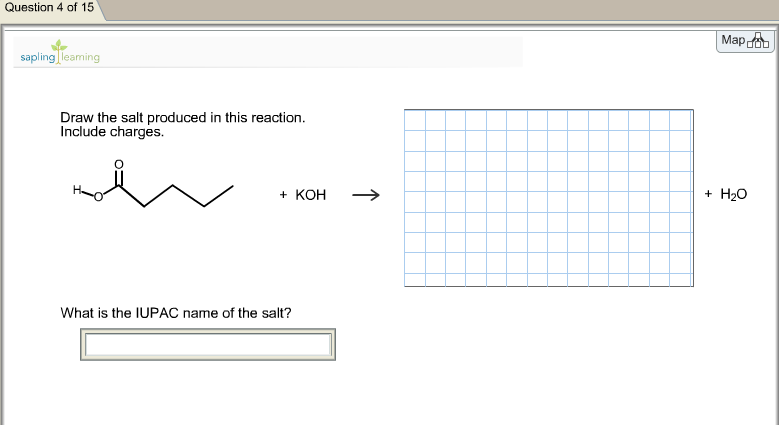
Draw the salt produced in this reaction. Include charges. Whatis the IUPAC name of the salt? Question
Fantastic news! We've Found the answer you've been seeking!
Question:
Draw the salt produced in this reaction. Include charges. Whatis the IUPAC name of the salt?
Transcribed Image Text:
Question 4 of 15 sapling] leaming Draw the salt produced in this reaction. Include charges. + KOH What is the IUPAC name of the salt? Map + H₂O Question 4 of 15 sapling] leaming Draw the salt produced in this reaction. Include charges. + KOH What is the IUPAC name of the salt? Map + H₂O Question 4 of 15 sapling] leaming Draw the salt produced in this reaction. Include charges. + KOH What is the IUPAC name of the salt? Map + H₂O Question 4 of 15 sapling] leaming Draw the salt produced in this reaction. Include charges. + KOH What is the IUPAC name of the salt? Map + H₂O Question 4 of 15 sapling] leaming Draw the salt produced in this reaction. Include charges. + KOH What is the IUPAC name of the salt? Map + H₂O Question 4 of 15 sapling] leaming Draw the salt produced in this reaction. Include charges. + KOH What is the IUPAC name of the salt? Map + H₂O Question 4 of 15 sapling] leaming Draw the salt produced in this reaction. Include charges. + KOH What is the IUPAC name of the salt? Map + H₂O Question 4 of 15 sapling] leaming Draw the salt produced in this reaction. Include charges. + KOH What is the IUPAC name of the salt? Map + H₂O
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Name the following molecules according to the IUPAC nomenclature system. (a) (b) (c) (d) (e) (f) CH32 Cl CH3 Br
-
Name the following compounds by IUPAC rules: a. b. H-C CH,CH-CH
-
The IUPAC name for a carboxylic acid is based on the name of the hydrocarbon with the same number of carbon atoms. The ending -oic is appended, as in ethanoic acid, which is the IUPAC name for acetic...
-
A machine was sold in December 20x3 for $13,000. It was purchased in January 20x1 for $19,000, and depreciation of $16,000 was recorded from the date of purchase through the date of disposal....
-
Data for Communication Accessories is provided in P11-4C. In P11-4C, the income statement, balance sheets, and additional information for Communication Accessories are provided. Required: Prepare the...
-
Horvath Company uses an imprest petty cash system. The fund was established on March 1 with a balance of $100. During March, the following petty cash receipts were found in the petty cash box. The...
-
This appeal involves the validity of a will executed in contravention of an earlier contract to make mutual wills. A husband and wife signed a contract to make mutual wills and then executed those...
-
On January 2, 2013, Benson Hospital purchased a $100,000 special radiology scanner from Picard Inc. The scanner had a useful life of 4 years and was estimated to have no disposal value at the end of...
-
1. Mr. i holds the following preferences over outcomes w, x, y, and z: xPw, xPy, zPx, yPz, wPy, and wPz. (a) Identify all five possible subsets of these outcomes that contain three or more outcomes....
-
The case study shown below provides an overview of Road Transit Systems (RTS) - a specialist preventative vehicle maintenance program operator in NSW, Australia. A number of attributes have been...
-
The wheel shown in (Figure 1) is subjected to a force of P = 16 kN from the axle. Assume the rim is rigid and the spokes are made of the same material, and each has the same cross-sectional area....
-
Air conditioning for a college dormitory will cost $2.1 million to install and $170,000 per year to operate. The system should last 19 years. The real cost of capital is 9%, and the college pays no...
-
A project has an initial cost of $41,025, expected net cash inflows of $9,000 per year for 7 years, and a cost of capital of13%. What is the project's MIRR? Do not round intermediatecalculations....
-
How do microbial symbionts influence the evolution and ecological success of their host organisms, and what implications does this have for understanding microbial diversity and ecosystem functioning?
-
During the year, Kaplan Pharma increased its accounts receivable by $200, decreased its inventory by $100, and decreased its accounts payable by $110. How did these three accounts affect the sources...
-
4 . What is the duration of a four-year, $1,500 bond that pays a coupon (annual) of 12% that trades at a yield of 16%. Calculate is the expected change in the bonds price if interest rates fall by...
-
Ron Abrams, VP Operations for Wilson Bros. has come wandering into your office muttering under his breath (clearly exasperated) after reading the financial statements for one of the plants in Western...
-
Explain the Hawthorne effect.
-
Use the data in the table below to answer the questions which follow. Element ............ Ar H ......................... 1 C ........................ 12 N ........................ 14 O...
-
a. State which of the following processes is endothermic and which is exothermic. (i) The breaking of a chemical bond. (ii) The forming of a chemical bond. b. The table below shows the bond energy...
-
The iron obtained from the blast furnace is known as pig or cast iron. Because of the presence of impurities, such as carbon, it has a hard and brittle nature. Most of this type of iron is therefore...
-
Air enclosed in a cylinder has density \(ho=1.4 \mathrm{~kg} / \mathrm{m}^{3}\). a. What will be the density of the air if the length of the cylinder is doubled while the radius is unchanged? b. What...
-
Given the data of the graph, approximately how much energy, in Calories, would a \(200 \mathrm{~g}\) rat use during the course of a day? A. 10 B. 20 C. 100 D. 200
-
The passage proposes that there are quantitative "laws" of biology that have their basis in physical principles, using the scaling of specific metabolic rate with body mass as an example. Which of...

Study smarter with the SolutionInn App



