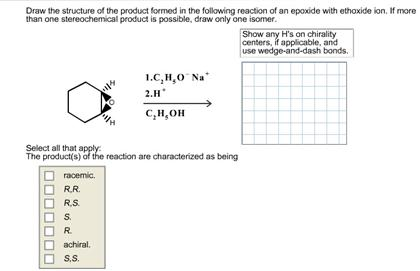
Draw the structure of the product formed in the following reaction of an epoxide with ethoxide...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Draw the structure of the product formed in the following reaction of an epoxide with ethoxide ion. If more than one stereochemical product is possible, draw only one isomer. S. R. 1.C,H,O Na 2.H The product(s) of the reaction are characterized as being racemic. R.R. R.S. achiral. S.S. C,H,OH Show any H's on chirality centers, if applicable, and use wedge-and-dash bonds. Draw the structure of the product formed in the following reaction of an epoxide with ethoxide ion. If more than one stereochemical product is possible, draw only one isomer. S. R. 1.C,H,O Na 2.H The product(s) of the reaction are characterized as being racemic. R.R. R.S. achiral. S.S. C,H,OH Show any H's on chirality centers, if applicable, and use wedge-and-dash bonds.
Expert Answer:
Answer rating: 100% (QA)
Explanation In the first step ethoxide ion act as a strong base and result in the epoxide ring openi... View the full answer

Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Posted Date:
Students also viewed these chemistry questions
-
Draw the structure of the product with molecular formula C 10 H 10 O that is obtained when the compound below is heated with aqueous acid. CN CN C10H100 Heat
-
Predict the structure of the product formed in the reaction of the organic base pyridine with the organic acid acetic acid, and use curved arrows to indicate the direction of electronflow. Pyridine...
-
Draw the structure of each possible dichloride that can be used to prepare the following alkyne via elimination:
-
Find the laurents series f(z)= 1/(z-1)(z-2) (i) |z| <1 (ii) 1 2
-
John Doe had a checking account at Highland National Bank in New York. Two days after John Doe died in Florida, but before Highland National knew of his death, John's sister appeared at the bank. She...
-
On December 31, 2009, Marsh Company held 1,000 shares of X Company common stock in its portfolio of long-term investments in available-for-sale securities. The stock had cost $15 per share and has a...
-
Assuming the same data as given in problem 9, was the well in each case profitable? Discuss your answer. Problem 9:- Property cost (acquisition cost). Drilling cost (one well). Estimated completion...
-
Krauss Leasing Company signs a lease agreement on January 1, 2011, to lease electronic equipment to Stewart Company. The term of the non-cancelable lease is 2 years, and payments are required at the...
-
Explain why there are the same number of tables in the relational model as compared to the number of entities in the logical model.
-
The Tusquittee Company is a retail company that began operations on October 1, 2018, when it incorporated in the state of North Carolina. The Tusquittee Company is authorized to issue 100,000 shares...
-
Bevin borrowed money from three lenders; each loan was evidenced by a promissory note and secured by a deed of trust on Bevin's avocado orchard. Bevin first borrowed $200,000 from Larry, who recorded...
-
A new machine costs $42,000 and has a $2000 salvage value at the end of its 8-year useful life. Determine the straight-line (SL), and the double declining balance (DDB) depreciation schedules for the...
-
Explain the differences between Parametric and Nonparametric methods of testing a hypothesis.
-
You decided to create a new language! Your computer memory has 4 bits, so what is the Maximum number of symbols you can encode in this memory?
-
What do you think about a tax code that even the IRS and CPA's must look outside the IRS Code to find answers? Explain.
-
If the inflation rate is 3% and the nominal interest rate is 8%, what is the real interest rate? Your answer is referred to as the real pre tax interest rate.
-
The following information is taken from the Classic Car Shop records: Accounts receivable, 31 December 2024 $30,000 (debt) 2024 net credit sales 54,000 Accounts deleted as non-collectible through...
-
1. As a general strategy, would you recommend that Carl take an aggressive approach to capacity expansion or more of a wait-and-see approach? 2. Should Carl go with the option for one facility that...
-
If (2Z,4Z)-hexa-2,4-diene were able to undergo a Diels-Alder reaction with methyl propenoate, what would be the products? (Hint: There are four products comprised of two pairs of enantiomers. One...
-
Write a detailed mechanism for the following reaction. Br OH HBr
-
(a) Using reactions studied in this chapter, show steps by which this alkyne could be converted to the seven-membered ring homolog of the product obtained in Problem 7.44(b). (b) Could the homologous...
-
Draw a block diagram for the feedback control of a liquid-level system, which consists of a valve with a control knob (0\%-100\%) and a liquid-level sensor. Clearly label essential components and...
-
A control system is represented using the block diagram shown in Figure 10.59. Sketch the root locus with respect to the proportional control gain K. Determine all the values of \(K\) for which the...
-
Roughly sketch the root locus with respect to \(K\) for the equation of \(1+K L(s)=0\) and the following choices for \(L(s)\). Make sure to give the asymptotes, arrival or departure angles, and...

Study smarter with the SolutionInn App


