Esters (i) aliphatic: RCOR' M+ weak but observable note: M+ gets less intense as the alcohol...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
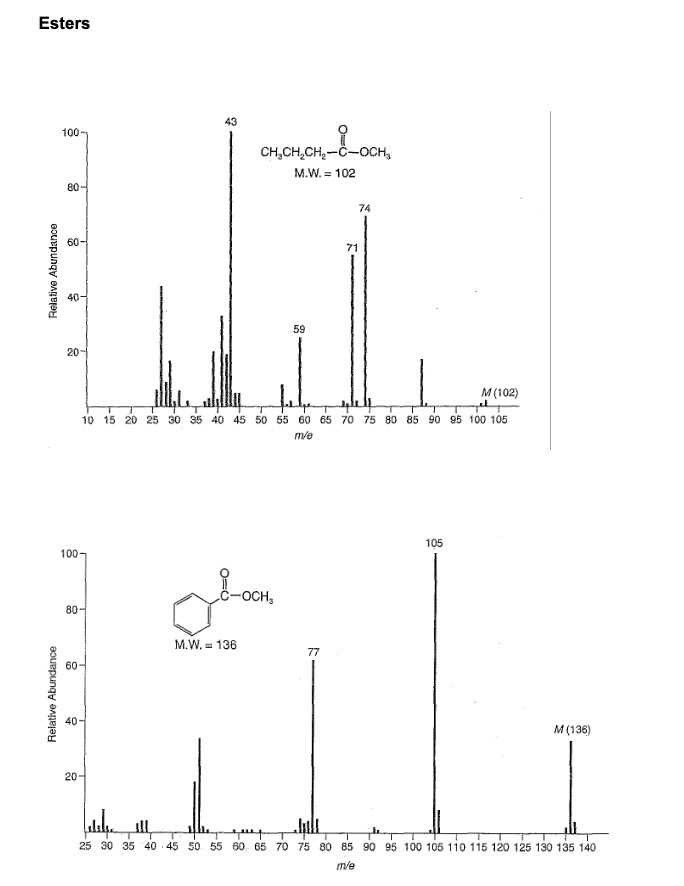
Esters (i) aliphatic: RCO₂R' M+ weak but observable note: M+ gets less intense as the alcohol portion ( ) increases in size: e.g. C4 very weak. M-R M - OR' 0+ R-C-O-R Ö+ R-C-O-R' Which peaks in the MS of methyl butanoate and methyl benzoate are due to these types of fragmentation ? Account for the peak at m/z = 74 in the methyl butanoate MS. Esters Relative Abundance Relative Abundance 100 80 60- 40- 20 100- 80- 60 40 43 20- CH₂CH₂CH₂-C-OCH₂ M.W. = 102 10 15 20 25 30 35 40 45 50 55 60 65 70 75 80 85 90 95 100 105 m/e 3-com. M.W. 136 59 71 77 74 M (102) 105 M (136) elekea ill T quep T 25 30 35 40 45 50 55 60 65 70 75 80 85 90 95 100 105 110 115 120 125 130 135 140 m/e Esters (i) aliphatic: RCO₂R' M+ weak but observable note: M+ gets less intense as the alcohol portion ( ) increases in size: e.g. C4 very weak. M-R M - OR' 0+ R-C-O-R Ö+ R-C-O-R' Which peaks in the MS of methyl butanoate and methyl benzoate are due to these types of fragmentation ? Account for the peak at m/z = 74 in the methyl butanoate MS. Esters Relative Abundance Relative Abundance 100 80 60- 40- 20 100- 80- 60 40 43 20- CH₂CH₂CH₂-C-OCH₂ M.W. = 102 10 15 20 25 30 35 40 45 50 55 60 65 70 75 80 85 90 95 100 105 m/e 3-com. M.W. 136 59 71 77 74 M (102) 105 M (136) elekea ill T quep T 25 30 35 40 45 50 55 60 65 70 75 80 85 90 95 100 105 110 115 120 125 130 135 140 m/e
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
I have 4 lbm of liquid water at 70 F, 15 psia. I now add 20 Btu of energy at a constant pressure. How hot does it get if it is heated? How fast does it move if it is pushed by a constant horizontal...
-
The base peak appears at m/z 105 for one of the following compounds and at m/z 119 for the other two. Match the compounds with the appropriate m/z values for their base peaks.
-
For a distortion less line with Z0 50 , a 20 (mNp/m), up 2.5 x 108 (m/s), find the line parameters and at 100 MHz.
-
The accounting records of Shumway Ag Implement show the following data. Determine the cost of goods sold during the period under a periodic inventory system using (a) the FIFO method, (b) the LIFO...
-
In 2015, Thom Inc. discovered an error in its 2012 financial statements. The firm recorded $ 8,500,000 of depreciation expense on its equipment instead of recording $ 9,500,000. Thom has a constant...
-
Consider the three sets from = LETTERS: A = {U, X, M, J, B, D}, B = {N, J, H, C, G, X}, and C = {H, V, N, K,D, F}. Using the operators intersect and union, for the sets A, B, and C, verify the...
-
In late 2004, a pain medication called Vioxx was removed voluntarily from the market by its manufacturer, Merck, Inc., due to a risk of heart-related problems. Merck lost a great deal of income and...
-
List six drawbacks of using only subjective information in formulating strategies.
-
Question 2 (b) (i)TS acquired an item of plant at a cost of $800 000 on 1 April2014. The plant had an estimated residual value of $50 000, and anestimated life of five years, neither of which has c 1...
-
Reported in the table below are estimation results for three residential housing price models. The dependent variable is defined as home sales price measured in thousands of dollars. The models...
-
What would this filter be in mindbridge software? 0 Example 2: Create a ?lter to identify transactions that trigger the 'reporting adjustment' control point. Please do this across the population. ...
-
When valuing securities with embedded options, why is it important to consider volatility explicitly?
-
What does the volatility parameter in an interest rate model represent?
-
Explain how the value of an embedded call option is determined.
-
What accounts are most important when forecasting a new ventures balance sheet? Which of these lend themselves to using yardsticks and which are better estimated based on fundamental analysis?
-
How is the interest rate tree rendered arbitrage-free?
-
Question 4 of b Current Attempt in Progress Grete Clark formed a dog grooming and training business called Grete Kanines on September 1, 2024. After consulting with a friend who had taken...
-
In a large midwestern university, 30% of the students live in apartments. If 200 students are randomly selected, find the probability that the number of them living in apartments will be between 55...
-
Expand the following expressions, using the rules for summation within parentheses and for binomial expansion (Numerals are always constants; thus, N is always a constant): a. (1 + N) __________ b....
-
Indicate the first letter (N, 0, I, R) of the highest possible scale for each of the following measures, where N is lowest and R is highest: Measure Highest Scale a. Type of disease b. Business...
-
Below are the arithmetic test scores of Ms. Teachwell's fourth-grade class. The statewide average on this same test for all fourth graders is 77.0. The statewide standard deviation is 7.0. You want...
-
Why are Cynthia Cooper and Scott Sullivan significant in detecting financial reporting fraud?
-
Why might management be interested in forensic accounting services?
-
What is a red flag with respect to earnings?

Study smarter with the SolutionInn App



