Draw an alternative Lewis structure for CH2Br. I can't seem toget this right :( Determine the formal
Fantastic news! We've Found the answer you've been seeking!
Question:
Draw an alternative Lewis structure for CH2Br. I can't seem toget this right :(
Transcribed Image Text:
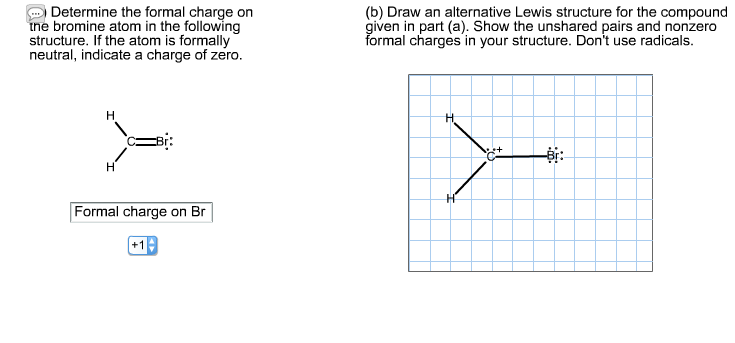
Determine the formal charge on the bromine atom in the following structure. If the atom is formally neutral, indicate a charge of zero. H - H Formal charge on Br +19 (b) Draw an alternative Lewis structure for the compound given in part (a). Show the unshared pairs and nonzero formal charges in your structure. Don't use radicals. H H C+ Determine the formal charge on the bromine atom in the following structure. If the atom is formally neutral, indicate a charge of zero. H - H Formal charge on Br +19 (b) Draw an alternative Lewis structure for the compound given in part (a). Show the unshared pairs and nonzero formal charges in your structure. Don't use radicals. H H C+
Expert Answer:
Answer rating: 100% (QA)
Bromine has 7 valence electrons It forms 2 bonds with its adjacent atoms Hence the number of b... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Draw a Lewis structure for the N, N-dimethyl formamide molecule. The skeletal structure is Various types of evidence lead to the conclusion that there is some double- bond character to one of the CN...
-
Draw the Lewis structure for the ligand shown in the next column. Which atoms can serve as donor atoms? Classify this ligand as monodentate, bidentate, or tridentate. How many of these ligands are...
-
Draw the Lewis structure for the iodine dioxide difluoride ion, IO2F2-. Which statements are correct regarding its Lewis structure? (Note, all the atoms are bonded to the central I atom.)Steric...
-
A geostationary satellite orbiting at a height 36,000km operates at 4GHz using a transmitter antenna that has a gain of 15dB. On earth the receiver antenna has a gain of 45dB. Calculate; (a) Free...
-
Would PPP be more likely to hold between the United States and Hungary if trade barriers were completely removed and if Hungarys currency were allowed to float without any government intervention?...
-
Pascale Corp. has the following securities (all purchased in 2014) in its investment ponfolio on December 31, 2014: (1) 2,500 Anderson Corp. common shares, which cost $48,750 (2) 10,000 Munter Ltd....
-
At December 31, 2019, the portfolio of investments in equity securities measured at FV-NI held by Athletes Inc. is as follows. Investment Security Badger Common Stock (1,000 shares) Spartan Common...
-
Each of the following situations involves possible violations of the AICPA's Code of Professional Conduct. For each situation, state whether it is a violation of the Code. In those cases in which it...
-
Arnell Industries has just issued 20 million in debt? (at par).The firm will pay interest only on this debt.? Arnell's marginaltax rate is expected to be 21% for the foreseeable future. a.Suppo 2...
-
John and Ellen Brire are married and file a joint return. They have no dependents. John owns an unincorporated specialty electrical lighting retail store, Brite-On. Brite-On had the following assets...
-
3.5 - 2 HP 1 1/2 HP 3 HP 1 HP- 3/4 HP 1/2 HP 1/3 HP 2.0 Static Pressure in inches W.G. 1.5 1.0 0.5 NOT SELECT TO THE LEFT OF THIS SYSTEM CURVE RPM Density 0.075 lb/ft BHP ---- -5 HP 1200 680 1070 940...
-
An ant, crazed by the Sun on a hot Texas afternoon, darts over an xy plane scratched in the dirt. The x and y components of four consecutive darts are the following, all in centimeters: (27.0, 37.0),...
-
K In each of the following geometric sequences, find (i) the 7th term; (ii) the nth term. 1 4 16 a. 3 9 27 b. 5, 5, 55 C. 1 4 9 3 9 - 16 27
-
The combined electrical resistance R of two resistors R, and R, connected in parallel, is given by the equation below, where R, R, and R are measured in ohms. R and R are increasing at rates of 0.7...
-
If f(x)=(8x3+9) and g(x) = -(9x + 19), then d dx [(2)-9(2)] -0
-
If f(x)=6x+7 and g(x) = 8x + 15, then d [f(x) dx g(x) 10
-
Reply individually to Whitney and Jennie for DB Reply to Whitney Recent studies have examined adolescents and their social media use. The goal of the study was to find out if the three subgroups...
-
Find the equation of the plane passing through the points P 5,4,3 ,Q 4,3,1 and R 1,5,4
-
Deduce the structure of each compound from the information given. All unknowns in this problem have molecular formula C8H12. (a) Upon catalytic hydrogenation, unknown W gives cyclooctane. Ozonolysis...
-
Write an equation for the reaction of vitamin E with an oxidizing radical (RO) to give ROH and a less reactive free radical.
-
When dichloromethane is treated with strong NaOH, an intermediate is generated that reacts like a carbene. Draw the structure of this reactive intermediate, and propose a mechanism for its formation.
-
2. YOU BE THE JUDGE WRITING PROBLEM An off-duty, out-of-uniform police officer and his son purchased some food from a 7-Eleven store and were still in the parking lot when a carload of teenagers...
-
3. You sign a two-year lease with a landlord for an apartment. The rent will be $1,000 per month. A clause in the lease requires payment on the first of every month. The clause states that the...
-
4. Federal antitrust statutes are complex, but the basic goal is straightforward: to prevent a major industry from being so dominated by a small group of corporations that they destroy competition...

Study smarter with the SolutionInn App


