Draw the Lewis structure of SF4 showing all lone pairs. What is the hybridization of the...
Fantastic news! We've Found the answer you've been seeking!
Question:

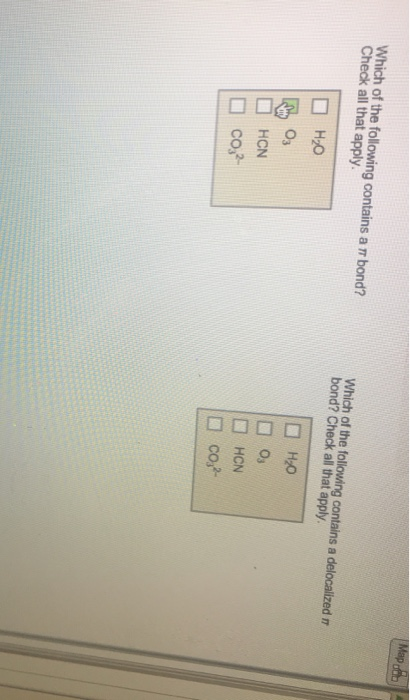
Transcribed Image Text:
Draw the Lewis structure of SF4 showing all lone pairs. What is the hybridization of the central atom? sp sp³d sp³d An SF4 molecule is polar. nonpolar. Identify the molecular geometry of SF4. square pyramidal linear bent tetrahedral octahedral see-saw T-shaped trigonal pyramidal square planar trigonal planar trigonal bipyramidal What are the ideal bond angles of this geometry? Choose all that apply. 90 degrees 109.5 degrees 120 degrees 180 degrees yious Give Up & View Solulion Check Answer Next Exit Which of the following contains a r bond? Check all that apply. H₂O 03 HCN co²- Which of the following contains a delocalized n bond? Check all that apply. H₂O 0₁ HCN co²- Map 000 Draw the Lewis structure of SF4 showing all lone pairs. What is the hybridization of the central atom? sp sp³d sp³d An SF4 molecule is polar. nonpolar. Identify the molecular geometry of SF4. square pyramidal linear bent tetrahedral octahedral see-saw T-shaped trigonal pyramidal square planar trigonal planar trigonal bipyramidal What are the ideal bond angles of this geometry? Choose all that apply. 90 degrees 109.5 degrees 120 degrees 180 degrees yious Give Up & View Solulion Check Answer Next Exit Which of the following contains a r bond? Check all that apply. H₂O 03 HCN co²- Which of the following contains a delocalized n bond? Check all that apply. H₂O 0₁ HCN co²- Map 000
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
What is the hybridization of the central atom in (a) SiCl4 (b) HCN (c) SO3 (d) TeCl2.
-
Draw the Lewis structure of O2F2. Assign oxidation states and formal charges to the atoms in O2F2. The com-pound O2F2 is a vigorous and potent oxidizing and fluo-rinating agent. Are oxidation states...
-
Draw the lewis structure and identify the three molecular geometry for CH3SO2H, C(CH2)2 and CH3NCO
-
Below is the comparative income statement of Variline, Inc. Requirements 1. Prepare a horizontal analysis of the comparative income statement of Variline, Inc. Round percentage changes to the nearest...
-
Arch Corporation manufactures specialty lines of women's apparel. During February, the company worked on three special orders: A-25, A-27, and B-14. Cost and production data for each order are as...
-
For each of the following, evaluate E f dV. a) f(x, y) = (1 + x2)-1 and E is bounded by x = 1, y = 0, and y = x3. b) f(x, y) = x + y and E is the triangle with vertices (0, 0), (0, 1), and (2, 0). c)...
-
When was the Sarbanes-Oxley Act signed into law by President George W. Bush?
-
Rocky Mountain Corporation makes two types of hiking bootsXactive and the Pathbreaker. Data concerning these two product lines appear below: The company has a traditional costing system in which...
-
Nova Inc. has announced a rights offer that will take 6 rights to buy a new share in the offering at a subscription price of $25. The current price per share is $39. Assume that immediately...
-
David R. and Ella M. Cole (ages 39 and 38, respectively) are husband and wife who live at 1820 Elk Avenue, Denver, CO 80202. David is a self-employed consultant, specializing in retail management and...
-
Kenneth Company uses activity-based costing. It budgets $1,650,000 of overhead cost to sustainably dispose of 6,600 tons of hazardous waste. The company disposed of 10 tons of hazardous waste in...
-
Ski West, Incorporated, operates a downhill ski area near Lake Tahoe, California. An all-day adult lift ticket can be purchased for $85. Adult customers also can purchase a season pass that entitles...
-
The axis of rotation of an object passes perpendicularly through its center. What is the approximate length of the lever arm if a force is applied at a location 4 meters directly above the center of...
-
A metal rod of length 29 cm is placed in a magnetic field of strength 2.5 T, oriented perpendicular to the field. 33%Part (a) Determine the induced emf, in volts, between the ends of the rod when the...
-
A moving company needs to lift a 700 lb. (320kg) piano to the top floor of an apartment building. They set up a rope and pulley system on the balcony of the upper story apartment, and pull the piano...
-
Zimmerman Company manufactures two products, Board 12 and Case 165. Zimmerman's overhead costs consist of setting up machines, $2,400,000; machining. $5,400,000; and inspecting. $1,800,000....
-
. What are the risks and rewards of the contract management ? How can the team balance these risks and rewards?
-
Catherine (aged 42) and Johnson (aged 45) have been married for 12 years. Johnson is a project manager of an event company at a monthly salary of $55,000 with an additional one-month salary of...
-
The heat of fusion of water is 6.008 10 3 J mol 1 at its normal melting point of 273.15 K. Calculate the freezing point depression constant K f .
-
Use the logic diagram of Figure 27.2 to determine the point group for PCl 5 . Indicate your decision-making process as was done in the text for NH 3 . a. linear? b. C n axis? c. more than 1C n axis?...
-
Consider the Daniell cell, for which the overall cell reaction is Zn(s) + Cu 2+ (aq) Zn 2+ (aq) + Cu(s). The concentrations of CuSO 4 and ZnSO 4 are 2.50 10 3 and 1.10 10 3 m, respectively. a....
-
The following information has been extracted from the draft financial information of V Ltd: Dividend paid = 52,000 (a) You are required to prepare a cash flow statement for V Ltd for the year ended...
-
Write the following in standard form. a. \((4+5 i)(2-3 i)\). b. \((1+i)^{3}\). c. \(\frac{5+3 i}{1-i}\).
-
Write the following in polar form, \(z=r e^{i \theta}\). a. \(i-1\). b. \(-2 i\). c. \(\sqrt{3}+3 i\).

Study smarter with the SolutionInn App


