Estos edificios que estn ah son salones de clase y laboratorios para las clases de...
Fantastic news! We've Found the answer you've been seeking!
Question:
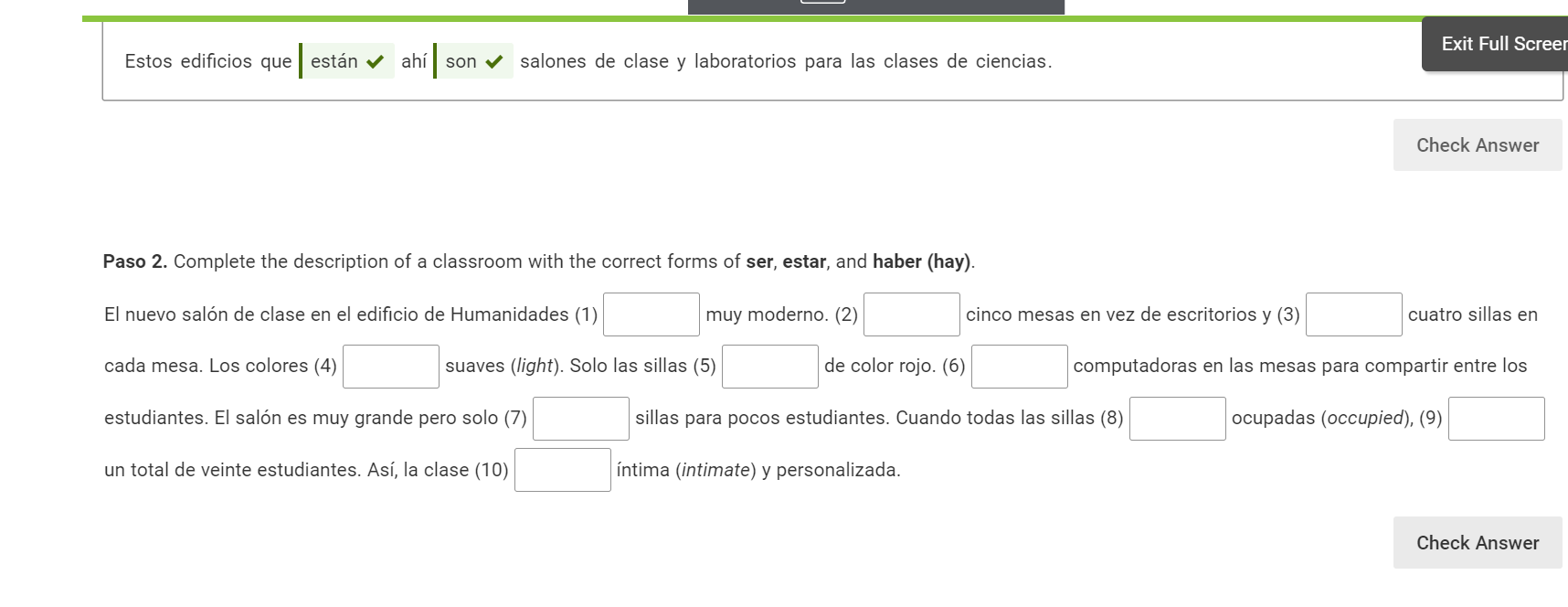
Transcribed Image Text:
Estos edificios que están ahí son ✔ salones de clase y laboratorios para las clases de ciencias. Paso 2. Complete the description of a classroom with the correct forms of ser, estar, and haber (hay). muy moderno. (2) El nuevo salón de clase en el edificio de Humanidades (1) cada mesa. Los colores (4) estudiantes. El salón es muy grande pero solo (7) un total de veinte estudiantes. Así, la clase (10) cinco mesas en vez de escritorios y (3) suaves (light). Solo las sillas (5) de color rojo. (6) sillas para pocos estudiantes. Cuando todas las sillas (8) íntima (intimate) y personalizada. Exit Full Screer Check Answer cuatro sillas en computadoras en las mesas para compartir entre los ocupadas (occupied), (9) Check Answer Estos edificios que están ahí son ✔ salones de clase y laboratorios para las clases de ciencias. Paso 2. Complete the description of a classroom with the correct forms of ser, estar, and haber (hay). muy moderno. (2) El nuevo salón de clase en el edificio de Humanidades (1) cada mesa. Los colores (4) estudiantes. El salón es muy grande pero solo (7) un total de veinte estudiantes. Así, la clase (10) cinco mesas en vez de escritorios y (3) suaves (light). Solo las sillas (5) de color rojo. (6) sillas para pocos estudiantes. Cuando todas las sillas (8) íntima (intimate) y personalizada. Exit Full Screer Check Answer cuatro sillas en computadoras en las mesas para compartir entre los ocupadas (occupied), (9) Check Answer
Expert Answer:
Answer rating: 100% (QA)
k Answer 1 hay 2 Hay 3 Hay 4 son 5 son 6 rojo 7 hay 8 estn 9 hay 10 es Explanatio... View the full answer

