e here to search Given the following thermodynamic values, calculate AS for this reaction. 2SO(g) +...
Fantastic news! We've Found the answer you've been seeking!
Question:
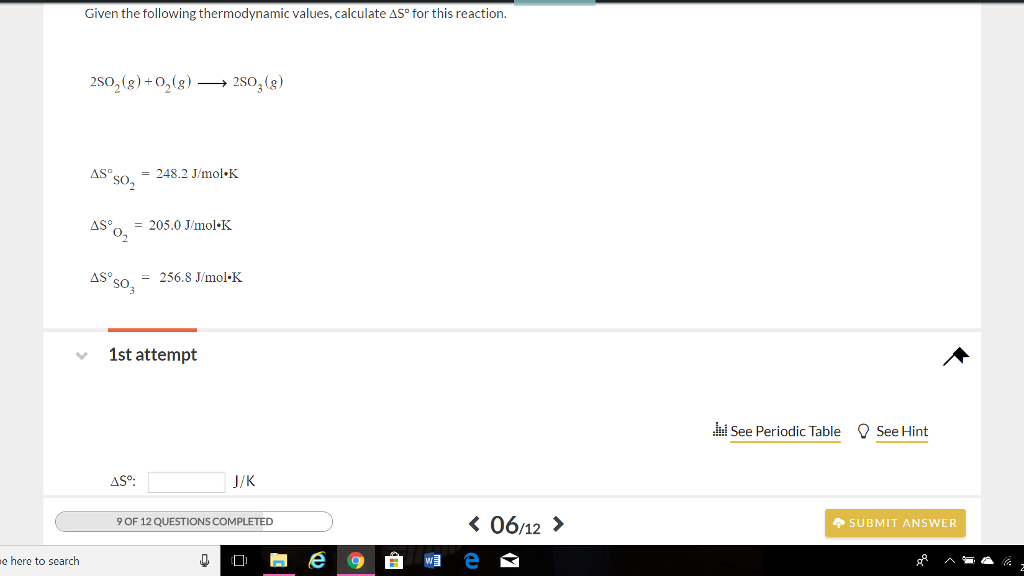
Transcribed Image Text:
e here to search Given the following thermodynamic values, calculate AS° for this reaction. 2SO₂(g) + O₂(g) →→→→ 25O₂ (g) AS S0₂ AS⁹ 0₂ ASOS = 205.0 J/mol K SO₂ = 248.2 J/mol K ASº: = 256.8 J/mol.K 1st attempt J/K 9 OF 12 QUESTIONS COMPLETED ↓ M e < 06/12 > See Periodic Table See Hint SUBMIT ANSWER e here to search Given the following thermodynamic values, calculate AS° for this reaction. 2SO₂(g) + O₂(g) →→→→ 25O₂ (g) AS S0₂ AS⁹ 0₂ ASOS = 205.0 J/mol K SO₂ = 248.2 J/mol K ASº: = 256.8 J/mol.K 1st attempt J/K 9 OF 12 QUESTIONS COMPLETED ↓ M e < 06/12 > See Periodic Table See Hint SUBMIT ANSWER
Expert Answer:
Answer rating: 100% (QA)
We Know That AS reaction Given that 48 AS AS Given balanced chemical equation is as ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Given the following Thermodynamic Quantities at 298.15 K (25 C): Substance AH: (kJ/mol) S (J/K-mol) Cl2 (8) 222.96 P2 (g) PCI3 (g) 144.3 218.1 -288.1 311.7 P2 (g) + 3CI2 (g) 2PCI3 (g) For the...
-
Given the following thermodynamic data, calculate the lattice energy of CaBr2(s). Term Value (kJ/mol) ?H?f[CaBr2(s)] -675 ?H?f[Ca(g)] 179 ?H?f[Br(g)] 112 I1(Ca) 590. I2(Ca) 1145 E(Br) -325 Express...
