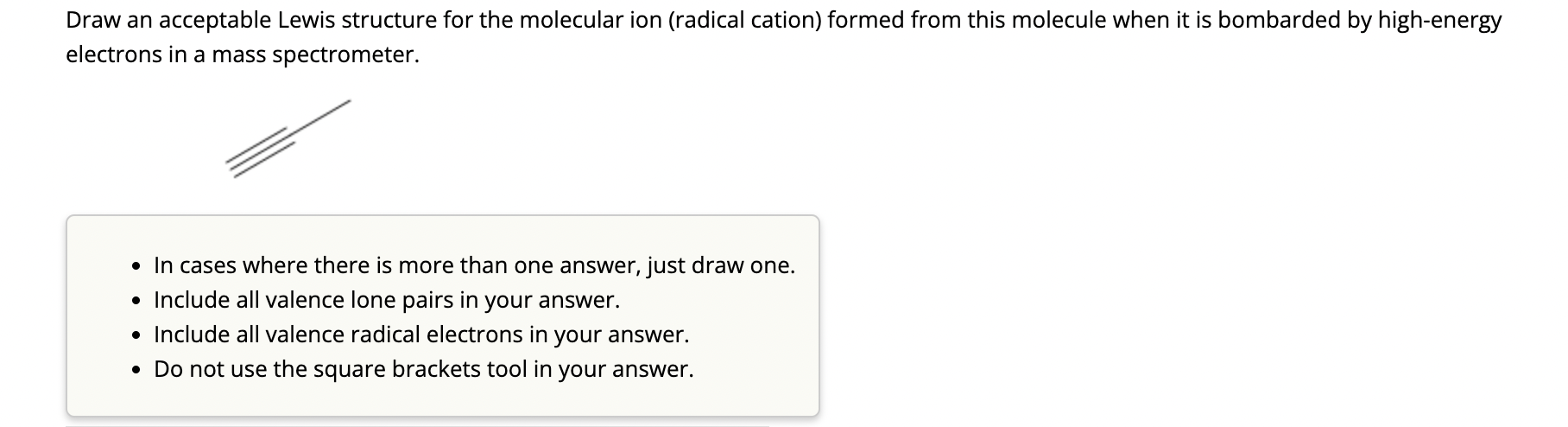
Draw an acceptable Lewis structure for the molecular ion (radical cation) formed from this molecule when...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Draw an acceptable Lewis structure for the molecular ion (radical cation) formed from this molecule when it is bombarded by high-energy electrons in a mass spectrometer. In cases where there is more than one answer, just draw one. Include all valence lone pairs in your answer. Include all valence radical electrons in your answer. Do not use the square brackets tool in your answer. Draw an acceptable Lewis structure for the molecular ion (radical cation) formed from this molecule when it is bombarded by high-energy electrons in a mass spectrometer. In cases where there is more than one answer, just draw one. Include all valence lone pairs in your answer. Include all valence radical electrons in your answer. Do not use the square brackets tool in your answer.
Expert Answer:
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemical engineering questions
-
ABC Manufactures is a manufacturing company, and your audit team is conducting the annual audit of its financial statements. As part of the audit procedures, you are tasked with testing the accounts...
-
we 7 Four friends, Tim, Thabo, Tracy and Thandi, each received a lump sum of R12 000. Tim invested his money in shares for a new company called Newco, Tracy invested her money in shares for a company...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
b) Salt enhances bioavailability. Taking a weak acid as an example, explain how salt in gastric fluid has better absorption compared to its acid form.
-
Show how each of the following compounds reacts with HO-: a. CH3OH b. +NH4 d. BF3 e. +CH3 f. FeBr3 g. AlCl3 h. CH3COOH CH3NH3
-
Which of the following is not possible? A An acute scalene triangle C An obtuse isosceles triangle B A triangle with two acute exterior angles D An equiangular acute triangle
-
Fayols principle of esprit de corps refers to: A. Subordination of individual interest to the common good. B. Development of a high level of employee morale. C. Encouragement of initiative and risk...
-
At the end of its first year, the trial balance of Jules Company shows Equipment $22,000 and zero balances in Accumulated DepreciationEquipment and Depreciation Expense. Depreciation for the year is...
-
can you explain how they got the answer for this WORKSHEET EXAMPLE-Fall 2023 The following information was taken from Dodgers Co's general ledger at 12/31/23. The balances are before any...
-
A manufacturer of prefabricated homes has decided to subcontract four components of the homes. Several companies are interested in receiving this business, but none can handle more than one...
-
Oneida Company's operations began in August. August sales were $190,000 and purchases were $105,000. The beginning cash balance for September is $30,500. Oneida's owner approaches the bank for a...
-
Perry Company employs a job-order costing system. Only three jobs-Job #205, Job #206, and Job #207-were worked on during January and February. Job #205 was completed February 10; the other two jobs...
-
Odilon Company makes a household appliance with model number X500. The goal for 2019 is to reduce direct materials usage per unit. No defective units are currently produced. Manufacturing conversion...
-
What are the potential benefits and risks of investing in securities? 13. How are securities priced in the market and what factors influence their value? 14. Are there any specific rules and...
-
The business-to-business strategic marketing planning process In business-to-business marketing, as in all other types of organisations, the overall business strategy will guide the development of...
-
Tom is evaluating a project that costs $3,500,000, has a five-year life, and has no salvage value. Assume that depreciation is straight-line to zero over the life of the project. Sales are projected...
-
Apex Corporation's economic unit estimates that the probability of a good business environment next year is equal to the probability of a bad environment. Knowing that, the managers of Apex must...
-
Cornell and Roberts are partners who agree to admit Stanley to their partnership. Cornell has a capital balance of $80,000 and Roberts has a capital balance of $120,000. Cornell and Roberts share net...
-
An organic compound was found to contain only C, H, and Cl. When a 1.50-g sample of the compound was completely combusted in air, 3.52 g of CO2 was formed. In a separate experiment the chlorine in a...
-
For the majority of the compounds listed in Appendix C, the value of Gf is more positive (or less negative) than the value of Gf. (a) Explain this observation, using NH3(g), CCl4(l), and KNO3(s) as...
-
(a) Write an equation that expresses the first law of thermodynamics in terms of heat and work. (b) Under what conditions will the quantities q and w be negative numbers?
-
What happens to the oligopolists profits if entry is easy?
-
What is price leadership?
-
In an oligopoly such as the U.S. domestic airline industry, a firm such as United Airlines would a. carefully anticipate Delta, JetBlue, and Southwests likely responses before it raised or lowered...

Study smarter with the SolutionInn App