Related Book For 

College Mathematics for Business Economics Life Sciences and Social Sciences
ISBN: 978-0321614001
12th edition
Authors: Raymond A. Barnett, Michael R. Ziegler, Karl E. Byleen
Posted Date:
Students also viewed these psychology questions
-
5 non face cards? Refer to the description of a standard deck of 52 cards and Figure 4 on page 363. An experiment consists of dealing 5 cards from a standard 52-card deck. In Problem, what is the...
-
A six or club Refer to the description of a standard deck of 52 cards and Figure 4 on page 363. An experiment consists of drawing 1 card from a standard 52-card deck. In Problem, what is the...
-
5 hearts? Refer to the description of a standard deck of 52 cards and Figure 4 on page 363. An experiment consists of dealing 5 cards from a standard 52-card deck. In Problem, what is the probability...
-
If there is a decrease in the demand for Canadian dollars relative to U.S. dollars, a. The price and quantity of Canadian dollars traded will fall. b. The price and quantity of Canadian dollars will...
-
Why would a client want to issue OCBOA financial statements when it is specifically noted that they are not prepared in accordance with GAAP?
-
For the fully differential circuit in Fig. 12.32 a, assume the op amp is ideal with R i = , R o = 0, a dm = , and a cm = 0. Find the closed-loop gains A dm = v od /v sd , A cm = v oc /v sc , A dmcm =...
-
If the row variable and column variable are interchanged, how are the expected frequencies affected? Is the value of the chi-square statistic affected? Is the number of degrees of freedom affected?
-
Read the following description of Drea Tech Company and identify the elements of inherent risk associated with the revenue cycle. Determine the appropriate audit response (audit procedure) to address...
-
Product Decisions Under Bottlenecked Operations Youngstown Glass Company manufactures three types of safety plate glass: large, medium, and small. All three products have high demand. Thus,...
-
Aaron Rivers, CPA, is auditing the financial statements of Charger Company, a client for the past five years. During past audits of Charger, Rivers identified some immaterial misstatements (most of...
-
In a new subdivision, six homes cost $60000, eight homes cost $80000, and six homes cost $110000. What is the sum of the weighted average and the mode of these homes' costs?
-
calculate the compound amount and compound interest for the following investment. Click here for Table 11-1 . Round your answers to the nearest cent. Principal Time Period (years) Nominal Rate (%)...
-
What is Media? And describe how Media can adding value and sentiment to society.
-
6-1 YIELD CURVES Assume that yields on U.S. Treasury securities were as follows: Rate Term 6 months 4.69% 5.49 1 year 5.66 2 years 5.71 3 years 5.89 4 years 6.05 5 years 10 years 6.12 20 years 6.64...
-
An investment manager collected historical data of the market over the past 5 years and summarised the results in the table below. He is going to make some investment decision base on the following...
-
c. Growth Company has existing debt issued three years ago with a coupon rate of 5.7%. The firm just issued new debt at par with a coupon rate of 6.9%. What is Growth Company's cost of debt? (Select...
-
Recently, the FASB and International Accounting Standards Board (IASB) have been working on several projects in order to harmonize the accounting rules between GAAP and IFRS. This project requires...
-
I frequently use NY Times and CNN and am aware of Fox News but I never use it. I visit these sites, NY Times and CNN, a few times a week whenever I have to research something or see something on...
-
Interpret the results of Problem 22 with both a graph and a description of the graph.
-
(A) If A and B are disjoint, then n(AB)=n(A) + n(B). (B) If n(AUB)=n(A) + n(B),then A and Bare disjoint. In Problem, discuss the validity of each statement. If the statement is always true, explain...
-
In Problem, use the price-demand equation to find the values of p for which demand is elastic and the values for which demand is inelastic. Assume that price and demand are both positive. x = f(p) =...
-
The heat of vaporization of ether is \(25.98 \mathrm{~kJ} / \mathrm{mol}\) at its boiling point, \(34.5^{\circ} \mathrm{C}\). (a) Calculate the rate of change of vapour pressure with temperature...
-
Show that the internal energy of an ideal gas is a function only of temperature.
-
For a gas obeying the equation of state \(V=B+\frac{R T}{P}\), the Joule-Thomson coefficient is given by \[ \mu_{\mathrm{JT}}=-\frac{1}{C_{P}}\left(T \frac{d B}{d T}-B ight) \]

Study smarter with the SolutionInn App