-
Given that Som = 29.79 J K-I mol-1 for bismuth at 100 K and the following tabulated heat capacities data (D.G. Archer,]. Chem. Eng. Data 40, 1015 (1995)), compute the standard molar entropy of...
-
Featherstone Inc. reported the following data: Net income ................................... $296,000 Depreciation expense ................... 113,100 Gain on disposal of equipment ...... 58,500...
-
Ventris Company found that one of its manufacturing cells had actual cycle time of 15 minutes per unit. The theoretical cycle time for this cell was nine minutes per unit. Required: 1. Calculate the...
-
Refer to the data given in Exercise 7 and assume that the necessary conditions of normality and variance are met. a. Find the predicted size of a household that discards 1.25 kg of plastic. b. Find...
-
How does Porters competitive forces model help companies develop competitive strategies using information systems? Define Porters competitive forces model and explain how it works. Describe what...
-
The supervisor at the Precision Machine Shop wants to determine the staffing policy that minimizes total operating costs. The average arrival rate at the tool crib, where tools are dispensed to the...
-
. Blue, Inc. includes the following account among its trade receivables. Hopkins Co. 1/1 Balance forward 686 1/28 Cash (#1710) 1,078 1/20 Invoice #1710 1,078 4/2 Cash (#2116) 1,323 3/14 Invoice...
-
This and similar cases in later chapters focus on the financial statements of a real company Target Corporation, a discount merchandiser that sells a wide assortment of general merchandise and food....
-
Discuss the various ways in which industrial psychology is applied in industry. Q2. Describe the Hawthorne studies and its contribution to industrial psychology. Q3. Discuss the various Learning...
-
Power Drive Corporation designs and produces a line of golf equipment and golf apparel. Power Drive has 100,000 shares of common stock outstanding as of the beginning of 2024. Power Drive has the...
-
Two 2.2 g point charges on 1.0-m-long threads repel each other after being equally charged, as shown in the figure.(Figure 1) Assume that 0= 26. Figure A 1.0 m. 0 0 1.0 m 1 of 1 Part A What is the...
-
California Surf Clothing Company issues 1,000 shares of $1 par value common stock at $35 per share. Later in the year, the company decides to purchase 100 shares at a cost of $38 per share. Assume...
-
It was May 2017 when Neeraj Kumar, the owner of Kumar Food Corporation (KFC), held a brainstorming session with his marketing and sales team to find ways of increasing the value of his company to...
-
What technology would you use to assure consistent delivery of the training message in every restaurant every time it is given, and why. Assume the maximum training time per topic is 10 minutes and...
-
Explain the concept of the "product life cycle" and its implications for production and marketing strategies.
-
The figure shows six containers, each of which is filled from the top. Assume that water is poured into the containers at a constant rate and each container is filled in 10 seconds. Assume also that...
-
Sketch the molecular orbital and label its type (Ï or p; bonding or antibonding) that would be formed when the following atomic orbitals overlap. Explain your labels. a. b. d.
-
Calculate the kinetic energy possessed by 1.00 1020 molecules of methane gas (CH4) at T = 27oC, assuming ideal behavior.
-
Assign oxidation states to all of the following atoms. a. UO 2 2+ b. As 2 O 3 c. NaBiO 3 d. As 4 e. HAsO 2 f. Mg 2 P 2 O 7 g. Na 2 S 2 O 3 h. Hg 2 Cl 2 i. Ca(NO 3 ) 2
-
4. Jean-Franois, a French wine exporter, sues Bob Joe, a Texas importer, claiming that Bob Joe owes him $2 million for wine. Jean-Franois takes the witness stand to describe how the contract was...
-
5. The Kyrgyz Republic is one of the new nations that broke away from the old Soviet Union. In September 1994, the government of Kyrgyzstan made two independent announcements: (1) it was abolishing...
-
6. The Instituto de Auxilios y Viviendas is a government agency of the Dominican Republic. Dr. Marion Fernandez, the general administrator of the Instituto and Secretary of the Republic, sought a...

Study smarter with the SolutionInn App


