Disinfection is one of the major steps in the drinking water treatment process. Depending on the...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
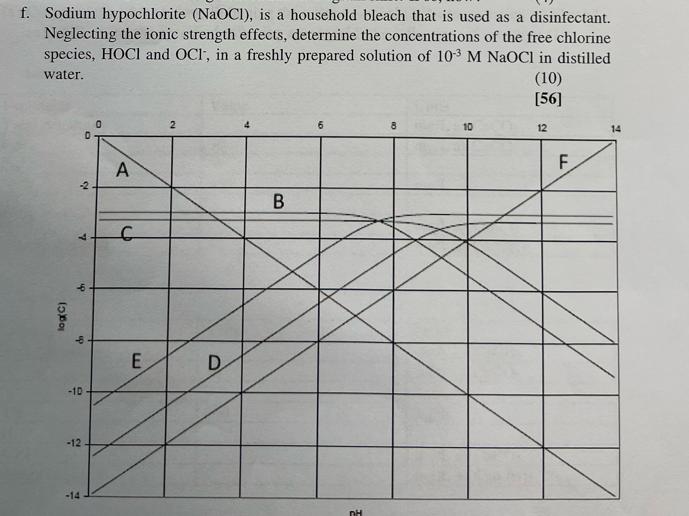
Disinfection is one of the major steps in the drinking water treatment process. Depending on the source of water being treated, there is high likelihood of producing disinfection by- products (DBP) when chlorine is used as disinfectant. a. Describe how the formation of disinfection by products takes place, discuss which water sources are more likely to present this problem and propose methods that can be used to mitigate DBP formation. (10) b. Chlorine dioxide has been proposed as a possible solution in the prevention of DBP formation. Discuss mechanism by which chlorine dioxide imparts disinfection in water. Briefly discuss the advantages and disadvantages associated with the use of chlorine dioxide. NB. You are encouraged to use and cite journal articles for this question. (10) c. In order to obtain a free residual of chlorine of 0.5 mg/L, a dose of 10 mg/L is added to a 36400 m/day water treatment plant. NB It is common practice to apply Cl:NH3 dosage ratios of 3:1 (i) Calculate the amount of Cl2 and NH3 required to achieve this. (8) (ii) Explain why it is sometimes necessary to add ammonia during the disinfection process and what is the reasoning behind using a 3:1 Cl2:NH3 dosage ratio (6) Chlorine usage in a 20 000 m/day treatment plant is 8 kg. The residual after 10 min contact time is 0.15 mg/L. What is the chlorine demand of the water? (4) d. e. The graph below shows the log-concentration diagram of a system containing 5 x 104 M NH3 and an unknown amount of Sodium hypochlorite. Determine. i. The unknown species A to F ii. The sodium concentration iii. iv. (6) (2) The pH (2) In a similar system, the NH3 is substituted with 2.5 x10-4 M NH4C1 and 2.5 x104 M NH3 Will the log-concentration diagram shift? If so, how? (4) f. Sodium hypochlorite (NaOCI), is a household bleach that is used as a disinfectant. Neglecting the ionic strength effects, determine the concentrations of the free chlorine species, HOCI and OCI, in a freshly prepared solution of 10 M NaOCl in distilled water. log(C) 7 -2 ch ip D -10- -12 -14 0 A C E 2 D B 6 nH 8 10 (10) [56] 12 F. 14 Disinfection is one of the major steps in the drinking water treatment process. Depending on the source of water being treated, there is high likelihood of producing disinfection by- products (DBP) when chlorine is used as disinfectant. a. Describe how the formation of disinfection by products takes place, discuss which water sources are more likely to present this problem and propose methods that can be used to mitigate DBP formation. (10) b. Chlorine dioxide has been proposed as a possible solution in the prevention of DBP formation. Discuss mechanism by which chlorine dioxide imparts disinfection in water. Briefly discuss the advantages and disadvantages associated with the use of chlorine dioxide. NB. You are encouraged to use and cite journal articles for this question. (10) c. In order to obtain a free residual of chlorine of 0.5 mg/L, a dose of 10 mg/L is added to a 36400 m/day water treatment plant. NB It is common practice to apply Cl:NH3 dosage ratios of 3:1 (i) Calculate the amount of Cl2 and NH3 required to achieve this. (8) (ii) Explain why it is sometimes necessary to add ammonia during the disinfection process and what is the reasoning behind using a 3:1 Cl2:NH3 dosage ratio (6) Chlorine usage in a 20 000 m/day treatment plant is 8 kg. The residual after 10 min contact time is 0.15 mg/L. What is the chlorine demand of the water? (4) d. e. The graph below shows the log-concentration diagram of a system containing 5 x 104 M NH3 and an unknown amount of Sodium hypochlorite. Determine. i. The unknown species A to F ii. The sodium concentration iii. iv. (6) (2) The pH (2) In a similar system, the NH3 is substituted with 2.5 x10-4 M NH4C1 and 2.5 x104 M NH3 Will the log-concentration diagram shift? If so, how? (4) f. Sodium hypochlorite (NaOCI), is a household bleach that is used as a disinfectant. Neglecting the ionic strength effects, determine the concentrations of the free chlorine species, HOCI and OCI, in a freshly prepared solution of 10 M NaOCl in distilled water. log(C) 7 -2 ch ip D -10- -12 -14 0 A C E 2 D B 6 nH 8 10 (10) [56] 12 F. 14
Expert Answer:

