Which of the following properties of zinc are chemical properties? Check all that apply. It has...
Fantastic news! We've Found the answer you've been seeking!
Question:
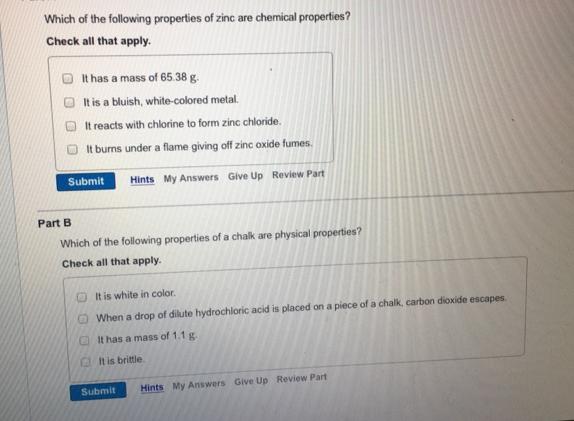
Transcribed Image Text:
Which of the following properties of zinc are chemical properties? Check all that apply. It has a mass of 65.38 g. It is a bluish, white-colored metal. It reacts with chlorine to form zinc chloride. It burns under a flame giving off zinc oxide fumes. Submit Hints My Answers Give Up Review Part Part B Which of the following properties of a chalk are physical properties? Check all that apply. It is white in color. When a drop of dilute hydrochloric acid is placed on a piece of a chalk, carbon dioxide escapes. It has a mass of 1.1 g. It is brittle Submit Hints My Answers Give Up Review Part Which of the following properties of zinc are chemical properties? Check all that apply. It has a mass of 65.38 g. It is a bluish, white-colored metal. It reacts with chlorine to form zinc chloride. It burns under a flame giving off zinc oxide fumes. Submit Hints My Answers Give Up Review Part Part B Which of the following properties of a chalk are physical properties? Check all that apply. It is white in color. When a drop of dilute hydrochloric acid is placed on a piece of a chalk, carbon dioxide escapes. It has a mass of 1.1 g. It is brittle Submit Hints My Answers Give Up Review Part
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below 1 The following properties of zinc are ... View the full answer

Related Book For 

Equity Asset Valuation
ISBN: 978-0470571439
2nd Edition
Authors: Jerald E. Pinto, Elaine Henry, Thomas R. Robinson, John D. Stowe, Abby Cohen
Posted Date:
Students also viewed these chemistry questions
-
Which of the following properties indicates very strong intermolecular forces in a liquid? (a) Very low surface tension, (b) Very low critical temperature, (c) Very low boiling point, (d) Very low...
-
A flask containing dilute hydrochloric acid was placed on a digital balance. An excess of limestone chippings was added to this acid, a plug of cotton wool was placed in the neck of the flask and the...
-
When resistors are connected in series, which of the following would be the same for each resistor: potential difference, current, power?
-
Solve the inequality. Write the solution in interval notation. |15 = x < 7
-
Toledo Mud Hens Baseball Club, Inc., is a not-for-profit organization that is a separate legal entity and can be sued in its own right. The Toledo Mud Hens are probably the most famous team in all of...
-
Give an example of three types of different levels of costs in a customer cost hierarchy.
-
Apply the central difference formula (4.10) to approximate the first derivatives of the functions \(u_{1}(x)=\sin (x), u_{2}(x)=\sin (3 x)\), \(u_{3}(x)=\sin (10 x)\) at \(x=0\). Use the uniform grid...
-
What is the internal rate of return on the following projects, each of which requires a $20,000 cash outlay now and returns the cash flows indicated? a. $10,426.72 at the end of Years 1 and 2. b....
-
ForCo, a compnay incorporated in foreign country F, sells widgets in the United States via mail order. In New York, Chicago, and Los Angeles. ForCo has facilities at prestigious real estate locati...
-
This is the code followed by the questions :) // Execute in strict mode to prevent some common mistakes "use strict"; /** * Make diary data item * @param type Type of item to create, either "text" or...
-
https://youtu.be/TbpZMSCx7uk. Watch the Week 5 Discussion Video. The information presented in the video will prepare you to answer your reply prompts. 2. Initial Post: Create a new thread and answer...
-
Visit the webpage of three of the marketing research firms listed in Table 1.1 in Chapter 1. What types of research designs have been implemented recently by these firms?
-
Define common-base current gain and common-emitter current gain.
-
Visit the website of the U.S. Census Bureau (www.census.gov). Write a report about the secondary data available from the bureau that would be useful to a fast-food firm such as McDonald's for the...
-
Define the conditions for cutoff, forward-active mode, and saturation mode for a pnp bipolar transistor.
-
Based on the following declarations, tell whether each statement below is syntactically legal (yes) or illegal (no). int* p; int* q; int* r; int a; int b; int c; int* p; int* q: int* r; int a; int b;...
-
Give a typical heading for the statement of cost of goods manufactured. Three examples needed.
-
AB CORPORATION ISSUED THE FOLLOWING 850 COMMON STOCKS PAR VALUE P100 750 PARTICIPATING PREFERRED STOCKS PAR VALUE P100 AT 3% AB CORPORATION DECLARED P100,000.00 DIVIDEND IN 2022.
-
The text defined intrinsic value as the value of an asset given a hypothetically complete understanding of the assets investment characteristics. Discuss why hypothetically is included in the...
-
Watson Dunn is planning to value BCC Corporation, a provider of a variety of industrial metals and minerals. Dunn uses a single - stage FCFF approach. The financial information Dunn has assembled for...
-
An analyst plans to use P/E and the method of comparables as a basis for recommending purchasing shares of one of two peer - group companies in the business of manufacturing personal digital...
-
1. ETHICS Lee McNeely told Hardee's officials that he was interested in purchasing multiple restaurants in Arkansas. A Hardee's officer assured him that any of the company-owned stores in Arkansas...
-
Question: In which one or more of the following forms of organization is it true that none of the partners are liable for the debts of the partnership? A. General partnership B. Limited liability...
-
2. CPA QUESTION: Assuming all other requirements are met, a corporation may elect to be treated as an S corporation under the Internal Revenue Code if it has: (a) Both common and preferred...

Study smarter with the SolutionInn App


