Exercise 3-5 (Algo) Balance sheet preparation [LO3-2, 3-3] The following are the ending balances of accounts...
Fantastic news! We've Found the answer you've been seeking!
Question:
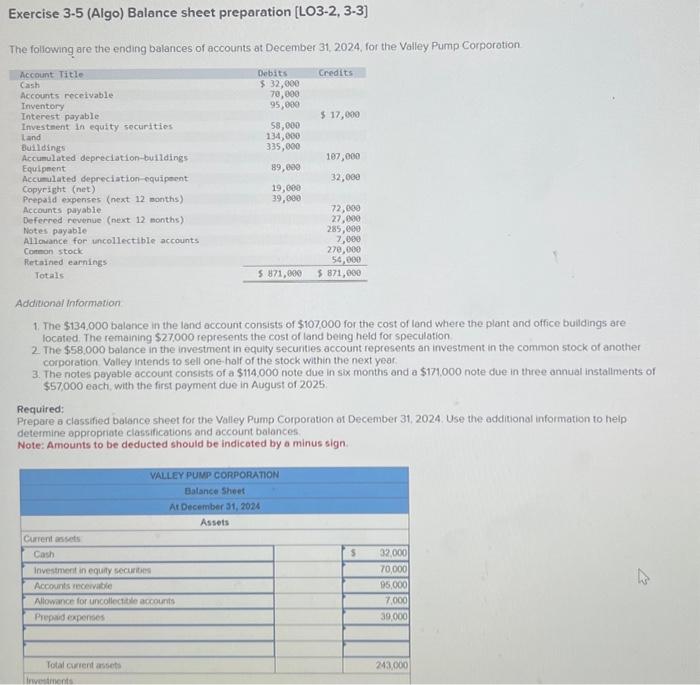
Transcribed Image Text:
Exercise 3-5 (Algo) Balance sheet preparation [LO3-2, 3-3] The following are the ending balances of accounts at December 31, 2024, for the Valley Pump Corporation Account Title Cash Accounts receivable Inventory Interest payable Investment in equity securities Land Buildings Accumulated depreciation-buildings Equipment Accumulated depreciation equipment Copyright (net) Prepaid expenses (next 12 months) Accounts payable Deferred revenue (next 12 months) Notes payable Allowance for uncollectible accounts Common stock Retained earnings Totals Current assets Cash Debits $ 32,000 70,000 95,000 Investment in equity securities Accounts receivable Allowance for uncollectible accounts Prepaid expenses Total current assets 58,000 134,000 335,000 89,000 19,000 39,000 Investments Credits Additional Information 1. The $134,000 balance in the land account consists of $107,000 for the cost of land where the plant and office buildings are located. The remaining $27,000 represents the cost of land being held for speculation. 2. The $58,000 balance in the investment in equity securities account represents an investment in the common stock of another corporation Valley intends to sell one-half of the stock within the next year. 3. The notes payable account consists of a $114,000 note due in six months and a $171,000 note due in three annual installments of $57,000 each, with the first payment due in August of 2025. $ 17,000 Required: Prepare a classified balance sheet for the Valley Pump Corporation of December 31, 2024. Use the additional information to help determine appropriate classifications and account balances. Note: Amounts to be deducted should be indicated by a minus sign.. VALLEY PUMP CORPORATION Balance Sheet At December 31, 2024 Assets 107,000 32,000 72,000 27,000 285,000 7,000 270,000 54,000 $ 871,000 $ 871,000 S 32,000 70,000 95,000 7,000 39,000 243,000 4 Exercise 3-5 (Algo) Balance sheet preparation [LO3-2, 3-3] The following are the ending balances of accounts at December 31, 2024, for the Valley Pump Corporation Account Title Cash Accounts receivable Inventory Interest payable Investment in equity securities Land Buildings Accumulated depreciation-buildings Equipment Accumulated depreciation equipment Copyright (net) Prepaid expenses (next 12 months) Accounts payable Deferred revenue (next 12 months) Notes payable Allowance for uncollectible accounts Common stock Retained earnings Totals Current assets Cash Debits $ 32,000 70,000 95,000 Investment in equity securities Accounts receivable Allowance for uncollectible accounts Prepaid expenses Total current assets 58,000 134,000 335,000 89,000 19,000 39,000 Investments Credits Additional Information 1. The $134,000 balance in the land account consists of $107,000 for the cost of land where the plant and office buildings are located. The remaining $27,000 represents the cost of land being held for speculation. 2. The $58,000 balance in the investment in equity securities account represents an investment in the common stock of another corporation Valley intends to sell one-half of the stock within the next year. 3. The notes payable account consists of a $114,000 note due in six months and a $171,000 note due in three annual installments of $57,000 each, with the first payment due in August of 2025. $ 17,000 Required: Prepare a classified balance sheet for the Valley Pump Corporation of December 31, 2024. Use the additional information to help determine appropriate classifications and account balances. Note: Amounts to be deducted should be indicated by a minus sign.. VALLEY PUMP CORPORATION Balance Sheet At December 31, 2024 Assets 107,000 32,000 72,000 27,000 285,000 7,000 270,000 54,000 $ 871,000 $ 871,000 S 32,000 70,000 95,000 7,000 39,000 243,000 4
Expert Answer:
Answer rating: 100% (QA)
A classified balance sheet for the Valley Pump Corporation at December 31 2024 Valley Pump Corporati... View the full answer

