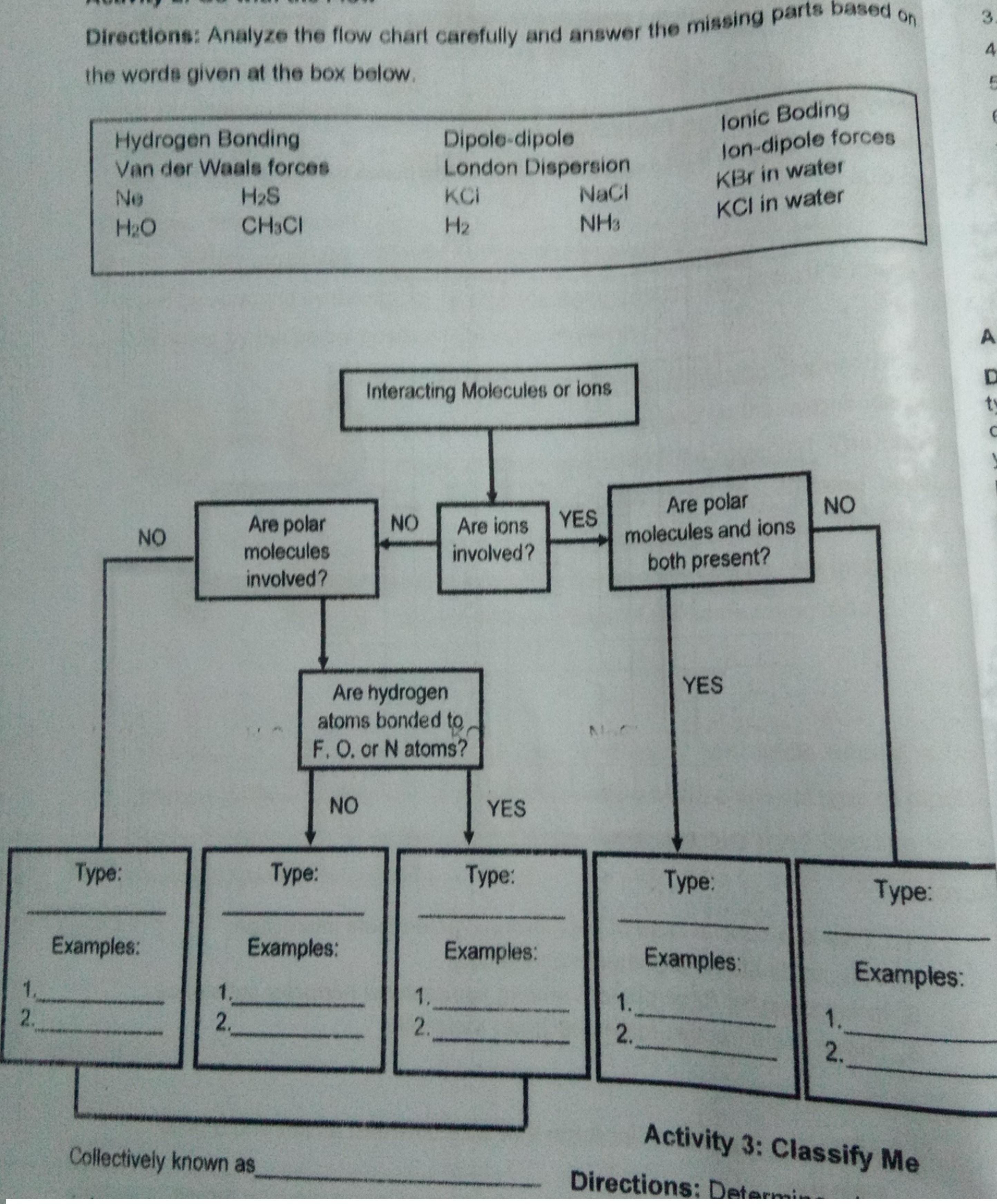
Directions: Analyze the flow chart carefully and answer the missing parts based on the words given...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Directions: Analyze the flow chart carefully and answer the missing parts based on the words given at the box below. 4. Hydrogen Bonding Van der Waals forces H2S Dipole-dipole London Dispersion NaCl lonic Boding lon-dipole forces KBr in water KCI in water Ne KCI H2O CH3CI H2 NH3 Interacting Molecules or ions tv Are polar NO Are polar molecules Are ions YES involved? NO NO molecules and ions both present? involved? YES Are hydrogen atoms bonded to, F. O. or N atoms? NO YES Туре: Турeе: Type: Туpе: Турeе: Examples: Examples: Examples: Examples: Examples: 1. 2. 1. 1. 1. 2. 2. 2. 2. Activity 3: Classify Me Collectively known as Directions: Determi Directions: Analyze the flow chart carefully and answer the missing parts based on the words given at the box below. 4. Hydrogen Bonding Van der Waals forces H2S Dipole-dipole London Dispersion NaCl lonic Boding lon-dipole forces KBr in water KCI in water Ne KCI H2O CH3CI H2 NH3 Interacting Molecules or ions tv Are polar NO Are polar molecules Are ions YES involved? NO NO molecules and ions both present? involved? YES Are hydrogen atoms bonded to, F. O. or N atoms? NO YES Туре: Турeе: Type: Туpе: Турeе: Examples: Examples: Examples: Examples: Examples: 1. 2. 1. 1. 1. 2. 2. 2. 2. Activity 3: Classify Me Collectively known as Directions: Determi
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Van der Waals forces between atoms and between molecules arise from a. uniform charge distributions b. nonuniform charge distributions c. electron transfer d. electron sharing
-
Van der Waals forces are strong enough to hold inert gas atoms together to form liquids at low temperatures, but these forces do not lead to inert gas molecules at higher temperatures. Why not?