Related Book For 

International Marketing And Export Management
ISBN: 9781292016924
8th Edition
Authors: Gerald Albaum , Alexander Josiassen , Edwin Duerr
Posted Date:
Students also viewed these chemical engineering questions
-
A client wishes to form a corporation in Georgia. What ele- ments must be included in the articles of incorporation filed with the state? Lisa a Minnesota resident is makin
-
The Crazy Eddie fraud may appear smaller and gentler than the massive billion-dollar frauds exposed in recent times, such as Bernie Madoffs Ponzi scheme, frauds in the subprime mortgage market, the...
-
Let A, B be sets. Define: (a) the Cartesian product (A B) (b) the set of relations R between A and B (c) the identity relation A on the set A [3 marks] Suppose S, T are relations between A and B, and...
-
Using the following information, determine the activity rate for the quality inspections activity. Budgeted Activity Cost Pool Activity Setups Estimated Activity Base 1,000 $60,000 Purchase orders...
-
If rent controls are so counterproductive, why do cities impose them? How else might the housing problems of poor people be solved?
-
At night the pupils of a certain womans eyes are 8 mm in diameter. (a) How many kilometers away from a car facing her will the woman be able to distinguish its headlights from each other? (b) What...
-
Redo the exercise above, assuming in the first case that the Faurecia share rises to 40 or falls to 25. What is the impact on the value of the option? What basic feature of the option have you...
-
Marathon, Inc., manufactures two types of shoes: X-Trainer and Court. Last year, Marathon had the following costs and revenues: Marathon, Inc., currently uses labor costs to allocate all overhead,...
-
Part A Sim Lim Company has the following closing balances for the year ended December 30, 2020. Direct materials $15,000 Dr Work-in-Process $34,500 Dr Finished Goods $49,500 Dr Cost of Goods Sold...
-
Your parents are considering investing in Apple Inc. common stock. They ask you, as an accounting expert, to make an analysis of the company for them. Financial statements of Apple are available...
-
Given an acceleration vector, initial velocity (40, Yo), and initial position (X),Yo), find the velocity and position vectors for t 0. a(t) = (cost, 3 sint), (u,v) = (0,5), (x,y) = (1,0) What is the...
-
Why is it necessary to use two subscripts, \(i\) and \(t\), to describe panel data? What does \(i\) refer to? What does \(t\) refer to?
-
During the Great Recession, many news stories focused on a rising number of discouraged workers. The implication of many of these stories is that the unemployment situation was worse than indicated...
-
Cindy gains utility from consumption C and leisure L. The most leisure she can consume in any given week is 110 hours. Her utility function is U(C, L) = C L. This functional form implies that Cindys...
-
Explain the difference between \(\hat{\beta}_{1}\) and \(\beta_{1}\); between the residual \(\hat{u}_{i}\) and the regression error \(u_{i}\); and between the OLS predicted value \(\hat{Y}_{i}\) and...
-
Suppose the economys labor market is competitive and that labor demand can be written as w = 50 0.3E while labor supply can be written as w = 8 + 0.2E where E is the total amount of employment in...
-
Discussion on Aims and principals of health promotion , protections and improvement and how this can benefit to community to improve health , physical, and Behavioural Chosen topic for this is child...
-
Four GWU students have been selected to taste food sold by 3 different food trucks labeled as food truck A, B and C on H & 22nd Streets every Monday for 3-weeks. For each student, food trucks are...
-
Introduction In 2010, New United Motor Manufacturing, Inc., a unique and highly successful joint venture between Americas General Motors and Japans Toyota, went out of business. NUMMI, as the...
-
Explain when the export firm should no longer be concerned with the pricing of its products.
-
Discuss the importance of using ecologically correct packaging materials and the major impact of packaging policy. Is eco-labeling necessary? Why?
-
When calculating WACC, if we wanted to abide by common practice, we would: a. Always use nominal rates b. Always use effective rates c. Use nominal rates on bonds only d. Use nominal rates on stock...
-
Given the following information, what is WBM Corporation's WACC? Common Stock: 1 million shares outstanding, \($40\) per share, \($1\) par value, beta = 1.3 Bonds: 10,000 bonds outstanding,...
-
Assuming that a firm uses at least some debt, which of the following expressions must be correct? a. Rx(1-T)>R> WACC b. WACC>R>RX (1-T) c. R>WACC>R, x(1-T) d. R,x(1-T)> WACC>R E

Study smarter with the SolutionInn App