Related Book For 

Posted Date:
Students also viewed these finance questions
-
The following are the ending balances of accounts at December 31, 2024, for the Valley Pump Corporation. Additional Information: 1. The $120,000 balance in the land account consists of $100,000 for...
-
Caswell Pharmaceutical has a shareholders' equity account as shown below. The firm's ordinary shares currently sell for $20 per share. Preference shares $ 500,000 Ordinary shares (2,000,000 shares) $...
-
Your client has offered a 5-year, $1,000 par value bond with a 10 percent coupon. Interest on this bond is paid quarterly. 1) If your client is to earn a nominal rate of return of 12 percent,...
-
Ross Company implemented a quality improvement program and tracked the following for the five years: By cost category as a percentage of sales for the same period of time: Required: 1. Prepare a bar...
-
Hiatt Hurricane Preparedness Corporation accounts for its inventory using LIFO and is applies the LCM rule. The company has compiled the following information regarding the cost, replacement cost,...
-
Refer to Exercise 9. Can you reject H0 at the = 0.01 level? State a conclusion. Exercise 9 Two suppliers of machine parts delivered large shipments. A simple random sample of 150 parts was chosen...
-
Starbucks is hoping to make use of its excess restaurant capacity in the evenings by experimenting with selling beer and wine. It speculates that the only additional costs are hiring more of the same...
-
Choice Bank's stock portfolio has a market value of $500 million. The beta of the portfolio approximates the market portfolio, whose standard deviation (s m ) has been estimated at 2.25 per cent....
-
George and Harry Haygood are building contractors who specialize in the construction of private home dwellings, storage warehouses, and small businesses (less than 20,000 sq. ft. of floor space)....
-
Which of the following can form hydrogen bonds with water? a) CH3OCH3 b) CH4 c) F2 d) HCOOH e) Na
-
Fill in the Blank. The equations of motion of a multi-degree-of-freedom system can be expressed in terms of _________ coefficients.
-
Fill in the Blank. The stiffness matrix of a semidefinite system is _________ .
-
Fill in the Blank. The value of the Kronecker delta \(\left(\delta_{i j} ight)\) is 1 for \(i=j\) and _________ for \(i eq j\).
-
Fill in the Blank. Lagrange's equations are expressed in terms of _________ coordinates.
-
Fill in the Blank. The representation of an arbitrary \(n\)-dimensional vector as a linear combination of \(n\)-linearly independent vectors is known as _________ theorem.
-
Consider a three-stage game: First, the incumbent may sign a quantity contract with consumers A contract commits the incumbent to a quantity qfix (but not a price) - Incumbent must pay a legal cost...
-
Wal-Mart is the second largest retailer in the world. The data file on the disk holds monthly data on Wal-Marts revenue, along with several possibly related economic variables. a) Using computer...
-
On May 1, 2024, Hecala Mining entered into an agreement with the state of New Mexico to obtain the rights to operate a mineral mine in New Mexico for $10 million. Additional costs and purchases...
-
Aquatic Equipment Corporation decided to switch from the LIFO method of costing inventories to the FIFO method at the beginning of 2024. The inventory as reported at the end of 2023 using LIFO would...
-
Note: The loss included in NI equals the difference between the proceeds ($30 million) and the carrying value of the investment ($35 million). An additional $15 million was recognized in net income...
-
The following reaction takes place in a system consisting of \(3 \mathrm{~mol} \mathrm{CH}_{4}, 5 \mathrm{~mol} \mathrm{H}_{2} \mathrm{O}\), \(1 \mathrm{~mol} \mathrm{CO}\) and \(4 \mathrm{~mol}...
-
A system is said to be in thermodynamic equilibrium if its (a) Temperature remains unchanged (b) Pressure remains unchanged (c) Chemical potential remains unchanged (d) Temperature, pressure and...
-
A system initially containing \(3 \mathrm{~mol} \mathrm{CO}_{2}, 5 \mathrm{~mol} \mathrm{H}_{2}\) and \(1 \mathrm{~mol} \mathrm{H}_{2} \mathrm{O}\) is undergoing the following reactions: \[...

Study smarter with the SolutionInn App