-
Van der Waals Equation and Critical Points (a) In p V- diagrams the slope p/V along an isotherm is never positive. Explain why. (b) Regions where p/V = 0 represent equilibrium between two phases;...
-
dentify a true statement about penetration pricing. It emphasizes a company's current performance but can sacrifice long-term performance. It requires a company to price goods to cover variable costs...
-
The Falcon Corporation has $68,000 in taxable income. Its accountant uncovered $87,000 in net positive adjustments and $2,000 of preference items in determining its alternative minimum taxable...
-
Compute the velocity of light in calcium fluoride (CaF2), which has a dielectric constant r of 2.056 (at frequencies within the visible range) and a magnetic susceptibility of -1.43 x 10-5.
-
You want to retire in 30 years. You are starting to invest in a growth income fund that promises an ambitious rate of $15 \%$. You can put in $\$ 200$ per month. How much will you have in 30 years?
-
In Section 5.5, we showed the following two-person, zero-sum game had a mixed strategy: a. Use dominance to reduce the game to a 2 2 game. Which strategies are dominated? b. Determine the optimal...
-
Image transcription text v X aXcelerate - Learner + X - C A entryeducation.app.axcelerate.com/learner/course/class/13950840/mod/19654/assessment/14117890/run Q Item List All BSBTEC301 - Design and...
-
The following trial balance was extracted from the books of Alice Ltd, a wine merchant, at the close of business on 31 December 2020 Trial Balance Capital Purchases Sales Revenue Trade payable Trade...
-
A survey of 1,085 adults asked, Do you enjoy shopping for clothing for yourself? The results indicated that 51% of the females enjoyed shopping for clothing for themselves as compared to 44% of the...
-
Crain Company has a manufacturing subsidiary in Singapore that produces high-end exercise equipment for U.S. consumers. The manufacturing subsidiary has total manufacturing costs of $1,460,000, plus...
-
Creative Computing sells a tablet computer called the Protab. The $740 sales price of a Protab Package includes the following: One Protab computer. A six-month limited warranty. This warranty...
-
Prepared from a situation suggested by Professor John W. Hardy.) Lone Star Meat Packers is a major processor of beef and other meat products. The company has a large amount of T-bone steak on hand,...
-
Ziegler Inc. has decided to use the high-low method to estimate the total cost and the fixed and variable cost components of the total cost. The data for various levels of production are as follows:...
-
Yale Company manufactures hair brushes that sell at wholesale for $3 per unit. The company had no beginning inventory in the prior year. These data summarize the current and prior year operations:...
-
Research the following theories that may influence criminal behavior: social structure theories, social process theories, and social conflict theories. Describe each of these theories and the...
-
Interview managers at three companies in your area about their use of ERP. How have their experiences been similar? What accounts for the similarities and differences?
-
The density of freshwater is 1.00 g/cm 3 and that of seawater is 1.03 g/cm 3 . A ship will float a. higher in freshwater than in seawater b. lower in freshwater than in seawater c. at the same level...
-
Rocks that have been altered by heat and pressure beneath the earths surface are called a. igneous rocks b. sedimentary rocks c. metamorphic rocks d. precipitated rocks
-
Asteroids are found a. Spread evenly throughout the solar system b. Mainly in a belt between the earth and the moon c. Mainly in a belt between the earth and Mars d. Mainly in a belt between Mars and...
-
Initially, both legs of a mercury manometer are open to the atmosphere, and the mercury height in the left leg is \(40.0 \mathrm{~mm}\). You inflate a balloon until it has a surface area of \(0.300...
-
At an atmospheric pressure of \(1.00 \mathrm{~atm}\), the height of the liquid in the tube of a mercury barometer is \(760 \mathrm{~mm}\) only if you ignore capillary rise in the tube. Obtain an...
-
A hydrogen-filled balloon is used to lift a \(125-\mathrm{kg}\) stone off the ground. The basket holding the stone has a mass of \(15.0 \mathrm{~kg}\). What must the minimum radius \(R\) of the...

Study smarter with the SolutionInn App


