Elemental iridium is composed of two stable isotopes, iridium-191 and iridium-193. (a) How many protons and...
Fantastic news! We've Found the answer you've been seeking!
Question:
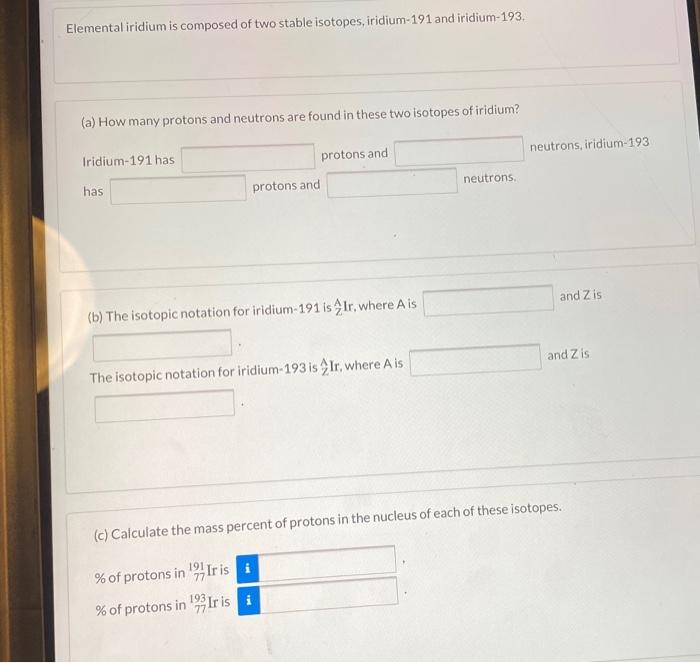
Transcribed Image Text:
Elemental iridium is composed of two stable isotopes, iridium-191 and iridium-193. (a) How many protons and neutrons are found in these two isotopes of iridium? Iridium-191 has has protons and protons and (b) The isotopic notation for iridium-191 is Ir, where A is The isotopic notation for iridium-193 is Ir, where A is 193 neutrons. neutrons, iridium-193 and Z is and Z is (c) Calculate the mass percent of protons in the nucleus of each of these isotopes. % of protons in Iris % of protons in Ir is i Elemental iridium is composed of two stable isotopes, iridium-191 and iridium-193. (a) How many protons and neutrons are found in these two isotopes of iridium? Iridium-191 has has protons and protons and (b) The isotopic notation for iridium-191 is Ir, where A is The isotopic notation for iridium-193 is Ir, where A is 193 neutrons. neutrons, iridium-193 and Z is and Z is (c) Calculate the mass percent of protons in the nucleus of each of these isotopes. % of protons in Iris % of protons in Ir is i
Expert Answer:
Answer rating: 100% (QA)
77 B Ir c 191 193 Ir O A A Ir 2 A Ir 2 Profons 77 Electrons77 Pr... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
How many protons and neutrons are in each isotope? c. 56Fe d. 224Rn EAl
-
How many protons and neutrons are in each isotope? c. 252Es d. 40K 48
-
A cascade system is composed of two ideal refrigeration cycles. The high-temperature cycle uses R-22. Saturated liquid leaves the condenser at 40C, and saturated vapor leaves the heat...
-
Which of the following statements about Erlang are TRUE? Check all that apply All functions in an .erl file are public by default, i.e. callable from the shell or from any other file All parameters...
-
What is the profit-leverage effect of purchasing? What is the return-on-assets effect of purchasing?
-
Wilbur DuVal has an apartment full of antique furniture. A fire in the apartment destroys a large part of his furnishings. The destroyed furnishings had a fair market value of $40,000, and Wilburs...
-
Enter the following in the personal accounts only. Do not write up the other accounts. Balance down each personal account at the end of the month. After completing this, state which of the balances...
-
Brainiac Company purchased a delivery truck for $30,000 on January 1, 2010.The truck has an expected salvage value of $2,000, and is expected to be driven 100,000 miles over its estimated useful life...
-
Surf Company can sell all of the two surfboard models it produces, but it has only 448 direct labor hours available. The Glide model requires 2 direct labor hours per unit. The Ultra model requires 4...
-
Review the methods used by supply chain managers to mitigate SCIS implementation risks.
-
Explain in detail the Wealth Management and key takeaway, which you will implement in your life. Please explain with relevant real life example which you will implement in your life.
-
How do the positions of the SEC and the AICPA differ with respect to the independence of a CPA who performs routine accounting services for a client?
-
The partnership of Smith, Frank \& Clark, a CPA firm, has been the auditor of Greenleaf, Inc., for many years. During the annual audit of the financial statements for the year ended December 31, \(20...
-
Why was the Independence Standards Board formed? What is the source of the board's authority?
-
What are related-party transactions?
-
Criticize the working paper on the next page that you are reviewing as senior auditor on the December 31 audit of Pratt Company.
-
Interest revenue to be recorded by a lessor under a direct financing lease should be
-
Use the graphs of f and g to graph h(x) = (f + g) (x). To print an enlarged copy of the graph, go to MathGraphs.com. 1. 2. y 24 8. 2. -2 -2 4 6
-
Predict the major product for each proposed Diels-Alder reaction. Include stereochemistry where appropriate. (a) (b) (c) Ph 0 Ph
-
Glyptal resin makes a strong, solid polymer matrix for electronic parts. Glyptal is made from terephthalic acid and glycerol. Draw the structure of Glyptal, and explain its remarkable strength and...
-
Draw a structure for each compound (includes old and new names). (a) 3-methylpent-1-ene (b) cis-3-methyl-3-hexene (c) 3,4-dibromobut-1-ene (d) 1,3-cyclohexadiene (e) cycloocta-1,4-diene (f)...
-
B Mann has the following purchases for the month of May 19X8: Required: (a) Enter up the purchases journal for the month. (b) Post the transactions to the suppliers accounts. (c) Transfer the total...
-
A Rowland has the following purchases for the month of June 19X9: Required: (a) Enter up the purchases journal for the month. (b) Post the items to the suppliers accounts. (c) Transfer the total to...
-
C Phillips, a sole trader, has the following purchases and sales for March 19X9: Required: (a) Prepare the purchases and sales journals of C Phillips from the above. (b) Post the items to the...

Study smarter with the SolutionInn App


