: 100% (QA)
Dipole moment occur when there is separation of charge imic band ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
For ten paired samples, the data are as listed here. For d = the difference between paired observations (x1 x2), use the 0.10 level of significance in testing H0: md = 0 versus H1: md 0. Sample: 1 2...
-
For nine paired samples, the data are as listed in the table. For d = the difference between paired observations (x1 x2), use the 0.10 level of significance in testing H0: md ¤ 0 versus H1: md...
-
Factor A Factor B 1 2 3 4 Xj for Factor B 1 2 3 4 1 2500 2 9 10 6 9 8500 3 14 11 16 12 13250 Xi for Factor A 8333 8000 9000 7333 X8083
-
Christine has three cars that must be overhauled by her ace mechanic, Megan. Given the following data about the cars, use least slack per remaining operation to determine Megans scheduling priority...
-
Redo Exercise 14.61 when the dosage is 20 milligrams. Note the greatly increased width of the confidence limits for the expected number of deaths and of the limits of prediction. This example...
-
Explain how, and why, investment income differs when an investor accounts for a strategic long-term equity investment using the (a) Cost model, (b) Equity method, and (c) Fair value through profit or...
-
What book introduced the structural and behavioral consideration of fraud? What is the iceberg theory?
-
You have $1,000,000 worth of equipment at the job site and wish to minimize your risk of direct property damage by taking out an insurance policy. The insurance company provides you with its...
-
. You have been asked to develop a pro forma statement of cash flow for Bets distribution center, and Internet based order fulfillment and warehouse property. In addition to recoverable operating...
-
A solar collector design consists of several parallel tubes each enclosed concentrically in an outer tube which is transparent to solar radiation. The tubes are thin walled with diameter of the inner...
-
During the good times at Toys 'R' Us, the company gained business by offering popular toys at competitive prices. How do you think, they were able to offer lower prices? During the good times at Toys...
-
Journalize basic transactions and adjusting entries. Instructions 7/31 Supplies Expense 900 Supplies 7/1 Bal. 1,100 7/31 900 7/10 650 Accounts Receivable 7/31 500 Salaries and Wages Expense 7/15 7/31...
-
Common Stockholders' Profitability Analysis A company reports the following: Net income Preferred dividends Average stockholders' equity $210,000 8,400 1,981,132 983,415 Average common stockholders'...
-
What is the Operating profit margin for Sam's appliances for the year ending 2018?
-
A portfolio has 30% of its value in IBM shares and the rest in Microsoft (MSFT). The volatility of IBM and MSFT are 25% and 33%, respectively, and the correlation between IBM and MSFT is 0.6. What is...
-
5x-6 if x3 a. Graph f(x)= x if x>3 b. Find f(1) and f(5). c. State the domain of the function.
-
Profit is the result of sales minus all variable costs. True False Cost of sales are variable; cost of labour also contains variable elements and never contains fixed elements. True False The...
-
Several months have passed and the Managing Partner approved and properly filed the Complaint and properly submitted the Request for Production of Documents that you drafted. In fact, it has been 75...
-
During May 2009, visitors to usatoday.com spent an average of 12.2 minutes per visit, compared to 11.0 minutes for visitors to washingtonpost.com. Assuming that file XR11030 contains the sample data...
-
A company executive has read with interest the finding that the average U.S. office worker receives about 60 e-mails per day. Assume that an executive, wishing to replicate this study within her own...
-
A national political candidate has commissioned a study to determine the percentage of registered voters who intend to vote for him in the upcoming election. To have 95% confidence that the sample...
-
Outline the steps in the fraud hypothesis testing approach. What are Type II errors? How does one avoid Type II errors?
-
You determine the following data about a local government in the southeast. Determine the ratios of unreserved fund balance and reserved fund balance to total revenues. Are these ratios favorable?...
-
Assume that Debt Service Expenditures is \(\$ 16.2\) million and Total Revenues is \(\$ 70.1\) million. Calculate the Debt Service/total revenue ratio. Is the ratio favorable?

Study smarter with the SolutionInn App


